Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Sulfite AI simulator
(@Sulfite_simulator)
Hub AI
Sulfite AI simulator
(@Sulfite_simulator)
Sulfite
Sulfites or sulphites are compounds that contain the sulfite ion (systematic name: sulfate(IV) ion), SO2−
3. The sulfite ion is the conjugate base of bisulfite. Although its acid (sulfurous acid) is elusive, its salts are widely used.
Sulfites are substances that naturally occur in some foods and the human body. They are also used as regulated food additives. When in food or drink, sulfites are often lumped together with sulfur dioxide.
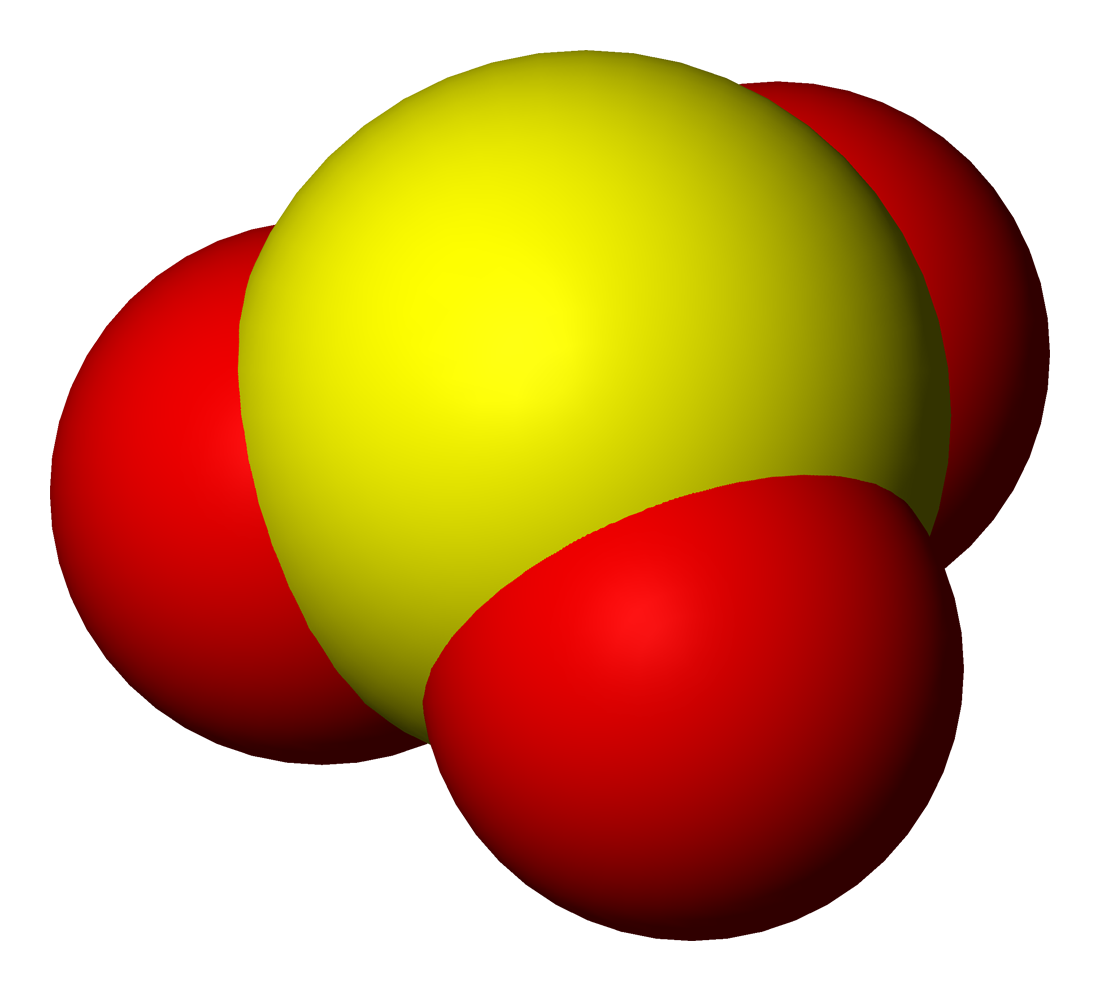
The structure of the sulfite anion can be described with three equivalent resonance structures. In each resonance structure, the sulfur atom is double-bonded to one oxygen atom with a formal charge of zero (neutral), and sulfur is singly bonded to the other two oxygen atoms, which each carry a formal charge of −1, together accounting for the −2 charge on the anion. There is also a non-bonded lone pair on the sulfur, so the structure predicted by VSEPR theory is trigonal pyramidal, as in ammonia (NH3). In the hybrid resonance structure, the S−O bonds are equivalently of bond order one and one-third.
Evidence from 17O NMR spectroscopic data suggests that protonation of the sulfite ion gives a mixture of isomers:
Sulfites are used as a food preservative or enhancer. They may come in various forms, such as:
Sulfites occur naturally in all wines to some extent. Sulfites are commonly introduced to arrest fermentation at a desired time, and may also be added to wine as preservatives to prevent spoilage and oxidation at several stages of the winemaking. Sulfur dioxide (SO2) protects wine not only from oxidation, but also from bacteria.
Organic wines are not necessarily sulfite-free, but generally have lower amounts and regulations stipulate lower maximum sulfite contents for these wines. In general, white wines contain more sulfites than red wines and sweeter wines contain more sulfites than drier ones.
In the United States, wines bottled after mid-1987 must have a label stating that they contain sulfites if they contain more than 10 parts per million (ppm). In the European Union an equivalent regulation came into force in November 2005. This includes sulfur dioxide, and the limit is on the milligrams per kilogram or per litre of sulfur dioxide equivalent. In 2012, a new EU regulation for organic wines came into force. In the United Kingdom, similar laws apply. Bottles of wine that contain over 10 mg/L (ppm) of "sulfites" (or sulfur dioxide) are required to bear "contains sulphites" on the label. This does not differ if sulfites are naturally occurring or added in the winemaking process.
Sulfite
Sulfites or sulphites are compounds that contain the sulfite ion (systematic name: sulfate(IV) ion), SO2−
3. The sulfite ion is the conjugate base of bisulfite. Although its acid (sulfurous acid) is elusive, its salts are widely used.
Sulfites are substances that naturally occur in some foods and the human body. They are also used as regulated food additives. When in food or drink, sulfites are often lumped together with sulfur dioxide.
The structure of the sulfite anion can be described with three equivalent resonance structures. In each resonance structure, the sulfur atom is double-bonded to one oxygen atom with a formal charge of zero (neutral), and sulfur is singly bonded to the other two oxygen atoms, which each carry a formal charge of −1, together accounting for the −2 charge on the anion. There is also a non-bonded lone pair on the sulfur, so the structure predicted by VSEPR theory is trigonal pyramidal, as in ammonia (NH3). In the hybrid resonance structure, the S−O bonds are equivalently of bond order one and one-third.
Evidence from 17O NMR spectroscopic data suggests that protonation of the sulfite ion gives a mixture of isomers:
Sulfites are used as a food preservative or enhancer. They may come in various forms, such as:
Sulfites occur naturally in all wines to some extent. Sulfites are commonly introduced to arrest fermentation at a desired time, and may also be added to wine as preservatives to prevent spoilage and oxidation at several stages of the winemaking. Sulfur dioxide (SO2) protects wine not only from oxidation, but also from bacteria.
Organic wines are not necessarily sulfite-free, but generally have lower amounts and regulations stipulate lower maximum sulfite contents for these wines. In general, white wines contain more sulfites than red wines and sweeter wines contain more sulfites than drier ones.
In the United States, wines bottled after mid-1987 must have a label stating that they contain sulfites if they contain more than 10 parts per million (ppm). In the European Union an equivalent regulation came into force in November 2005. This includes sulfur dioxide, and the limit is on the milligrams per kilogram or per litre of sulfur dioxide equivalent. In 2012, a new EU regulation for organic wines came into force. In the United Kingdom, similar laws apply. Bottles of wine that contain over 10 mg/L (ppm) of "sulfites" (or sulfur dioxide) are required to bear "contains sulphites" on the label. This does not differ if sulfites are naturally occurring or added in the winemaking process.