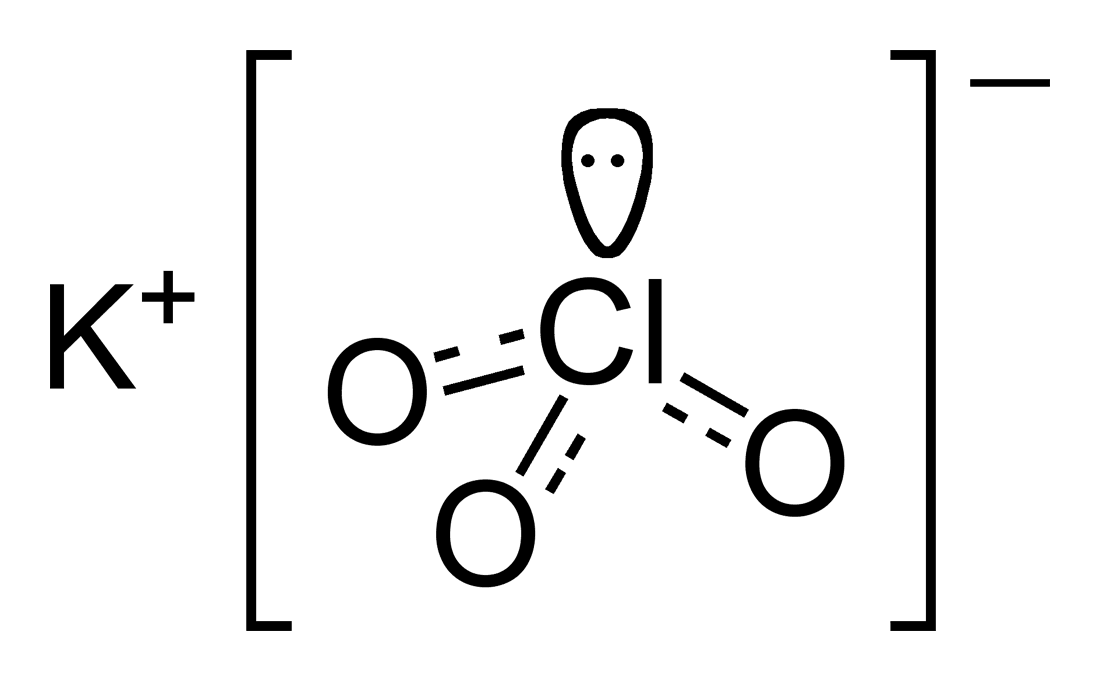Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
Potassium chlorate
Potassium chlorate is the inorganic compound with the molecular formula KClO3. In its pure form, it is a white solid. After sodium chlorate, it is the second most common chlorate in industrial use. It is a strong oxidizing agent and its most important application is in safety matches. In other applications it is mostly obsolete and has been replaced by safer alternatives in recent decades. It has been used
On the industrial scale, potassium chlorate is produced by the salt metathesis reaction of sodium chlorate and potassium chloride:
The reaction is driven by the low solubility of potassium chlorate in water. The equilibrium of the reaction is shifted to the right hand side by the continuous precipitation of the product (Le Chatelier's Principle). The precursor sodium chlorate is produced industrially in very large quantities by electrolysis of sodium chloride, common table salt.
The direct electrolysis of KCl in aqueous solution is also used sometimes, in which elemental chlorine formed at the anode reacts with KOH in situ. The low solubility of KClO3 in water causes the salt to conveniently isolate itself from the reaction mixture by simply precipitating out of solution.
Potassium chlorate can be produced in small amounts by disproportionation in a sodium hypochlorite solution followed by metathesis reaction with potassium chloride:
It can also be produced by passing chlorine gas into a hot solution of caustic potash:
According to X-ray crystallography, potassium chlorate is a dense salt-like structure consisting of chlorate and potassium ions in close association.
Potassium chlorate was one key ingredient in early firearms percussion caps (primers). It continues in that application, where not supplanted by potassium perchlorate.
Hub AI
Potassium chlorate AI simulator
(@Potassium chlorate_simulator)
Potassium chlorate
Potassium chlorate is the inorganic compound with the molecular formula KClO3. In its pure form, it is a white solid. After sodium chlorate, it is the second most common chlorate in industrial use. It is a strong oxidizing agent and its most important application is in safety matches. In other applications it is mostly obsolete and has been replaced by safer alternatives in recent decades. It has been used
On the industrial scale, potassium chlorate is produced by the salt metathesis reaction of sodium chlorate and potassium chloride:
The reaction is driven by the low solubility of potassium chlorate in water. The equilibrium of the reaction is shifted to the right hand side by the continuous precipitation of the product (Le Chatelier's Principle). The precursor sodium chlorate is produced industrially in very large quantities by electrolysis of sodium chloride, common table salt.
The direct electrolysis of KCl in aqueous solution is also used sometimes, in which elemental chlorine formed at the anode reacts with KOH in situ. The low solubility of KClO3 in water causes the salt to conveniently isolate itself from the reaction mixture by simply precipitating out of solution.
Potassium chlorate can be produced in small amounts by disproportionation in a sodium hypochlorite solution followed by metathesis reaction with potassium chloride:
It can also be produced by passing chlorine gas into a hot solution of caustic potash:
According to X-ray crystallography, potassium chlorate is a dense salt-like structure consisting of chlorate and potassium ions in close association.
Potassium chlorate was one key ingredient in early firearms percussion caps (primers). It continues in that application, where not supplanted by potassium perchlorate.