Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Mustard gas AI simulator
(@Mustard gas_simulator)
Hub AI
Mustard gas AI simulator
(@Mustard gas_simulator)
Mustard gas
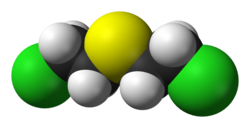
Mustard gas or sulfur mustard are names commonly used for the organosulfur chemical compound bis(2-chloroethyl) sulfide, which has the chemical structure S(CH2CH2Cl)2, as well as other species. In the wider sense, compounds with the substituents −SCH2CH2X or −N(CH2CH2X)2 are known as sulfur mustards or nitrogen mustards, respectively, where X = Cl or Br. Such compounds are potent alkylating agents, making mustard gas acutely and severely toxic. Mustard gas is a carcinogen. There is no preventative agent against mustard gas, with protection depending entirely on skin and airways protection, and no antidote exists for mustard poisoning.
Also known as mustard agents, this family of compounds comprises infamous cytotoxins and blister agents with a long history of use as chemical weapons. The name mustard gas is technically incorrect; the substances, when dispersed, are often not gases but a fine mist of liquid droplets that can be readily absorbed through the skin and by inhalation. The skin can be affected by contact with either the liquid or vapor. The rate of penetration into skin is proportional to dose, temperature and humidity.
Sulfur mustards are viscous liquids at room temperature and have an odor resembling mustard plants, garlic, or horseradish, hence the name. When pure, they are colorless, but when used in impure forms, such as in warfare, they are usually yellow-brown. Mustard gases form blisters on exposed skin and in the lungs, often resulting in prolonged illness ending in death.
The name of mustard gas derived from its yellow color, smell of mustard, and burning sensation on eyes. The term was first used in 1917 during World War I when Germans used the poison in combat.
Sulfur mustard is a type of chemical warfare agent. As a chemical weapon, mustard gas has been used in several armed conflicts since World War I, including the Iran–Iraq War, resulting in more than 100,000 casualties. Sulfur-based and nitrogen-based mustard agents are regulated under Schedule 1 of the 1993 Chemical Weapons Convention, as substances with few uses other than in chemical warfare. Mustard agents can be deployed by means of artillery shells, aerial bombs, rockets, or by spraying from aircraft.
Mustard gases have powerful blistering effects on victims. They are also carcinogenic and mutagenic alkylating agents. Their high lipophilicity accelerates their absorption into the body. Because mustard agents often do not elicit immediate symptoms, contaminated areas may appear normal. Within 24 hours of exposure, victims experience intense itching and skin irritation. If this irritation goes untreated, blisters filled with pus can form wherever the agent contacted the skin. As chemical burns, these are severely debilitating. Mustard gas can have the effect of turning a patient's skin different colors due to melanogenesis.
If the victim's eyes were exposed, then they become sore, starting with conjunctivitis (also known as pink eye), after which the eyelids swell, resulting in temporary blindness. Extreme ocular exposure to mustard gas vapors may result in corneal ulceration, anterior chamber scarring, and neovascularization. In these severe and infrequent cases, corneal transplantation has been used as a treatment. Miosis, when the pupil constricts more than usual, may also occur, which may be the result of the cholinomimetic activity of mustard. If inhaled in high concentrations, mustard agents cause bleeding and blistering within the respiratory system, damaging mucous membranes and causing pulmonary edema. Depending on the level of contamination, mustard agent burns can vary between first and second degree burns. They can also be as severe, disfiguring, and dangerous as third degree burns. Some 80% of sulfur mustard in contact with the skin evaporates, while 10% stays in the skin and 10% is absorbed and circulated in the blood.
The carcinogenic and mutagenic effects of exposure to mustard gas increase the risk of developing cancer later in life. In a study of patients 25 years after wartime exposure to chemical weaponry, c-DNA microarray profiling indicated that 122 genes were significantly mutated in the lungs and airways of mustard gas victims. Those genes all correspond to functions commonly affected by mustard gas exposure, including apoptosis, inflammation, and stress responses. The long-term ocular complications include burning, tearing, itching, photophobia, presbyopia, pain, and foreign-body sensations.
Mustard gas
Mustard gas or sulfur mustard are names commonly used for the organosulfur chemical compound bis(2-chloroethyl) sulfide, which has the chemical structure S(CH2CH2Cl)2, as well as other species. In the wider sense, compounds with the substituents −SCH2CH2X or −N(CH2CH2X)2 are known as sulfur mustards or nitrogen mustards, respectively, where X = Cl or Br. Such compounds are potent alkylating agents, making mustard gas acutely and severely toxic. Mustard gas is a carcinogen. There is no preventative agent against mustard gas, with protection depending entirely on skin and airways protection, and no antidote exists for mustard poisoning.
Also known as mustard agents, this family of compounds comprises infamous cytotoxins and blister agents with a long history of use as chemical weapons. The name mustard gas is technically incorrect; the substances, when dispersed, are often not gases but a fine mist of liquid droplets that can be readily absorbed through the skin and by inhalation. The skin can be affected by contact with either the liquid or vapor. The rate of penetration into skin is proportional to dose, temperature and humidity.
Sulfur mustards are viscous liquids at room temperature and have an odor resembling mustard plants, garlic, or horseradish, hence the name. When pure, they are colorless, but when used in impure forms, such as in warfare, they are usually yellow-brown. Mustard gases form blisters on exposed skin and in the lungs, often resulting in prolonged illness ending in death.
The name of mustard gas derived from its yellow color, smell of mustard, and burning sensation on eyes. The term was first used in 1917 during World War I when Germans used the poison in combat.
Sulfur mustard is a type of chemical warfare agent. As a chemical weapon, mustard gas has been used in several armed conflicts since World War I, including the Iran–Iraq War, resulting in more than 100,000 casualties. Sulfur-based and nitrogen-based mustard agents are regulated under Schedule 1 of the 1993 Chemical Weapons Convention, as substances with few uses other than in chemical warfare. Mustard agents can be deployed by means of artillery shells, aerial bombs, rockets, or by spraying from aircraft.
Mustard gases have powerful blistering effects on victims. They are also carcinogenic and mutagenic alkylating agents. Their high lipophilicity accelerates their absorption into the body. Because mustard agents often do not elicit immediate symptoms, contaminated areas may appear normal. Within 24 hours of exposure, victims experience intense itching and skin irritation. If this irritation goes untreated, blisters filled with pus can form wherever the agent contacted the skin. As chemical burns, these are severely debilitating. Mustard gas can have the effect of turning a patient's skin different colors due to melanogenesis.
If the victim's eyes were exposed, then they become sore, starting with conjunctivitis (also known as pink eye), after which the eyelids swell, resulting in temporary blindness. Extreme ocular exposure to mustard gas vapors may result in corneal ulceration, anterior chamber scarring, and neovascularization. In these severe and infrequent cases, corneal transplantation has been used as a treatment. Miosis, when the pupil constricts more than usual, may also occur, which may be the result of the cholinomimetic activity of mustard. If inhaled in high concentrations, mustard agents cause bleeding and blistering within the respiratory system, damaging mucous membranes and causing pulmonary edema. Depending on the level of contamination, mustard agent burns can vary between first and second degree burns. They can also be as severe, disfiguring, and dangerous as third degree burns. Some 80% of sulfur mustard in contact with the skin evaporates, while 10% stays in the skin and 10% is absorbed and circulated in the blood.
The carcinogenic and mutagenic effects of exposure to mustard gas increase the risk of developing cancer later in life. In a study of patients 25 years after wartime exposure to chemical weaponry, c-DNA microarray profiling indicated that 122 genes were significantly mutated in the lungs and airways of mustard gas victims. Those genes all correspond to functions commonly affected by mustard gas exposure, including apoptosis, inflammation, and stress responses. The long-term ocular complications include burning, tearing, itching, photophobia, presbyopia, pain, and foreign-body sensations.