Recent from talks
Nothing was collected or created yet.
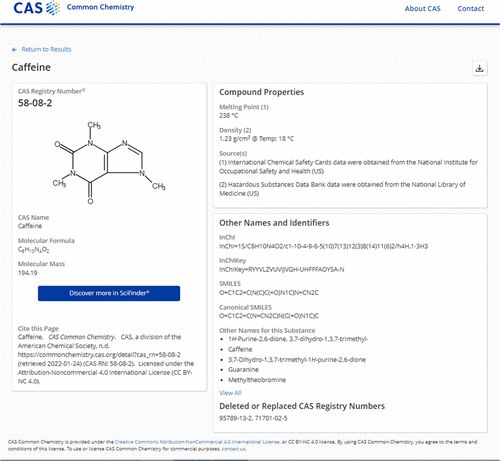
CAS Registry Number
View on Wikipedia

A CAS Registry Number[1] (also referred to as CAS RN[2] or informally CAS Number) is a unique identification number, assigned by the Chemical Abstracts Service (CAS) in the US to every chemical substance described in the open scientific literature, in order to index the substance in the CAS Registry. This registry includes all substances described since 1957, plus some substances from as far back as the early 1800s.[3] It is a chemical database that includes organic and inorganic compounds, minerals, isotopes, alloys, mixtures, and nonstructurable materials (UVCBs - substances of unknown or variable composition, complex reaction products, or biological origin).[4] CAS RNs are generally serial numbers (with a check digit), so they do not contain any information about the structures themselves the way SMILES and InChI strings do.
The CAS Registry is an authoritative collection of disclosed chemical substance information. It identifies more than 204 million unique organic and inorganic substances and 69 million protein and DNA sequences,[3] plus additional information about each substance. It is updated with around 15,000 additional new substances daily.[5] A collection of almost 500 thousand CAS registry numbers is made available under a CC BY-NC license at ACS Commons Chemistry.[6]
History and use
[edit]Historically, chemicals have been identified by a wide variety of synonyms and properties. One of the biggest challenges in the early development of substance indexing, a task undertaken by the Chemical Abstracts Service, was in identifying if a substance in literature was new or if it had been previously discovered. Well-known chemicals may be known via multiple generic, historical, commercial, and/or (black)-market names, and even systematic nomenclature based on structure alone was not universally useful. An algorithm was developed to translate the structural formula of a chemical into a computer-searchable table, which provided a basis for the service that listed each chemical with its CAS Registry Number, the CAS Chemical Registry System, which became operational in 1965.[7]
CAS Registry Numbers (CAS RN) are simple and regular, convenient for database searches. They offer a reliable, common and international link to every specific substance across the various nomenclatures and disciplines used by branches of science, industry, and regulatory bodies. Almost all molecule databases today allow searching by CAS Registry Number, and it is used as a global standard.[8]
Format
[edit]A CAS Registry Number has no inherent meaning, but is assigned in sequential, increasing order when the substance is identified by CAS scientists for inclusion in the CAS Registry database.
A CAS RN is separated by hyphens into three parts, the first consisting from two up to seven digits,[9] the second consisting of two digits, and the third consisting of a single digit serving as a check digit. This format gives CAS a maximum capacity of 1,000,000,000 unique numbers.
The check digit is found by taking the last digit times 1, the preceding digit times 2, the preceding digit times 3 etc., adding all these up and computing the sum modulo 10. For example, the CAS number of water is 7732-18-5: the checksum 5 is calculated as (8×1 + 1×2 + 2×3 + 3×4 + 7×5 + 7×6) = 105; 105 mod 10 = 5.
Granularity
[edit]- Stereoisomers and racemic mixtures are assigned discrete CAS Registry Numbers: L-epinephrine has 51-43-4, D-epinephrine has 150-05-0, and racemic DL-epinephrine has 329-65-7
- Different phases do not receive different CAS RNs (liquid water and ice both have 7732-18-5), but different crystal structures do (carbon in general is 7440-44-0, graphite is 7782-42-5 and diamond is 7782-40-3)
- Commonly encountered mixtures of known or unknown composition may receive a CAS RN; examples are Leishman stain (12627-53-1) and mustard oil (8007-40-7).
- Some chemical elements are discerned by their oxidation state, e.g. the element chromium has 7440-47-3, the trivalent Cr(III) has 16065-83-1 and the hexavalent Cr(VI) species have 18540-29-9.
- Occasionally whole classes of molecules receive a single CAS RN: the class of enzymes known as alcohol dehydrogenases has 9031-72-5.
Search engines
[edit]- CHEMINDEX Search via Canadian Centre for Occupational Health and Safety[10]
- ChemIDplus Advanced via United States National Library of Medicine[11]
- Common Chemistry[12][13] via Australian Inventory of Chemical Substances[14]
- European chemical Substances Information System[15] via the website of Royal Society of Chemistry[16]
- HSNO Chemical Classification Information Database via Environmental Risk Management Authority[17]
- Search Tool of Australian Inventory of Chemical Substances[18]
- USEPA CompTox Chemicals Dashboard[19]
See also
[edit]- Academic publishing
- Beilstein Registry Number
- Chemical file format
- Dictionary of chemical formulas
- EC# (EINECS and ELINCS, European Community)
- EC number (Enzyme Commission)
- International Union of Pure and Applied Chemistry
- List of CAS numbers by chemical compound
- MDL number
- PubChem
- Registration authority
- UN number
References
[edit]- ^ CAS registry description Archived 2008-07-25 at the Wayback Machine, by Chemical Abstracts Service
- ^ "CAS Registry Number Verified Partner Program". CAS. Retrieved 4 August 2021.
- ^ a b "CAS Content: Substances". www.cas.org. Retrieved 10 August 2020.
- ^ American Chemical Society. "CAS Registry and CASRNs". Archived from the original on 25 July 2008. Retrieved 25 July 2009.
- ^ "CAS REGISTRY". www.cas.org.
- ^ "CAS Common Chemistry expands collection of publicly available chemical information". CAS. Retrieved 17 March 2021.
- ^ Shively, Eric (11 June 2007). "CAS Surveys Its First 100 Years". Chemical & Engineering News. Retrieved 16 April 2024.
- ^ Jacobs, Andrea; Williams, Dustin; Hickey, Katherine; Patrick, Nathan; Williams, Antony J.; Chalk, Stuart; McEwen, Leah; Willighagen, Egon; Walker, Martin; Bolton, Evan; Sinclair, Gabriel; Sanford, Adam (13 June 2022). "CAS Common Chemistry in 2021: Expanding Access to Trusted Chemical Information for the Scientific Community". Journal of Chemical Information and Modeling. 62 (11): 2737–2743. doi:10.1021/acs.jcim.2c00268. ISSN 1549-9596. PMC 9199008. PMID 35559614.
- ^ 2014-06-18, https://www.cas.org/content/chemical-substances/faqs
- ^ Canadian Centre for Occupational Health and Safety. "CHEMINDEX Search". Retrieved 13 July 2009.
- ^ United States National Library of Medicine. "ChemIDplus Advanced". Archived from the original on 11 October 2016. Retrieved 28 June 2021.
- ^ American Chemical Society. "Substance Search". Retrieved 8 July 2009.
- ^ Jacobs, Andrea; Williams, Dustin; Hickey, Katherine; Patrick, Nathan; Williams, Antony J.; Chalk, Stuart; McEwen, Leah; Willighagen, Egon; Walker, Martin; Bolton, Evan; Sinclair, Gabriel; Sanford, Adam (13 May 2022). "CAS Common Chemistry in 2021: Expanding Access to Trusted Chemical Information for the Scientific Community". Journal of Chemical Information and Modeling. 62 (11): 2737–2743. doi:10.1021/acs.jcim.2c00268. PMC 9199008. PMID 35559614. S2CID 248778162.
- ^ National Industrial Chemicals Notification and Assessment Scheme. "AICS Detailed Help / Guidance Notes". Archived from the original on 9 July 2009. Retrieved 8 July 2009.
- ^ European Commission Joint research Centre. "ESIS : European chemical Substances Information System". Archived from the original on 1 January 2014. Retrieved 11 July 2009.
- ^ Library & Information Centre. "Finding a CAS Registry Number". Retrieved 11 July 2009.
- ^ Environmental Risk Management Authority. "HSNO Chemical Classification Information Database". Archived from the original on 11 July 2009. Retrieved 14 July 2009.
- ^ National Induscctrial Chemicals Notification and Assessment Scheme. "AICS Search Tool". Archived from the original on 14 May 2009. Retrieved 11 July 2009.
- ^ "CompTox Chemicals Dashboard | Home". comptox.epa.gov. Archived from the original on 24 December 2016. Retrieved 14 October 2021.
External links
[edit]- CAS registry description, by Chemical Abstracts Service
To find the CAS number of a compound given its name, formula or structure, the following free resources can be used: