Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
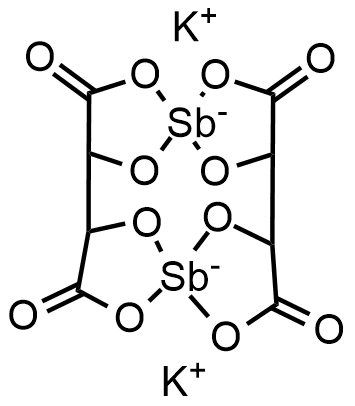
Antimony potassium tartrate
Antimony potassium tartrate, also known as potassium antimonyl tartrate, potassium antimontarterate, or tartar emetic, has the formula K2Sb2(C4H2O6)2. The compound has long been known as a powerful emetic, and was used in the treatment of schistosomiasis and leishmaniasis. It is used as a resolving agent. It typically is obtained as a hydrate.
The first treatment application against trypanosomiasis was tested in 1906, and the compound's use to treat other tropical diseases was researched. The treatment of leishmania with antimony potassium tartrate started in 1913. After the introduction of antimony(V) containing complexes like sodium stibogluconate and meglumine antimoniate, the use of antimony potassium tartrate was phased out. After British physician John Brian Christopherson's discovery in 1918 that antimony potassium tartrate could cure schistosomiasis, the antimonial drugs became widely used. However, the injection of antimony potassium tartrate had severe side effects such as Adams–Stokes syndrome and therefore alternative substances were under investigation. With the introduction and subsequent larger use of praziquantel in the 1970s, antimony-based treatments fell out of use.
Tartar emetic was used in the late 19th and early 20th century in patent medicine as a remedy for alcohol intoxication, and was first ruled ineffective in the United States in 1941, in United States v. 11 1/4 Dozen Packages of Articles Labeled in Part Mrs. Moffat's Shoo-Fly Powders for Drunkenness.
The New England Journal of Medicine reported a case study of a patient whose wife secretly gave him a dose of a product called "tartaro emetico" which contained trivalent antimony (antimony potassium tartrate) and is sold in Central America as an aversive treatment for alcohol use disorder. The patient, who had been out drinking the night before, developed persistent vomiting shortly after being given orange juice with the drug. When admitted to the hospital, and later in the intensive care unit, he experienced severe chest pains, cardiac abnormalities, renal and hepatic toxicity, and nearly died. The Journal reports that "Two years later, he [the patient] reports complete abstinence from alcohol."
Antimony potassium tartrate's potential as an emetic has been known since the Middle Ages. The compound itself was considered toxic and therefore a different way to administer it was found. Cups made from pure antimony were used to store wine for 24 hours and then the resulting solution of antimony potassium tartrate in wine was consumed in small portions until the wanted emetic effect was reached.
Poisoning by "tartarised antimony" or "emetic tartar" is a plot device in the first modern detective novel, The Notting Hill Mystery (1862). The emetic tartar was kept by a character in the novel because he was "addicted to the pleasures of the table, and was in the habit of taking an occasional emetic."
The compound is still used to induce vomiting in captured animals in order to study their diets.
Antimony potassium tartrate is used as an insecticide against thrips. It is in IRAC class 8E.
Hub AI
Antimony potassium tartrate AI simulator
(@Antimony potassium tartrate_simulator)
Antimony potassium tartrate
Antimony potassium tartrate, also known as potassium antimonyl tartrate, potassium antimontarterate, or tartar emetic, has the formula K2Sb2(C4H2O6)2. The compound has long been known as a powerful emetic, and was used in the treatment of schistosomiasis and leishmaniasis. It is used as a resolving agent. It typically is obtained as a hydrate.
The first treatment application against trypanosomiasis was tested in 1906, and the compound's use to treat other tropical diseases was researched. The treatment of leishmania with antimony potassium tartrate started in 1913. After the introduction of antimony(V) containing complexes like sodium stibogluconate and meglumine antimoniate, the use of antimony potassium tartrate was phased out. After British physician John Brian Christopherson's discovery in 1918 that antimony potassium tartrate could cure schistosomiasis, the antimonial drugs became widely used. However, the injection of antimony potassium tartrate had severe side effects such as Adams–Stokes syndrome and therefore alternative substances were under investigation. With the introduction and subsequent larger use of praziquantel in the 1970s, antimony-based treatments fell out of use.
Tartar emetic was used in the late 19th and early 20th century in patent medicine as a remedy for alcohol intoxication, and was first ruled ineffective in the United States in 1941, in United States v. 11 1/4 Dozen Packages of Articles Labeled in Part Mrs. Moffat's Shoo-Fly Powders for Drunkenness.
The New England Journal of Medicine reported a case study of a patient whose wife secretly gave him a dose of a product called "tartaro emetico" which contained trivalent antimony (antimony potassium tartrate) and is sold in Central America as an aversive treatment for alcohol use disorder. The patient, who had been out drinking the night before, developed persistent vomiting shortly after being given orange juice with the drug. When admitted to the hospital, and later in the intensive care unit, he experienced severe chest pains, cardiac abnormalities, renal and hepatic toxicity, and nearly died. The Journal reports that "Two years later, he [the patient] reports complete abstinence from alcohol."
Antimony potassium tartrate's potential as an emetic has been known since the Middle Ages. The compound itself was considered toxic and therefore a different way to administer it was found. Cups made from pure antimony were used to store wine for 24 hours and then the resulting solution of antimony potassium tartrate in wine was consumed in small portions until the wanted emetic effect was reached.
Poisoning by "tartarised antimony" or "emetic tartar" is a plot device in the first modern detective novel, The Notting Hill Mystery (1862). The emetic tartar was kept by a character in the novel because he was "addicted to the pleasures of the table, and was in the habit of taking an occasional emetic."
The compound is still used to induce vomiting in captured animals in order to study their diets.
Antimony potassium tartrate is used as an insecticide against thrips. It is in IRAC class 8E.