Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
Type III secretion system
The type III secretion system (T3SS or TTSS) is one of the bacterial secretion systems used by bacteria to secrete their effector proteins into the host's cells to promote virulence and colonisation. While the type III secretion system has been widely regarded as equivalent to the injectisome, many argue that the injectisome is only part of the type III secretion system, which also include structures like the flagellar export apparatus. The T3SS is a needle-like protein complex found in several species of pathogenic gram-negative bacteria.
The term 'Type III secretion system' was coined in 1993. This secretion system is distinguished from at least five other secretion systems found in gram-negative bacteria. Many animal and plant associated bacteria possess similar T3SSs. These T3SSs are similar as a result of convergent evolution and phylogenetic analysis supports a model in which gram-negative bacteria can transfer the T3SS gene cassette horizontally to other species. Some of the most researched T3SSs are from species of:[citation needed]
The T3SS is composed of approximately 30 different proteins, making it one of the most complex secretion systems. Its structure shows many similarities with bacterial flagella (long, rigid, extracellular structures used for motility). Some of the proteins participating in T3SS share amino-acid sequence homology to flagellar proteins. Some of the bacteria possessing a T3SS have flagella as well and are motile (Salmonella, for instance), and some do not (Shigella, for instance). Technically speaking, type III secretion is used both for secreting infection-related proteins and flagellar components. However, the term "type III secretion" is used mainly in relation to the infection apparatus. The bacterial flagellum shares a common ancestor with the type III secretion system.
T3SSs are essential for the pathogenicity (the ability to infect) of many pathogenic bacteria. Defects in the T3SS may render a bacterium non-pathogenic. It has been suggested that some non-invasive strains of gram-negative bacteria have lost the T3SS because the energetically costly system is no longer of use. Although traditional antibiotics were effective against these bacteria in the past, antibiotic-resistant strains constantly emerge. Understanding the way the T3SS works and developing drugs targeting it specifically have become an important goal of many research groups around the world since the late 1990s.
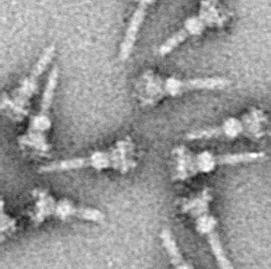
The hallmark of T3SS is the needle (more generally, the needle complex (NC) or the T3SS apparatus (T3SA); also called injectisome when the ATPase is excluded; see below). Bacterial proteins that need to be secreted pass from the bacterial cytoplasm through the needle directly into the host cytoplasm. Three membranes separate the two cytoplasms: the double membranes (inner and outer membranes) of the Gram-negative bacterium and the eukaryotic membrane. The needle provides a smooth passage through those highly selective and almost impermeable membranes. A single bacterium can have several hundred needle complexes spread across its membrane. It has been proposed that the needle complex is a universal feature of all T3SSs of pathogenic bacteria.
The needle complex starts at the cytoplasm of the bacterium, crosses the two membranes and protrudes from the cell. The part anchored in the membrane is the base (or basal body) of the T3SS. The extracellular part is the needle. A so-called inner rod connects the needle to the base. The needle itself, although the biggest and most prominent part of the T3SS, is made out of many units of a single protein. The majority of the different T3SS proteins are therefore those that build the base and those that are secreted into the host. As mentioned above, the needle complex shares similarities with bacterial flagella. More specifically, the base of the needle complex is structurally very similar to the flagellar base; the needle itself is analogous to the flagellar hook, a structure connecting the base to the flagellar filament.
The base is composed of several circular rings and is the first structure that is built in a new needle complex. Once the base is completed, it serves as a secretion machine for the outer proteins (the needle). Once the whole complex is completed the system switches to secreting proteins that are intended to be delivered into host cells. The needle is presumed to be built from bottom to top; units of needle monomer protein pile upon each other, so that the unit at the tip of the needle is the last one added. The needle subunit is one of the smallest T3SS proteins, measuring at around 9 kDa. 100−150 subunits comprise each needle.
The T3SS needle measures around 60−80 nm in length and 8 nm in external width. It needs to have a minimal length so that other extracellular bacterial structures (adhesins and the lipopolysaccharide layer, for instance) do not interfere with secretion. The hole of the needle has a 3 nm diameter. Most folded effector proteins are too large to pass through the needle opening, so most secreted proteins must pass through the needle unfolded, a task carried out by the ATPase at the base of the structure.
Hub AI
Type III secretion system AI simulator
(@Type III secretion system_simulator)
Type III secretion system
The type III secretion system (T3SS or TTSS) is one of the bacterial secretion systems used by bacteria to secrete their effector proteins into the host's cells to promote virulence and colonisation. While the type III secretion system has been widely regarded as equivalent to the injectisome, many argue that the injectisome is only part of the type III secretion system, which also include structures like the flagellar export apparatus. The T3SS is a needle-like protein complex found in several species of pathogenic gram-negative bacteria.
The term 'Type III secretion system' was coined in 1993. This secretion system is distinguished from at least five other secretion systems found in gram-negative bacteria. Many animal and plant associated bacteria possess similar T3SSs. These T3SSs are similar as a result of convergent evolution and phylogenetic analysis supports a model in which gram-negative bacteria can transfer the T3SS gene cassette horizontally to other species. Some of the most researched T3SSs are from species of:[citation needed]
The T3SS is composed of approximately 30 different proteins, making it one of the most complex secretion systems. Its structure shows many similarities with bacterial flagella (long, rigid, extracellular structures used for motility). Some of the proteins participating in T3SS share amino-acid sequence homology to flagellar proteins. Some of the bacteria possessing a T3SS have flagella as well and are motile (Salmonella, for instance), and some do not (Shigella, for instance). Technically speaking, type III secretion is used both for secreting infection-related proteins and flagellar components. However, the term "type III secretion" is used mainly in relation to the infection apparatus. The bacterial flagellum shares a common ancestor with the type III secretion system.
T3SSs are essential for the pathogenicity (the ability to infect) of many pathogenic bacteria. Defects in the T3SS may render a bacterium non-pathogenic. It has been suggested that some non-invasive strains of gram-negative bacteria have lost the T3SS because the energetically costly system is no longer of use. Although traditional antibiotics were effective against these bacteria in the past, antibiotic-resistant strains constantly emerge. Understanding the way the T3SS works and developing drugs targeting it specifically have become an important goal of many research groups around the world since the late 1990s.
The hallmark of T3SS is the needle (more generally, the needle complex (NC) or the T3SS apparatus (T3SA); also called injectisome when the ATPase is excluded; see below). Bacterial proteins that need to be secreted pass from the bacterial cytoplasm through the needle directly into the host cytoplasm. Three membranes separate the two cytoplasms: the double membranes (inner and outer membranes) of the Gram-negative bacterium and the eukaryotic membrane. The needle provides a smooth passage through those highly selective and almost impermeable membranes. A single bacterium can have several hundred needle complexes spread across its membrane. It has been proposed that the needle complex is a universal feature of all T3SSs of pathogenic bacteria.
The needle complex starts at the cytoplasm of the bacterium, crosses the two membranes and protrudes from the cell. The part anchored in the membrane is the base (or basal body) of the T3SS. The extracellular part is the needle. A so-called inner rod connects the needle to the base. The needle itself, although the biggest and most prominent part of the T3SS, is made out of many units of a single protein. The majority of the different T3SS proteins are therefore those that build the base and those that are secreted into the host. As mentioned above, the needle complex shares similarities with bacterial flagella. More specifically, the base of the needle complex is structurally very similar to the flagellar base; the needle itself is analogous to the flagellar hook, a structure connecting the base to the flagellar filament.
The base is composed of several circular rings and is the first structure that is built in a new needle complex. Once the base is completed, it serves as a secretion machine for the outer proteins (the needle). Once the whole complex is completed the system switches to secreting proteins that are intended to be delivered into host cells. The needle is presumed to be built from bottom to top; units of needle monomer protein pile upon each other, so that the unit at the tip of the needle is the last one added. The needle subunit is one of the smallest T3SS proteins, measuring at around 9 kDa. 100−150 subunits comprise each needle.
The T3SS needle measures around 60−80 nm in length and 8 nm in external width. It needs to have a minimal length so that other extracellular bacterial structures (adhesins and the lipopolysaccharide layer, for instance) do not interfere with secretion. The hole of the needle has a 3 nm diameter. Most folded effector proteins are too large to pass through the needle opening, so most secreted proteins must pass through the needle unfolded, a task carried out by the ATPase at the base of the structure.