Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Hemoglobin AI simulator
(@Hemoglobin_simulator)
Hub AI
Hemoglobin AI simulator
(@Hemoglobin_simulator)
Hemoglobin
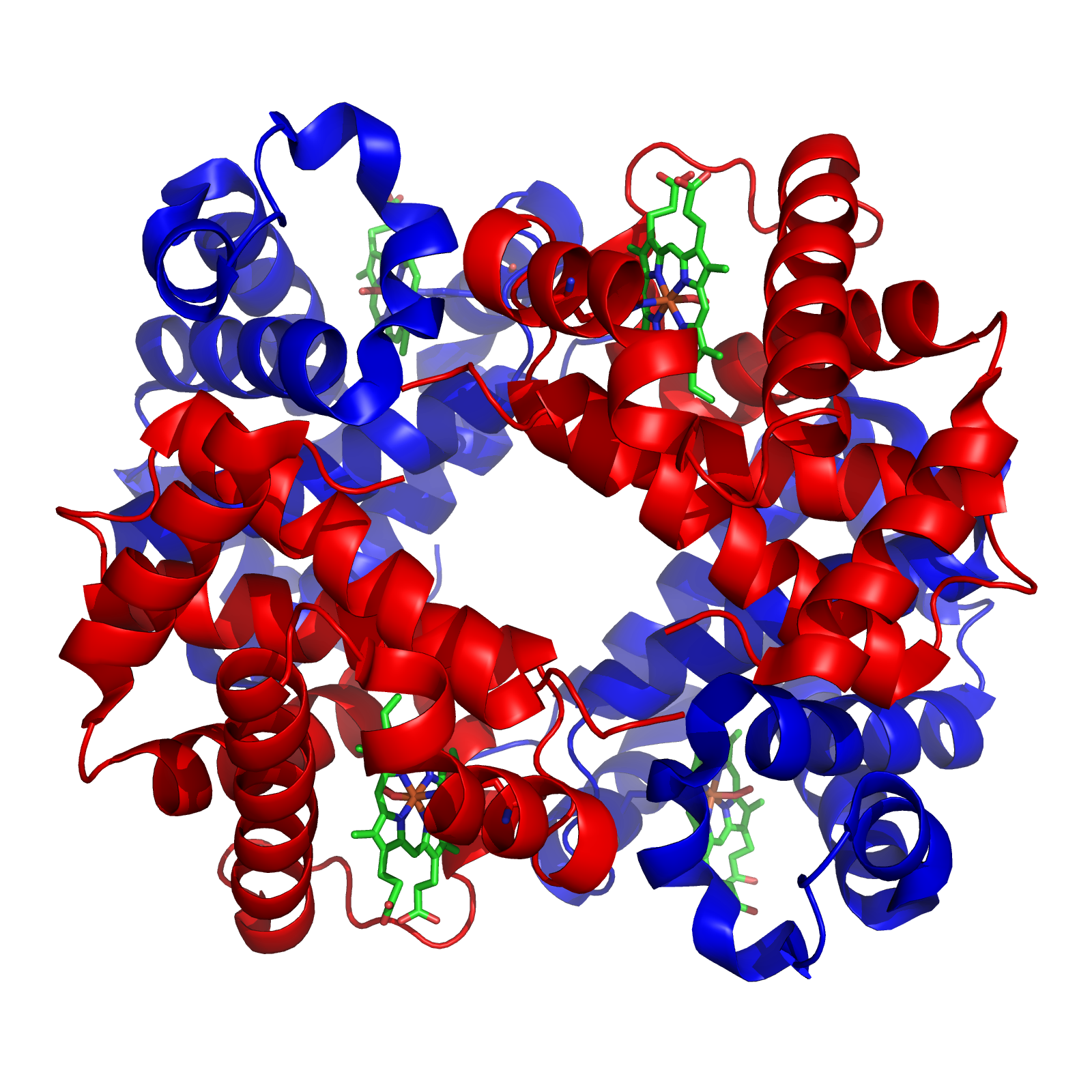
Hemoglobin (haemoglobin, Hb or Hgb) is a protein containing iron that facilitates the transportation of oxygen in red blood cells. Almost all vertebrates contain hemoglobin, with the sole exception of the fish family Channichthyidae. Hemoglobin in the blood carries oxygen from the respiratory organs (lungs or gills) to the other tissues of the body, where it releases the oxygen to enable aerobic respiration which powers an animal's metabolism. A healthy human has 12 to 20 grams of hemoglobin in every 100 mL of blood. Hemoglobin is a metalloprotein, a chromoprotein, and a globulin.
In mammals, hemoglobin makes up about 96% of a red blood cell's dry weight (excluding water), and around 35% of the total weight (including water). Hemoglobin has an oxygen-binding capacity of 1.34 mL of O2 per gram, which increases the total blood oxygen capacity seventy-fold compared to dissolved oxygen in blood plasma alone. The mammalian hemoglobin molecule can bind and transport up to four oxygen molecules.
Hemoglobin also transports other gases. It carries off some of the body's respiratory carbon dioxide (about 20–25% of the total) as carbaminohemoglobin, in which CO2 binds to the heme protein. The molecule also carries the important regulatory molecule nitric oxide bound to a thiol group in the globin protein, releasing it at the same time as oxygen.
Hemoglobin is also found in other cells, including in the A9 dopaminergic neurons of the substantia nigra, macrophages, alveolar cells, lungs, retinal pigment epithelium, hepatocytes, mesangial cells of the kidney, endometrial cells, cervical cells, and vaginal epithelial cells. In these tissues, hemoglobin absorbs unneeded oxygen as an antioxidant, and regulates iron metabolism. Excessive glucose in the blood can attach to hemoglobin and raise the level of hemoglobin A1c.
Hemoglobin and hemoglobin-like molecules are also found in many invertebrates, fungi, and plants. In these organisms, hemoglobins may carry oxygen, or they may transport and regulate other small molecules and ions such as carbon dioxide, nitric oxide, hydrogen sulfide and sulfide. A variant called leghemoglobin serves to scavenge oxygen away from anaerobic systems such as the nitrogen-fixing nodules of leguminous plants, preventing oxygen poisoning.
The medical condition hemoglobinemia, a form of anemia, is caused by intravascular hemolysis, in which hemoglobin leaks from red blood cells into the blood plasma.
Although blood had been known to carry oxygen since at least 1794, the oxygen-carrying property of hemoglobin was described by Hünefeld in 1840. In 1851, German physiologist Otto Funke published a series of articles in which he described growing hemoglobin crystals by successively diluting red blood cells with a solvent such as pure water, alcohol or ether, followed by slow evaporation of the solvent from the resulting protein solution. Hemoglobin's reversible oxygenation was described a few years later by Felix Hoppe-Seyler.
In 1825, Johann Friedrich Engelhart discovered that the ratio of iron to protein is identical in the hemoglobins of several species. From the known atomic mass of iron, he calculated the molecular mass of hemoglobin to n × 16000 (n=number of iron atoms per hemoglobin molecule, now known to be 4), the first determination of a protein's molecular mass. This "hasty conclusion" drew ridicule from colleagues who could not believe that any molecule could be so large. However, Gilbert Smithson Adair confirmed Engelhart's results in 1925 by measuring the osmotic pressure of hemoglobin solutions.
Hemoglobin
Hemoglobin (haemoglobin, Hb or Hgb) is a protein containing iron that facilitates the transportation of oxygen in red blood cells. Almost all vertebrates contain hemoglobin, with the sole exception of the fish family Channichthyidae. Hemoglobin in the blood carries oxygen from the respiratory organs (lungs or gills) to the other tissues of the body, where it releases the oxygen to enable aerobic respiration which powers an animal's metabolism. A healthy human has 12 to 20 grams of hemoglobin in every 100 mL of blood. Hemoglobin is a metalloprotein, a chromoprotein, and a globulin.
In mammals, hemoglobin makes up about 96% of a red blood cell's dry weight (excluding water), and around 35% of the total weight (including water). Hemoglobin has an oxygen-binding capacity of 1.34 mL of O2 per gram, which increases the total blood oxygen capacity seventy-fold compared to dissolved oxygen in blood plasma alone. The mammalian hemoglobin molecule can bind and transport up to four oxygen molecules.
Hemoglobin also transports other gases. It carries off some of the body's respiratory carbon dioxide (about 20–25% of the total) as carbaminohemoglobin, in which CO2 binds to the heme protein. The molecule also carries the important regulatory molecule nitric oxide bound to a thiol group in the globin protein, releasing it at the same time as oxygen.
Hemoglobin is also found in other cells, including in the A9 dopaminergic neurons of the substantia nigra, macrophages, alveolar cells, lungs, retinal pigment epithelium, hepatocytes, mesangial cells of the kidney, endometrial cells, cervical cells, and vaginal epithelial cells. In these tissues, hemoglobin absorbs unneeded oxygen as an antioxidant, and regulates iron metabolism. Excessive glucose in the blood can attach to hemoglobin and raise the level of hemoglobin A1c.
Hemoglobin and hemoglobin-like molecules are also found in many invertebrates, fungi, and plants. In these organisms, hemoglobins may carry oxygen, or they may transport and regulate other small molecules and ions such as carbon dioxide, nitric oxide, hydrogen sulfide and sulfide. A variant called leghemoglobin serves to scavenge oxygen away from anaerobic systems such as the nitrogen-fixing nodules of leguminous plants, preventing oxygen poisoning.
The medical condition hemoglobinemia, a form of anemia, is caused by intravascular hemolysis, in which hemoglobin leaks from red blood cells into the blood plasma.
Although blood had been known to carry oxygen since at least 1794, the oxygen-carrying property of hemoglobin was described by Hünefeld in 1840. In 1851, German physiologist Otto Funke published a series of articles in which he described growing hemoglobin crystals by successively diluting red blood cells with a solvent such as pure water, alcohol or ether, followed by slow evaporation of the solvent from the resulting protein solution. Hemoglobin's reversible oxygenation was described a few years later by Felix Hoppe-Seyler.
In 1825, Johann Friedrich Engelhart discovered that the ratio of iron to protein is identical in the hemoglobins of several species. From the known atomic mass of iron, he calculated the molecular mass of hemoglobin to n × 16000 (n=number of iron atoms per hemoglobin molecule, now known to be 4), the first determination of a protein's molecular mass. This "hasty conclusion" drew ridicule from colleagues who could not believe that any molecule could be so large. However, Gilbert Smithson Adair confirmed Engelhart's results in 1925 by measuring the osmotic pressure of hemoglobin solutions.