Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
Cluster decay
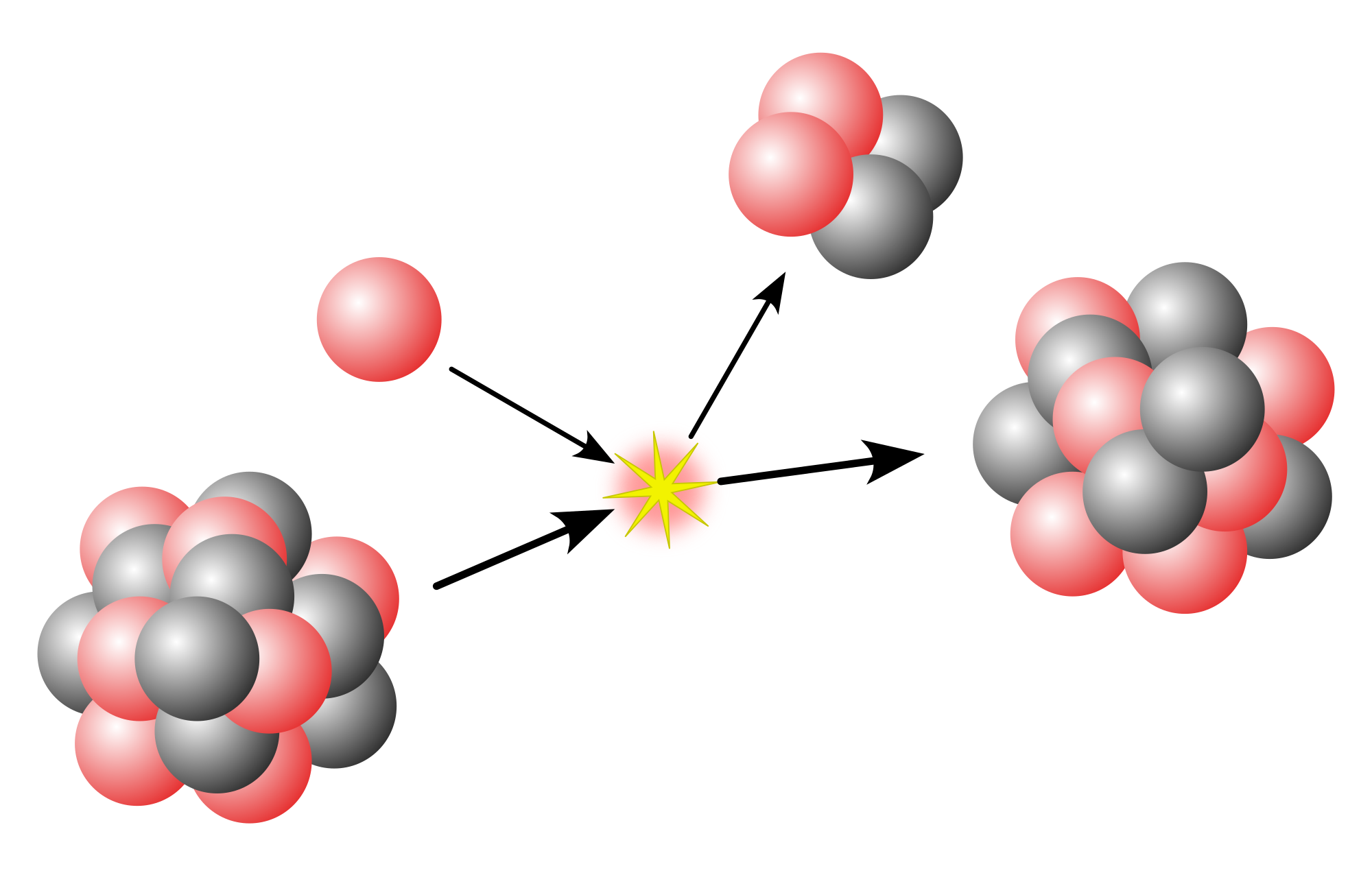
Cluster decay, also known as heavy particle radioactivity, is a rare type of radioactive decay in which an unstable atomic nucleus emits a small cluster of protons and neutrons. The emitted cluster is larger than an alpha particle (which has two protons and two neutrons) but smaller than the typical fragments produced in spontaneous fission.
This process is a way for a heavy, unstable atom to become more stable. For example, an atom of 223
88Ra can emit a 14
6C nucleus (which contains 6 protons and 8 neutrons) and transform into a more stable 209
82Pb atom.
Cluster decay was theoretically predicted in 1980 by Aureliu Săndulescu, Dorin N. Poenaru, and Walter Greiner, and was first experimentally confirmed in 1984 by H. J. Rose and G. A. Jones.
Like alpha decay, cluster decay is a quantum tunneling process. The cluster of protons and neutrons forms inside the parent nucleus and must penetrate a Coulomb barrier to escape. The process is highly unlikely, which is why cluster decay is a rare phenomenon, with branching ratios relative to alpha decay being very small.
The decay transforms the parent nucleus (with atomic number Z and mass number A) into a daughter nucleus (Zd, Ad) by emitting a nuclear cluster (Ze, Ae). The number of protons and neutrons is conserved:
The following example shows the decay of 223
88Ra into 209
82Pb by emission of a 14
6C cluster:
The energy released in the decay (the Q-value) is converted into the kinetic energy of the fragments. As required by the conservation of momentum, the lighter emitted cluster carries away most of this energy. The kinetic energy of the cluster, Ek, is approximately:
This decay mode is intermediate between standard alpha decay, where a light helium nucleus is emitted, and spontaneous fission, which splits a nucleus into two or more large fragments with a probabilistic mass distribution. In cluster decay, the emitted particle is a specific light nucleus, not a range of possible fragments.
Hub AI
Cluster decay AI simulator
(@Cluster decay_simulator)
Cluster decay
Cluster decay, also known as heavy particle radioactivity, is a rare type of radioactive decay in which an unstable atomic nucleus emits a small cluster of protons and neutrons. The emitted cluster is larger than an alpha particle (which has two protons and two neutrons) but smaller than the typical fragments produced in spontaneous fission.
This process is a way for a heavy, unstable atom to become more stable. For example, an atom of 223
88Ra can emit a 14
6C nucleus (which contains 6 protons and 8 neutrons) and transform into a more stable 209
82Pb atom.
Cluster decay was theoretically predicted in 1980 by Aureliu Săndulescu, Dorin N. Poenaru, and Walter Greiner, and was first experimentally confirmed in 1984 by H. J. Rose and G. A. Jones.
Like alpha decay, cluster decay is a quantum tunneling process. The cluster of protons and neutrons forms inside the parent nucleus and must penetrate a Coulomb barrier to escape. The process is highly unlikely, which is why cluster decay is a rare phenomenon, with branching ratios relative to alpha decay being very small.
The decay transforms the parent nucleus (with atomic number Z and mass number A) into a daughter nucleus (Zd, Ad) by emitting a nuclear cluster (Ze, Ae). The number of protons and neutrons is conserved:
The following example shows the decay of 223
88Ra into 209
82Pb by emission of a 14
6C cluster:
The energy released in the decay (the Q-value) is converted into the kinetic energy of the fragments. As required by the conservation of momentum, the lighter emitted cluster carries away most of this energy. The kinetic energy of the cluster, Ek, is approximately:
This decay mode is intermediate between standard alpha decay, where a light helium nucleus is emitted, and spontaneous fission, which splits a nucleus into two or more large fragments with a probabilistic mass distribution. In cluster decay, the emitted particle is a specific light nucleus, not a range of possible fragments.