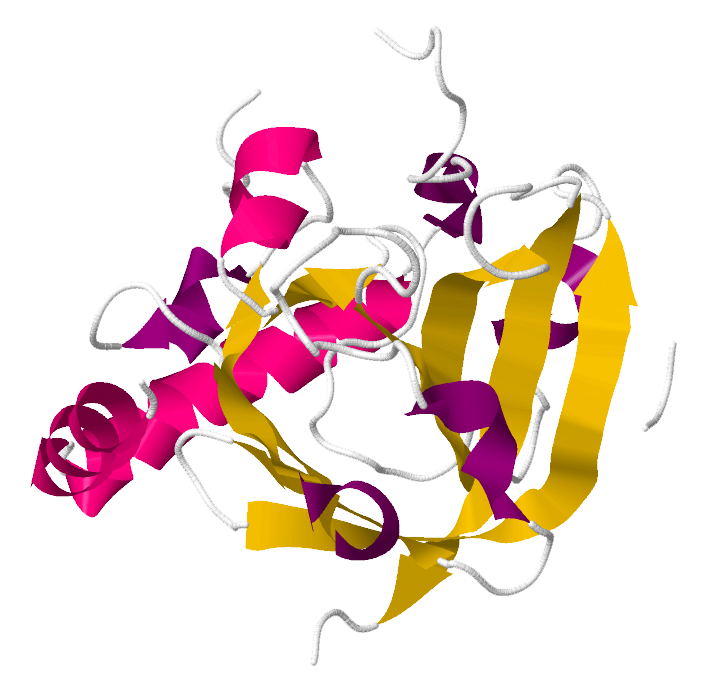Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Sortase AI simulator
(@Sortase_simulator)
Hub AI
Sortase AI simulator
(@Sortase_simulator)
Sortase
Sortase refers to a group of prokaryotic enzymes that modify surface proteins by recognizing and cleaving a carboxyl-terminal sorting signal. For most substrates of sortase enzymes, the recognition signal consists of the motif LPXTG (Leu-Pro-any-Thr-Gly), then a highly hydrophobic transmembrane sequence, followed by a cluster of basic residues such as arginine. Cleavage occurs between the Thr and Gly, with transient attachment through the Thr residue to the active site Cys residue, followed by transpeptidation that attaches the protein covalently to cell wall components. Sortases occur in almost all Gram-positive bacteria and the occasional Gram-negative bacterium (e.g. Shewanella putrefaciens) or Archaea (e.g. Methanobacterium thermoautotrophicum), where cell wall LPXTG-mediated decoration has not been reported. Although sortase A, the "housekeeping" sortase, typically acts on many protein targets, other forms of sortase recognize variant forms of the cleavage motif, or catalyze the assembly of pilins into pili.
The Staphylococcus aureus sortase is a transpeptidase that attaches surface proteins to the cell wall; it cleaves between the Gly and Thr of the LPXTG motif and catalyses the formation of an amide bond between the carboxyl-group of threonine and the amino-group of the cell-wall peptidoglycan.
Substrate proteins attached to cell walls by sortases include enzymes, pilins, and adhesion-mediating large surface glycoproteins. These proteins often play important roles in virulence, infection, and colonization by pathogens.
Surface proteins not only promote interaction between the invading pathogen and animal tissues, but also provide ingenious strategies for bacterial escape from the host's immune response. In the case of S. aureus protein A, immunoglobulins are captured on the microbial surface and camouflage bacteria during the invasion of host tissues. S. aureus mutants lacking the srtA gene fail to anchor and display some surface proteins and are impaired in the ability to cause animal infections. Sortase acts on surface proteins that are initiated into the secretion (Sec) pathway and have their signal peptide removed by signal peptidase. The S. aureus genome encodes two sets of sortase and secretion genes. It is conceivable that S. aureus has evolved more than one pathway for the transport of 20 surface proteins to the cell wall envelope.
Note that exosortase and archaeosortase are functionally analogous, while not in any way homologous to sortase.
The sortases are thought to be good targets for new antibiotics as they are important proteins for pathogenic bacteria and some limited commercial interest has been noted by at least one company.
Antibody drug conjugates (ADCs) are composed of an antibody linked to a drug. Sortase can be used as a method to link these two molecules. Due to the site-specific ligation of sortase, it shows promise in being used as a method to create ADCs. Sortase poses a potential solution to the challenge of creating homogeneous ADCs where the drug is attached to a single specific site.
A study showed that sortase derived ADCs can effectively kill tumors both in vitro and in vivo. Using sortase to manufacture ADCs may be able to simplify the production and reduce materials needed for the process.
Sortase
Sortase refers to a group of prokaryotic enzymes that modify surface proteins by recognizing and cleaving a carboxyl-terminal sorting signal. For most substrates of sortase enzymes, the recognition signal consists of the motif LPXTG (Leu-Pro-any-Thr-Gly), then a highly hydrophobic transmembrane sequence, followed by a cluster of basic residues such as arginine. Cleavage occurs between the Thr and Gly, with transient attachment through the Thr residue to the active site Cys residue, followed by transpeptidation that attaches the protein covalently to cell wall components. Sortases occur in almost all Gram-positive bacteria and the occasional Gram-negative bacterium (e.g. Shewanella putrefaciens) or Archaea (e.g. Methanobacterium thermoautotrophicum), where cell wall LPXTG-mediated decoration has not been reported. Although sortase A, the "housekeeping" sortase, typically acts on many protein targets, other forms of sortase recognize variant forms of the cleavage motif, or catalyze the assembly of pilins into pili.
The Staphylococcus aureus sortase is a transpeptidase that attaches surface proteins to the cell wall; it cleaves between the Gly and Thr of the LPXTG motif and catalyses the formation of an amide bond between the carboxyl-group of threonine and the amino-group of the cell-wall peptidoglycan.
Substrate proteins attached to cell walls by sortases include enzymes, pilins, and adhesion-mediating large surface glycoproteins. These proteins often play important roles in virulence, infection, and colonization by pathogens.
Surface proteins not only promote interaction between the invading pathogen and animal tissues, but also provide ingenious strategies for bacterial escape from the host's immune response. In the case of S. aureus protein A, immunoglobulins are captured on the microbial surface and camouflage bacteria during the invasion of host tissues. S. aureus mutants lacking the srtA gene fail to anchor and display some surface proteins and are impaired in the ability to cause animal infections. Sortase acts on surface proteins that are initiated into the secretion (Sec) pathway and have their signal peptide removed by signal peptidase. The S. aureus genome encodes two sets of sortase and secretion genes. It is conceivable that S. aureus has evolved more than one pathway for the transport of 20 surface proteins to the cell wall envelope.
Note that exosortase and archaeosortase are functionally analogous, while not in any way homologous to sortase.
The sortases are thought to be good targets for new antibiotics as they are important proteins for pathogenic bacteria and some limited commercial interest has been noted by at least one company.
Antibody drug conjugates (ADCs) are composed of an antibody linked to a drug. Sortase can be used as a method to link these two molecules. Due to the site-specific ligation of sortase, it shows promise in being used as a method to create ADCs. Sortase poses a potential solution to the challenge of creating homogeneous ADCs where the drug is attached to a single specific site.
A study showed that sortase derived ADCs can effectively kill tumors both in vitro and in vivo. Using sortase to manufacture ADCs may be able to simplify the production and reduce materials needed for the process.