Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
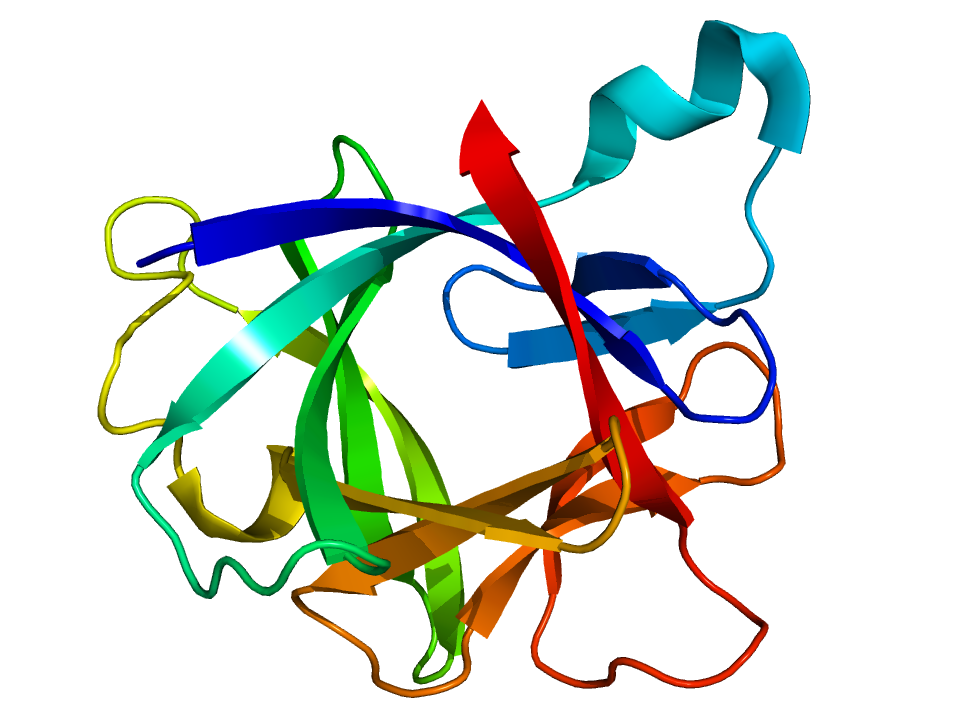
Interleukin 1 beta
Interleukin-1 beta (IL-1β) also known as leukocytic pyrogen, leukocytic endogenous mediator, mononuclear cell factor, lymphocyte activating factor and other names, is a cytokine protein that in humans is encoded by the IL1B gene. There are two genes for interleukin-1 (IL-1): IL-1 alpha and IL-1 beta (this gene). IL-1β precursor is cleaved by cytosolic caspase 1 (interleukin 1 beta convertase) to form mature IL-1β.
The fever-producing property of human leukocytic pyrogen (interleukin 1) was purified by Dinarello in 1977 with a specific activity of 10–20 nanograms/kg. In 1979, Dinarello reported that purified human leukocytic pyrogen was the same molecule that was described by Igal Gery in 1972. He named it lymphocyte-activating factor (LAF) because it was a lymphocyte mitogen. It was not until 1984 that interleukin 1 was discovered to consist of two distinct proteins, now called interleukin-1 alpha and interleukin-1 beta.
IL-1β is a member of the interleukin 1 family of cytokines. This cytokine is produced by activated macrophages, monocytes, and a subset of dendritic cells known as slanDC, as a proprotein, which is proteolytically processed to its active form by caspase 1 (CASP1/ICE). This cytokine is an important mediator of the inflammatory response, and is involved in a variety of cellular activities, including cell proliferation, differentiation, and apoptosis. The induction of cyclooxygenase-2 (PTGS2/COX2) by this cytokine in the central nervous system (CNS) is found to contribute to inflammatory pain hypersensitivity. This gene and eight other interleukin 1 family genes form a cytokine gene cluster on chromosome 2.
IL-1β, in combination with IL-23, induced expression of IL-17, IL-21 and IL-22 by γδ T cells. This induction of expression is in the absence of additional signals. That suggests IL-1β is involved in modulation of autoimmune inflammation
Different inflammasome complex — cytosolic molecular complex — have been described. Inflammasomes recognize danger signals and activate proinflammatory process and production of IL-1β and IL-18. NLRP3 (contains three domain: pyrin domain, a nucleotide-binding domain and a leucine-rich repeat) type of inflammasome is activated by various stimuli and there are documented several diseases connected to NLRP3 activation like type 2 diabetes mellitus, Alzheimer's disease, obesity and atherosclerosis.
Before cleavage by caspase 1, pro-IL-1β has a molecular weight of 37 kDa. The molecular weight of the proteolytically processed IL-1β is 17.5 kDa. IL-1β has the following amino acid sequence:
The physiological activity determined from the dose dependent proliferation of murine D10S cells is 2.5 x 108 to 7.1 x 108 units/mg.
IL-1β is present in other species of animals, however non-mammalian sequences of IL-1β lack a conserved cascase-1 cleavage site.
Hub AI
Interleukin 1 beta AI simulator
(@Interleukin 1 beta_simulator)
Interleukin 1 beta
Interleukin-1 beta (IL-1β) also known as leukocytic pyrogen, leukocytic endogenous mediator, mononuclear cell factor, lymphocyte activating factor and other names, is a cytokine protein that in humans is encoded by the IL1B gene. There are two genes for interleukin-1 (IL-1): IL-1 alpha and IL-1 beta (this gene). IL-1β precursor is cleaved by cytosolic caspase 1 (interleukin 1 beta convertase) to form mature IL-1β.
The fever-producing property of human leukocytic pyrogen (interleukin 1) was purified by Dinarello in 1977 with a specific activity of 10–20 nanograms/kg. In 1979, Dinarello reported that purified human leukocytic pyrogen was the same molecule that was described by Igal Gery in 1972. He named it lymphocyte-activating factor (LAF) because it was a lymphocyte mitogen. It was not until 1984 that interleukin 1 was discovered to consist of two distinct proteins, now called interleukin-1 alpha and interleukin-1 beta.
IL-1β is a member of the interleukin 1 family of cytokines. This cytokine is produced by activated macrophages, monocytes, and a subset of dendritic cells known as slanDC, as a proprotein, which is proteolytically processed to its active form by caspase 1 (CASP1/ICE). This cytokine is an important mediator of the inflammatory response, and is involved in a variety of cellular activities, including cell proliferation, differentiation, and apoptosis. The induction of cyclooxygenase-2 (PTGS2/COX2) by this cytokine in the central nervous system (CNS) is found to contribute to inflammatory pain hypersensitivity. This gene and eight other interleukin 1 family genes form a cytokine gene cluster on chromosome 2.
IL-1β, in combination with IL-23, induced expression of IL-17, IL-21 and IL-22 by γδ T cells. This induction of expression is in the absence of additional signals. That suggests IL-1β is involved in modulation of autoimmune inflammation
Different inflammasome complex — cytosolic molecular complex — have been described. Inflammasomes recognize danger signals and activate proinflammatory process and production of IL-1β and IL-18. NLRP3 (contains three domain: pyrin domain, a nucleotide-binding domain and a leucine-rich repeat) type of inflammasome is activated by various stimuli and there are documented several diseases connected to NLRP3 activation like type 2 diabetes mellitus, Alzheimer's disease, obesity and atherosclerosis.
Before cleavage by caspase 1, pro-IL-1β has a molecular weight of 37 kDa. The molecular weight of the proteolytically processed IL-1β is 17.5 kDa. IL-1β has the following amino acid sequence:
The physiological activity determined from the dose dependent proliferation of murine D10S cells is 2.5 x 108 to 7.1 x 108 units/mg.
IL-1β is present in other species of animals, however non-mammalian sequences of IL-1β lack a conserved cascase-1 cleavage site.