Cancer research
View on Wikipedia
Cancer research is research into cancer to identify causes and develop strategies for prevention, diagnosis, treatment, and cure.
Cancer research ranges from epidemiology, molecular bioscience to the performance of clinical trials to evaluate and compare applications of the various cancer treatments. These applications include surgery, radiation therapy, chemotherapy, hormone therapy, immunotherapy and combined treatment modalities such as chemo-radiotherapy. Starting in the mid-1990s, the emphasis in clinical cancer research shifted towards therapies derived from biotechnology research, such as cancer immunotherapy and gene therapy.
Cancer research is done in academia, research institutes, and corporate environments, and is largely government funded.[citation needed]
History
[edit]
Cancer research has been ongoing for centuries. Early research focused on the causes of cancer.[1] Percivall Pott identified the first environmental trigger (chimney soot) for cancer in 1775 and cigarette smoking was identified as a cause of lung cancer in 1950. Early cancer treatment focused on improving surgical techniques for removing tumors. Radiation therapy took hold in the 1900s. Chemotherapeutics were developed and refined throughout the 20th century.
The U.S. declared a "War on Cancer" in the 1970s, and increased the funding and support for cancer research.[2]
Seminal papers
[edit]Some of the most highly cited and most influential research reports include:
- The Hallmarks of Cancer, published in 2000, and Hallmarks of Cancer: The Next Generation, published in 2011, by Douglas Hanahan and Robert Weinberg. Together, these articles have been cited in over 30,000 published papers.
Types of research
[edit]Cancer research encompasses a variety of types and interdisciplinary areas of research. Scientists involved in cancer research may be trained in areas such as chemistry, biochemistry, molecular biology, physiology, medical physics, epidemiology, and biomedical engineering. Research performed on a foundational level is referred to as basic research and is intended to clarify scientific principles and mechanisms. Translational research aims to elucidate mechanisms of cancer development and progression and transform basic scientific findings into concepts that can be applicable to the treatment and prevention of cancer. Clinical research is devoted to the development of pharmaceuticals, surgical procedures, and medical technologies for the eventual treatment of patients.
Prevention and epidemiology
[edit]Epidemiologic analysis indicates that at least 35% of all cancer deaths in the world could now be avoided by primary prevention.[3] According to a newer GBD systematic analysis, in 2019, ~44% of all cancer deaths — or ~4.5 million deaths or ~105 million lost disability-adjusted life years — were due to known clearly preventable risk factors, led by smoking, alcohol use and high BMI.[4]
However, one 2015 study suggested that between ~70% and ~90% of cancers are due to environmental factors and therefore potentially preventable.[5][contradictory] Furthermore, it is estimated that with further research cancer death rates could be reduced by 70% around the world even without the development of any new therapies.[3] Cancer prevention research receives only 2–9% of global cancer research funding,[3] albeit many of the options for prevention are already well-known without further cancer-specific research but are not reflected in economics and policy. Mutational signatures of various cancers, for example, could reveal further causes of cancer and support causal attribution.[6][additional citation(s) needed]
Detection
[edit]Prompt detection of cancer is important, since it is usually more difficult to treat in later stages. Accurate detection of cancer is also important because false positives can cause harm from unnecessary medical procedures. Some screening protocols are currently not accurate (such as prostate-specific antigen testing). Others such as a colonoscopy or mammogram are unpleasant and as a result some patients may opt out. Active research is underway to address all these problems, to develop novel ways of cancer screening and to increase detection rates.[citation needed][further explanation needed]
For example:
- Multimodal learning AI systems are being developed to help detect many cancer types via integrating different types of data.[7][8]
- Scientists work on identifying and measurability of novel biomarkers or sets of such to detect cancer early, such as tumor-associated mycobiomes and bacterial microbiomes[9][10][11][further explanation needed][additional citation(s) needed]
- Researchers investigate whether ants could be used as biosensors to detect cancer via urine[12][additional citation(s) needed]
Treatment
[edit]Emerging topics of cancer treatment research include:
- Anti-cancer vaccines
- Oncophage[13]
- Sipuleucel-T (Provenge) is a prostate cancer vaccine
- Inactivated tumor cells are investigated as potential bifunctional cancer vaccines[14]
- Newer forms of chemotherapy
- Gene therapy[15][16][17]
- Photodynamic therapy
- Radiation therapy
- Reoviridae (Reolysin drug therapy)
- Targeted therapy
- Medical microbots (including bacterial),[18][19] nanobots[20] and bacterial 'cyborg cells'[21][additional citation(s) needed]
- Virotherapy[22][23]
- Antibodies[24][25][26][27][additional citation(s) needed]
- Photoimmunotherapy (for brain cancer)[28][29][additional citation(s) needed]
- Natural killer cells can induce immunological memory.[30] Research is being developed to modify their action against cancer.[30]
- How treatments can best be combined (in combination therapies)[31]
Cause and development of cancer
[edit]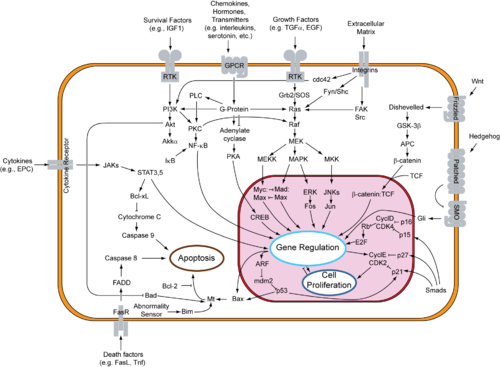
Research into the cause of cancer involves many different disciplines including genetics, diet, environmental factors (i.e. chemical carcinogens). In regard to investigation of causes and potential targets for therapy, the route used starts with data obtained from clinical observations, enters basic research, and, once convincing and independently confirmed results are obtained, proceeds with clinical research, involving appropriately designed trials on consenting human subjects, with the aim to test safety and efficiency of the therapeutic intervention method. An important part of basic research is characterization of the potential mechanisms of carcinogenesis, in regard to the types of genetic and epigenetic changes that are associated with cancer development. The mouse is often used as a mammalian model for manipulation of the function of genes that play a role in tumor formation, while basic aspects of tumor initiation, such as mutagenesis, are assayed on cultures of bacteria and mammalian cells.
Genes involved in cancer
[edit]The goal of oncogenomics is to identify new oncogenes or tumor suppressor genes that may provide new insights into cancer diagnosis, predicting clinical outcome of cancers, and new targets for cancer therapies. As the Cancer Genome Project stated in a 2004 review article, "a central aim of cancer research has been to identify the mutated genes that are causally implicated in oncogenesis (cancer genes)."[32] The Cancer Genome Atlas project is a related effort investigating the genomic changes associated with cancer, while the COSMIC cancer database documents acquired genetic mutations from hundreds of thousands of human cancer samples.[33]
These large scale projects, involving about 350 different types of cancer, have identified ~130,000 mutations in ~3000 genes that have been mutated in the tumors. The majority occurred in 319 genes, of which 286 were tumor suppressor genes and 33 oncogenes.
Several hereditary factors can increase the chance of cancer-causing mutations, including the activation of oncogenes or the inhibition of tumor suppressor genes. The functions of various onco- and tumor suppressor genes can be disrupted at different stages of tumor progression. Mutations in such genes can be used to classify the malignancy of a tumor.
In later stages, tumors can develop a resistance to cancer treatment. The identification of oncogenes and tumor suppressor genes is important to understand tumor progression and treatment success. The role of a given gene in cancer progression may vary tremendously, depending on the stage and type of cancer involved.[34]
Cancer epigenetics
[edit]Diet and cancer
[edit]
Many dietary recommendations have been proposed to reduce the risk of cancer, few have significant supporting scientific evidence.[42][43][44] Obesity and drinking alcohol have been correlated with the incidence and progression of some cancers.[42] Lowering the consumption of sweetened beverages is recommended as a measure to address obesity.[45]
Some specific foods are linked to specific cancers. There is strong evidence that processed meat and red meat intake increases risk of colorectal cancer.[46][47][48][49] Aflatoxin B1, a frequent food contaminant, increases risk of liver cancer,[50] while drinking coffee is associated with a reduced risk.[51] Betel nut chewing causes oral cancer.[50] Stomach cancer is more common in Japan due to its high-salt diet.[50][52]
Dietary recommendations for cancer prevention typically include weight management and eating a healthy diet, consisting mainly of "vegetables, fruit, whole grains and fish, and a reduced intake of red meat, animal fat, and refined sugar."[42] A healthy dietary pattern may lower cancer risk by 10–20%.[53] There is no clinical evidence that diets or specific foods can cure cancer.[54][55]Periods of intermittent fasting (time-restricted feeding which may not include caloric restriction) is investigated for potential usefulness in cancer prevention and treatment and as of 2021 additional trials are needed to elucidate the risks and benefits.[56][57][58][59] In some cases, "caloric restrictions could hinder both cancer growth and progression, besides enhancing the efficacy of chemotherapy and radiation therapy".[60] Caloric restriction mimetics, including some present in foods like spermidine, are also investigated for these or similar reasons.[61][62] Such and similar dietary supplements may contribute to prevention or treatment, with candidate substances including apigenin,[63][64][65] berberine,[66][67][68][69][70] jiaogulan,[71] and rhodiola rosea.[72][73]
Research funding
[edit]Cancer research is funded by government grants, charitable foundations and pharmaceutical and biotechnology companies.[74]
In the early 2000s, most funding for cancer research came from taxpayers and charities, rather than from corporations. In the US, less than 30% of all cancer research was funded by commercial researchers such as pharmaceutical companies.[75] Per capita, public spending on cancer research by taxpayers and charities in the US was five times as much in 2002–03 as public spending by taxpayers and charities in the 15 countries that were full members of the European Union.[75] As a percentage of GDP, the non-commercial funding of cancer research in the US was four times the amount dedicated to cancer research in Europe.[75] Half of Europe's non-commercial cancer research is funded by charitable organizations.[75]
The National Cancer Institute is the major funding institution in the United States. In the 2023 fiscal year, the NCI funded $7.1 billion in cancer research.[76]
Difficulties
[edit]Difficulties inherent to cancer research are shared with many types of biomedical research.
Cancer research processes have been criticised. These include, especially in the US, for the financial resources and positions required to conduct research. Other consequences of competition for research resources appear to be a substantial number of research publications whose results cannot be replicated.[77][78][79][80]
Replicability
[edit]
Public participation
[edit]Distributed computing
[edit]One can share computer time for distributed cancer research projects like Help Conquer Cancer.[86] World Community Grid also had a project called Help Defeat Cancer. Other related projects include the Folding@home and Rosetta@home projects, which focus on groundbreaking protein folding and protein structure prediction research. Vodafone has also partnered with the Garvan Institute to create the DreamLab Project, which uses distributed computing via an app on cellphones to perform cancer research.
Clinical trials
[edit]
Members of the public can also join clinical trials as healthy control subjects or for methods of cancer detection.
There could be software and data-related procedures that increase participation in trials and make them faster and less expensive. One open source platform matches genomically profiled cancer patients to precision medicine drug trials.[88][87]

Organizations
[edit]
Organizations exist as associations for scientists participating in cancer research, such as the American Association for Cancer Research and American Society of Clinical Oncology, and as foundations for public awareness or raising funds for cancer research, such as Relay For Life and the American Cancer Society.
Awareness campaigns
[edit]Supporters of different types of cancer have adopted different colored awareness ribbons and promote months of the year as being dedicated to the support of specific types of cancer.[89] The American Cancer Society began promoting October as Breast Cancer Awareness Month in the United States in the 1980s. Pink products are sold to both generate awareness and raise money to be donated for research purposes. This has led to pinkwashing, or the selling of ordinary products turned pink as a promotion for the company.
See also
[edit]References
[edit]- ^ "Early Theories about Cancer Causes". American Cancer Society. Archived from the original on 9 May 2018. Retrieved 9 May 2018.
- ^ "Milestone (1971): President Nixon declares war on cancer". dtp.cancer.gov. Archived from the original on 3 December 2017. Retrieved 9 May 2018.
- ^ a b c Song M, Vogelstein B, Giovannucci EL, Willett WC, Tomasetti C (September 2018). "Cancer prevention: Molecular and epidemiologic consensus". Science. 361 (6409): 1317–8. Bibcode:2018Sci...361.1317S. doi:10.1126/science.aau3830. PMC 6260589. PMID 30262488.
- ^ Tran, Khanh Bao; Lang, Justin J.; Compton, Kelly; Xu, Rixing; Acheson, Alistair R.; Henrikson, Hannah Jacqueline; Kocarnik, Jonathan M.; Penberthy, Louise; Aali, Amirali; Abbas, Qamar; et al. (20 August 2022). "The global burden of cancer attributable to risk factors, 2010–19: a systematic analysis for the Global Burden of Disease Study 2019". The Lancet. 400 (10352): 563–591. doi:10.1016/S0140-6736(22)01438-6. PMC 9395583. PMID 35988567.
- ^ Wu S, Powers S, Zhu W, Hannun YA (January 2016). "Substantial contribution of extrinsic risk factors to cancer development". Nature. 529 (7584): 43–7. Bibcode:2016Natur.529...43W. doi:10.1038/nature16166. PMC 4836858. PMID 26675728.
- ^ Degasperi, Andrea; Zou, Xueqing; Dias Amarante, Tauanne; Martinez-Martinez, Andrea; et al. (22 April 2022). "Substitution mutational signatures in whole-genome–sequenced cancers in the UK population". Science. 376 (6591) abl9283. doi:10.1126/science.abl9283. PMC 7613262. PMID 35949260. S2CID 248334490.
- University press release: "Largest study of whole genome sequencing data reveals 'treasure trove' of clues about causes of cancer". University of Cambridge. Retrieved 15 May 2022.
- ^ Quach, Katyanna. "Harvard boffins build multimodal AI system to predict cancer". The Register. Retrieved 16 September 2022.
- ^ Chen, Richard J.; Lu, Ming Y.; Williamson, Drew F. K.; Chen, Tiffany Y.; Lipkova, Jana; Noor, Zahra; Shaban, Muhammad; Shady, Maha; Williams, Mane; Joo, Bumjin; Mahmood, Faisal (8 August 2022). "Pan-cancer integrative histology-genomic analysis via multimodal deep learning". Cancer Cell. 40 (8): 865–878.e6. doi:10.1016/j.ccell.2022.07.004. PMC 10397370. PMID 35944502. S2CID 251456162.
- Teaching hospital press release: "New AI technology integrates multiple data types to predict cancer outcomes". Brigham and Women's Hospital via medicalxpress.com. Retrieved 18 September 2022.
- ^ Zimmer, Carl (29 September 2022). "A New Approach to Spotting Tumors: Look for Their Microbes". The New York Times. Retrieved 19 October 2022.
- ^ Dohlman, Anders B.; Klug, Jared; Mesko, Marissa; Gao, Iris H.; Lipkin, Steven M.; Shen, Xiling; Iliev, Iliyan D. (29 September 2022). "A pan-cancer mycobiome analysis reveals fungal involvement in gastrointestinal and lung tumors". Cell. 185 (20): 3807–22. doi:10.1016/j.cell.2022.09.015. PMC 9564002. PMID 36179671.
- ^ Narunsky-Haziza, Lian; Sepich-Poore, Gregory D.; Livyatan, Ilana; Asraf, Omer; Martino, Cameron; Nejman, Deborah; Gavert, Nancy; Stajich, Jason E.; Amit, Guy; González, Antonio; Wandro, Stephen; Perry, Gili; Ariel, Ruthie; Meltser, Arnon; Shaffer, Justin P.; Zhu, Qiyun; Balint-Lahat, Nora; Barshack, Iris; Dadiani, Maya; Gal-Yam, Einav N.; Patel, Sandip Pravin; Bashan, Amir; Swafford, Austin D.; Pilpel, Yitzhak; Knight, Rob; Straussman, Ravid (29 September 2022). "Pan-cancer analyses reveal cancer-type-specific fungal ecologies and bacteriome interactions". Cell. 185 (20): 3789–3806.e17. doi:10.1016/j.cell.2022.09.005. PMC 9567272. PMID 36179670.
- ^ Piqueret, Baptiste; Montaudon, Élodie; Devienne, Paul; Leroy, Chloé; Marangoni, Elisabetta; Sandoz, Jean-Christophe; d'Ettorre, Patrizia (25 January 2023). "Ants act as olfactory bio-detectors of tumours in patient-derived xenograft mice". Proceedings of the Royal Society B: Biological Sciences. 290 (1991) 20221962. doi:10.1098/rspb.2022.1962. PMC 9874262. PMID 36695032.
- ^ di Pietro A, Tosti G, Ferrucci PF, Testori A (December 2008). "Oncophage: step to the future for vaccine therapy in melanoma". Expert Opinion on Biological Therapy. 8 (12): 1973–84. doi:10.1517/14712590802517970. PMID 18990084. S2CID 83589014.
- ^ Chen, Kok-Siong; Reinshagen, Clemens; Van Schaik, Thijs A.; Rossignoli, Filippo; Borges, Paulo; Mendonca, Natalia Claire; Abdi, Reza; Simon, Brennan; Reardon, David A.; Wakimoto, Hiroaki; Shah, Khalid (4 January 2023). "Bifunctional cancer cell–based vaccine concomitantly drives direct tumor killing and antitumor immunity". Science Translational Medicine. 15 (677) eabo4778. doi:10.1126/scitranslmed.abo4778. PMC 10068810. PMID 36599004. S2CID 255416438.
- ^ "Gene Therapy, Cancer-Killing Viruses And New Drugs Highlight Novel Approaches To Cancer Treatment". Medical News Today. Retrieved 24 April 2007.
- ^ "World first gene therapy trial for leukaemia". LLR. Archived from the original on 2 August 2013. Retrieved 23 July 2013.
- ^ Chinese scientists to pioneer first human CRISPR trial
- ^ Schmidt, Christine K.; Medina-Sánchez, Mariana; Edmondson, Richard J.; Schmidt, Oliver G. (5 November 2020). "Engineering microrobots for targeted cancer therapies from a medical perspective". Nature Communications. 11 (1): 5618. Bibcode:2020NatCo..11.5618S. doi:10.1038/s41467-020-19322-7. ISSN 2041-1723. PMC 7645678. PMID 33154372.
- ^ Gwisai, T.; Mirkhani, N.; Christiansen, M. G.; Nguyen, T. T.; Ling, V.; Schuerle, S. (26 October 2022). "Magnetic torque–driven living microrobots for increased tumor infiltration". Science Robotics. 7 (71) eabo0665. bioRxiv 10.1101/2022.01.03.473989. doi:10.1126/scirobotics.abo0665. ISSN 2470-9476. PMID 36288270. S2CID 253160428.
- ^ Kishore, Chandra; Bhadra, Priyanka (July 2021). "Targeting Brain Cancer Cells by Nanorobot, a Promising Nanovehicle: New Challenges and Future Perspectives". CNS & Neurological Disorders Drug Targets. 20 (6): 531–9. doi:10.2174/1871527320666210526154801. PMID 34042038. S2CID 235217854.
- ^ Contreras-Llano, Luis E.; Liu, Yu-Han; Henson, Tanner; Meyer, Conary C.; Baghdasaryan, Ofelya; Khan, Shahid; Lin, Chi-Long; Wang, Aijun; Hu, Che-Ming J.; Tan, Cheemeng (11 January 2023). "Engineering Cyborg Bacteria Through Intracellular Hydrogelation". Advanced Science. 10 (9) 2204175. doi:10.1002/advs.202204175. ISSN 2198-3844. PMC 10037956. PMID 36628538.
- News report about the study: Firtina, Nergis (1 February 2023). "Semi-living 'cyborg cells' could treat cancer, suggests new study". Interesting Engineering. Archived from the original on 15 February 2023. Retrieved 15 February 2023.
- ^ Lawler, Sean E.; Speranza, Maria-Carmela; Cho, Choi-Fong; Chiocca, E. Antonio (1 June 2017). "Oncolytic Viruses in Cancer Treatment: A Review". JAMA Oncology. 3 (6): 841–9. doi:10.1001/jamaoncol.2016.2064. PMID 27441411. S2CID 39321536.
- ^ Harrington, Kevin; Freeman, Daniel J.; Kelly, Beth; Harper, James; Soria, Jean-Charles (September 2019). "Optimizing oncolytic virotherapy in cancer treatment". Nature Reviews Drug Discovery. 18 (9): 689–706. doi:10.1038/s41573-019-0029-0. ISSN 1474-1784. PMID 31292532. S2CID 256745869.
- ^ Osborne, Margaret. "Small Cancer Trial Resulted in Complete Remission for All Participants". Smithsonian Magazine. Retrieved 21 July 2022.
- ^ Cercek, Andrea; Lumish, Melissa; Sinopoli, Jenna; Weiss, Jill; Shia, Jinru; Lamendola-Essel, Michelle; El Dika, Imane H.; Segal, Neil; Shcherba, Marina; Sugarman, Ryan; Stadler, Zsofia; Yaeger, Rona; Smith, J. Joshua; Rousseau, Benoit; Argiles, Guillem; Patel, Miteshkumar; Desai, Avni; Saltz, Leonard B.; Widmar, Maria; Iyer, Krishna; Zhang, Janie; Gianino, Nicole; Crane, Christopher; Romesser, Paul B.; Pappou, Emmanouil P.; Paty, Philip; Garcia-Aguilar, Julio; Gonen, Mithat; Gollub, Marc; Weiser, Martin R.; Schalper, Kurt A.; Diaz, Luis A. (23 June 2022). "PD-1 Blockade in Mismatch Repair–Deficient, Locally Advanced Rectal Cancer". New England Journal of Medicine. 386 (25): 2363–76. doi:10.1056/NEJMoa2201445. ISSN 0028-4793. PMC 9492301. PMID 35660797. S2CID 249395846.
- ^ "Trastuzumab Deruxtecan Leads to Longer PFS and OS Compared with Chemotherapy in Previously Treated HER2-Low Unresectable or Metastatic Breast Cancer". www.esmo.org. Retrieved 21 July 2022.
- ^ Modi, Shanu; Jacot, William; Yamashita, Toshinari; Sohn, Joohyuk; Vidal, Maria; Tokunaga, Eriko; Tsurutani, Junji; Ueno, Naoto T.; Prat, Aleix; Chae, Yee Soo; Lee, Keun Seok; Niikura, Naoki; Park, Yeon Hee; Xu, Binghe; Wang, Xiaojia; Gil-Gil, Miguel; Li, Wei; Pierga, Jean-Yves; Im, Seock-Ah; Moore, Halle C. F.; Rugo, Hope S.; Yerushalmi, Rinat; Zagouri, Flora; Gombos, Andrea; Kim, Sung-Bae; Liu, Qiang; Luo, Ting; Saura, Cristina; Schmid, Peter; Sun, Tao; Gambhire, Dhiraj; Yung, Lotus; Wang, Yibin; Singh, Jasmeet; Vitazka, Patrik; Meinhardt, Gerold; Harbeck, Nadia; Cameron, David A. (5 June 2022). "Trastuzumab Deruxtecan in Previously Treated HER2-Low Advanced Breast Cancer". New England Journal of Medicine. 387 (1): 9–20. doi:10.1056/NEJMoa2203690. hdl:2445/197309. PMC 10561652. PMID 35665782. S2CID 249418284.
- ^ "Scientists harness light therapy to target and kill cancer cells in world first". The Guardian. 17 June 2022. Retrieved 21 June 2022.
- ^ Mączyńska, Justyna; Raes, Florian; Da Pieve, Chiara; Turnock, Stephen; Boult, Jessica K. R.; Hoebart, Julia; Niedbala, Marcin; Robinson, Simon P.; Harrington, Kevin J.; Kaspera, Wojciech; Kramer-Marek, Gabriela (21 January 2022). "Triggering anti-GBM immune response with EGFR-mediated photoimmunotherapy". BMC Medicine. 20 (1): 16. doi:10.1186/s12916-021-02213-z. ISSN 1741-7015. PMC 8780306. PMID 35057796.
- News release: "Light-activated 'photoimmunotherapy' could enhance brain cancer treatment". Institute of Cancer Research. 16 June 2022. Archived from the original on 20 June 2022. Retrieved 21 June 2022.
- ^ a b Cerwenka A, Lanier LL (February 2016). "Natural killer cell memory in infection, inflammation and cancer". Nature Reviews. Immunology. 16 (2): 112–123. doi:10.1038/nri.2015.9. PMID 26806484. S2CID 361806.
- ^ Zhu, Shaoming; Zhang, Tian; Zheng, Lei; Liu, Hongtao; Song, Wenru; Liu, Delong; Li, Zihai; Pan, Chong-xian (December 2021). "Combination strategies to maximize the benefits of cancer immunotherapy". Journal of Hematology & Oncology. 14 (1): 156. doi:10.1186/s13045-021-01164-5. PMC 8475356. PMID 34579759.
- ^ Futreal PA, Coin L, Marshall M, Down T, Hubbard T, Wooster R, Rahman N, Stratton MR (March 2004). "A census of human cancer genes". Nature Reviews. Cancer. 4 (3): 177–183. doi:10.1038/nrc1299. PMC 2665285. PMID 14993899.
- ^ Forbes S, Clements J, Dawson E, Bamford S, Webb T, Dogan A, Flanagan A, Teague J, Wooster R, Futreal PA, Stratton MR (January 2006). "COSMIC 2005". British Journal of Cancer. 94 (2): 318–322. doi:10.1038/sj.bjc.6602928. PMC 2361125. PMID 16421597.
- ^ Vlahopoulos SA, Logotheti S, Mikas D, Giarika A, Gorgoulis V, Zoumpourlis V.The role of ATF-2 in oncogenesis" Bioessays 2008 Apr;30(4) 314-27.
- ^ Sharma S, Kelly TK, Jones PA (January 2010). "Epigenetics in cancer". Carcinogenesis. 31 (1): 27–36. doi:10.1093/carcin/bgp220. PMC 2802667. PMID 19752007.
- ^ Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA, Kinzler KW (March 2013). "Cancer genome landscapes". Science. 339 (6127): 1546–1558. Bibcode:2013Sci...339.1546V. doi:10.1126/science.1235122. PMC 3749880. PMID 23539594.
- ^ Illingworth RS, Gruenewald-Schneider U, Webb S, Kerr AR, James KD, Turner DJ, et al. (September 2010). "Orphan CpG islands identify numerous conserved promoters in the mammalian genome". PLOS Genetics. 6 (9) e1001134. doi:10.1371/journal.pgen.1001134. PMC 2944787. PMID 20885785.
- ^ Wei J, Li G, Dang S, Zhou Y, Zeng K, Liu M (2016). "Discovery and Validation of Hypermethylated Markers for Colorectal Cancer". Disease Markers. 2016 2192853. doi:10.1155/2016/2192853. PMC 4963574. PMID 27493446.
- ^ Beggs AD, Jones A, El-Bahrawy M, El-Bahwary M, Abulafi M, Hodgson SV, Tomlinson IP (April 2013). "Whole-genome methylation analysis of benign and malignant colorectal tumours". The Journal of Pathology. 229 (5): 697–704. doi:10.1002/path.4132. PMC 3619233. PMID 23096130.
- ^ Novak K (December 2004). "Epigenetics changes in cancer cells". MedGenMed. 6 (4): 17. PMC 1480584. PMID 15775844.
- ^ Banno K, Kisu I, Yanokura M, Tsuji K, Masuda K, Ueki A, et al. (September 2012). "Epimutation and cancer: a new carcinogenic mechanism of Lynch syndrome (Review)". International Journal of Oncology. 41 (3): 793–797. doi:10.3892/ijo.2012.1528. PMC 3582986. PMID 22735547.
- ^ a b c Wicki A, Hagmann J (9 September 2011). "Diet and cancer". Swiss Medical Weekly. 141: w13250. doi:10.4414/smw.2011.13250. PMID 21904992.
- ^ Papadimitriou N, Markozannes G, Kanellopoulou A, Critselis E, Alhardan S, Karafousia V, Kasimis JC, Katsaraki C, Papadopoulou A, Zografou M, Lopez DS, Chan DS, Kyrgiou M, Ntzani E, Cross AJ, Marrone MT, Platz EA, Gunter MJ, Tsilidis KK (2021). "An umbrella review of the evidence associating diet and cancer risk at 11 anatomical sites". Nature Communications. 12 (1): 4579. Bibcode:2021NatCo..12.4579P. doi:10.1038/s41467-021-24861-8. PMC 8319326. PMID 34321471.
- ^ Jabbari M, Pourmoradian S, Eini-Zinab H, Mosharkesh E, Hosseini Balam F, Yaghmaei Y, Yadegari A, Amini B, Arman Moghadam D, Barati M, Hekmatdoost A (2022). "Levels of evidence for the association between different food groups/items consumption and the risk of various cancer sites: an umbrella review". Int J Food Sci Nutr. 73 (7): 861–874. doi:10.1080/09637486.2022.2103523. PMID 35920747. S2CID 251280745.
- ^ Stewart BW, Wild CP, eds. (2014). "Ch. 2: Cancer Etiology § 6 Diet, obesity and physical activity". World Cancer Report 2014. World Health Organization. pp. 124–33. ISBN 978-92-832-0429-9.
- ^ Vieira AR, Abar L, Chan DSM, Vingeliene S, Polemiti E, Stevens C, Greenwood D, Norat T. (2017). "Foods and beverages and colorectal cancer risk: a systematic review and meta-analysis of cohort studies, an update of the evidence of the WCRF-AICR Continuous Update Project". Annals of Oncology. 28 (8): 1788–1802. doi:10.1093/annonc/mdx171. hdl:10044/1/48313. PMID 28407090.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "Meat, fish, dairy and cancer risk". wcrf.org. Retrieved 24 April 2023.
- ^ "Red Meat and Processed Meat Consumption". progressreport.cancer.gov. Retrieved 24 April 2023.
- ^ "Red Meat (Beef, Pork, Lamb): Increases Risk of Colorectal Cancer". aicr.org. Retrieved 24 April 2023.
- ^ a b c Park S, Bae J, Nam BH, Yoo KY (2008). "Aetiology of cancer in Asia" (PDF). Asian Pacific Journal of Cancer Prevention. 9 (3): 371–380. PMID 18990005.
- ^ Yu C, Cao Q, Chen P, Yang S, Deng M, Wang Y, Li L (December 2016). "An updated dose-response meta-analysis of coffee consumption and liver cancer risk". Scientific Reports. 6 (1) 37488. Bibcode:2016NatSR...637488Y. doi:10.1038/srep37488. PMC 5133591. PMID 27910873.
- ^ Brenner H, Rothenbacher D, Arndt V (2009). "Epidemiology of Stomach Cancer". In Mukesh V (ed.). Cancer Epidemiology. Methods in Molecular Biology. Vol. 472. pp. 467–477. doi:10.1007/978-1-60327-492-0_23. ISBN 978-1-60327-491-3. PMC 2166976. PMID 19107449.
- ^ "Preventing Cancer". hsph.harvard.edu. Retrieved 24 April 2023.
- ^ "A healthy diet alone will not cure cancer". National Academies of Sciences, Engineering, and Medicine. 2024. Archived from the original on 28 April 2024.
- ^ Ilerhunmwuwa NP, Abdul Khader AHS, Smith C, Cliff ERS, Booth CM, Hottel E, Aziz M, Lee-Smith W, Goodman A, Chakraborty R, Mohyuddin GR. (2024). "Dietary interventions in cancer: a systematic review of all randomized controlled trials". Journal of the National Cancer Institute. 116 (7): 1026–1034. doi:10.1093/jnci/djae051. PMC 11223872. PMID 38429997.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Clifton, Katherine K.; Ma, Cynthia X.; Fontana, Luigi; Peterson, Lindsay L. (November 2021). "Intermittent fasting in the prevention and treatment of cancer". CA: A Cancer Journal for Clinicians. 71 (6): 527–546. doi:10.3322/caac.21694. ISSN 0007-9235. PMID 34383300. S2CID 236989849.
- ^ Manoogian, Emily N. C.; Panda, Satchidananda (1 October 2017). "Circadian rhythms, time-restricted feeding, and healthy aging". Ageing Research Reviews. 39: 59–67. doi:10.1016/j.arr.2016.12.006. ISSN 1568-1637. PMC 5814245. PMID 28017879.
- ^ Brandhorst, Sebastian; Longo, Valter D. (2016). "Fasting and Caloric Restriction in Cancer Prevention and Treatment". Metabolism in Cancer. Recent Results in Cancer Research. Vol. 207. Springer. pp. 241–266. doi:10.1007/978-3-319-42118-6_12. ISBN 978-3-319-42116-2. PMC 7476366. PMID 27557543. S2CID 42198775.
- ^ Alidadi, Mona; Banach, Maciej; Guest, Paul C.; Bo, Simona; Jamialahmadi, Tannaz; Sahebkar, Amirhossein (1 August 2021). "The effect of caloric restriction and fasting on cancer". Seminars in Cancer Biology. 73: 30–44. doi:10.1016/j.semcancer.2020.09.010. ISSN 1044-579X. PMID 32977005. S2CID 221938415.
- ^ Ibrahim, Ezzeldin M.; Al-Foheidi, Meteb H.; Al-Mansour, Mubarak M. (1 May 2021). "Energy and caloric restriction, and fasting and cancer: a narrative review". Supportive Care in Cancer. 29 (5): 2299–2304. doi:10.1007/s00520-020-05879-y. PMC 7981322. PMID 33190181. S2CID 226945778.
- ^ Hofer, Sebastian J.; Davinelli, Sergio; Bergmann, Martina; Scapagnini, Giovanni; Madeo, Frank (2021). "Caloric Restriction Mimetics in Nutrition and Clinical Trials". Frontiers in Nutrition. 8 717343. doi:10.3389/fnut.2021.717343. PMC 8450594. PMID 34552954.
- ^ Madeo, Frank; Eisenberg, Tobias; Pietrocola, Federico; Kroemer, Guido (26 January 2018). "Spermidine in health and disease". Science. 359 (6374) eaan2788. doi:10.1126/science.aan2788. ISSN 0036-8075. PMID 29371440. S2CID 206659415.
- ^ Imran, Muhammad; Aslam Gondal, Tanweer; Atif, Muhammad; Shahbaz, Muhammad; Batool Qaisarani, Tahira; Hanif Mughal, Muhammad; Salehi, Bahare; Martorell, Miquel; Sharifi-Rad, Javad (August 2020). "Apigenin as an anticancer agent". Phytotherapy Research. 34 (8): 1812–28. doi:10.1002/ptr.6647. ISSN 0951-418X. PMID 32059077. S2CID 211122428.
- ^ Shukla, Sanjeev; Gupta, Sanjay (1 June 2010). "Apigenin: A Promising Molecule for Cancer Prevention". Pharmaceutical Research. 27 (6): 962–978. doi:10.1007/s11095-010-0089-7. PMC 2874462. PMID 20306120.
- ^ Shankar, Eswar; Goel, Aditi; Gupta, Karishma; Gupta, Sanjay (1 December 2017). "Plant Flavone Apigenin: an Emerging Anticancer Agent". Current Pharmacology Reports. 3 (6): 423–446. doi:10.1007/s40495-017-0113-2. PMC 5791748. PMID 29399439.
- ^ Samadi, Parisa; Sarvarian, Parisa; Gholipour, Elham; Asenjan, Karim Shams; Aghebati-Maleki, Leili; Motavalli, Roza; Hojjat-Farsangi, Mohammad; Yousefi, Mehdi (October 2020). "Berberine: A novel therapeutic strategy for cancer". IUBMB Life. 72 (10): 2065–79. doi:10.1002/iub.2350. ISSN 1521-6543. PMID 32735398. S2CID 220893166.
- ^ Zhong, Xiao-Dan; Chen, Li-Juan; Xu, Xin-Yang; Liu, Yan-Jun; Tao, Fan; Zhu, Ming-Hui; Li, Chang-Yun; Zhao, Dan; Yang, Guan-Jun; Chen, Jiong (2022). "Berberine as a potential agent for breast cancer therapy". Frontiers in Oncology. 12 993775. doi:10.3389/fonc.2022.993775. PMC 9480097. PMID 36119505.
- ^ Wang, Ye; Liu, Yanfang; Du, Xinyang; Ma, Hong; Yao, Jing (30 January 2020). "The Anti-Cancer Mechanisms of Berberine: A Review". Cancer Management and Research. 12: 695–702. doi:10.2147/CMAR.S242329. PMC 6996556. PMID 32099466.
- ^ Vlavcheski, Filip; O'Neill, Eric J.; Gagacev, Filip; Tsiani, Evangelia (January 2022). "Effects of Berberine against Pancreatitis and Pancreatic Cancer". Molecules. 27 (23): 8630. doi:10.3390/molecules27238630. PMC 9738201. PMID 36500723.
- ^ Guamán Ortiz, Luis Miguel; Lombardi, Paolo; Tillhon, Micol; Scovassi, Anna Ivana (August 2014). "Berberine, an Epiphany Against Cancer". Molecules. 19 (8): 12349–67. doi:10.3390/molecules190812349. PMC 6271598. PMID 25153862.
- ^ Su, Chao; Li, Nan; Ren, Ruru; Wang, Yingli; Su, Xiaojuan; Lu, Fangfang; Zong, Rong; Yang, Lingling; Ma, Xueqin (January 2021). "Progress in the Medicinal Value, Bioactive Compounds, and Pharmacological Activities of Gynostemma pentaphyllum". Molecules. 26 (20): 6249. doi:10.3390/molecules26206249. PMC 8540791. PMID 34684830.
- ^ Pu, Wei-ling; Zhang, Meng-ying; Bai, Ru-yu; Sun, Li-kang; Li, Wen-hua; Yu, Ying-li; Zhang, Yue; Song, Lei; Wang, Zhao-xin; Peng, Yan-fei; Shi, Hong; Zhou, Kun; Li, Tian-xiang (1 January 2020). "Anti-inflammatory effects of Rhodiola rosea L.: A review". Biomedicine & Pharmacotherapy. 121 109552. doi:10.1016/j.biopha.2019.109552. ISSN 0753-3322. PMID 31715370. S2CID 207938536.
- ^ Magani, Sri Krishna Jayadev; Mupparthi, Sri Durgambica; Gollapalli, Bhanu Prakash; Shukla, Dhananjay; Tiwari, A. K.; Gorantala, Jyotsna; Yarla, Nagendra Sastry; Tantravahi, Srinivasan (2020). "Salidroside — Can it be a Multifunctional Drug?". Current Drug Metabolism. 21 (7): 512–524. doi:10.2174/1389200221666200610172105. PMID 32520682. S2CID 219588131.
- ^ "Federally Funded Cancer Research". asco.org. 8 February 2016. Archived from the original on 23 April 2018. Retrieved 9 May 2018.
- ^ a b c d Eckhouse S, Sullivan R (July 2006). "A survey of public funding of cancer research in the European union". PLOS Medicine. 3 (7) e267. doi:10.1371/journal.pmed.0030267. PMC 1513045. PMID 16842021.
- ^ "NCI Budget Fact Book". National Cancer Institute. 10 May 2022.
- ^ Alberts B, Kirschner MW, Tilghman S, Varmus H (April 2014). "Rescuing US biomedical research from its systemic flaws". Proceedings of the National Academy of Sciences of the United States of America. 111 (16): 5773–7. Bibcode:2014PNAS..111.5773A. doi:10.1073/pnas.1404402111. PMC 4000813. PMID 24733905.
- ^ Kolata G (23 April 2009). "Advances Elusive in the Drive to Cure Cancer". The New York Times. Archived from the original on 14 January 2012. Retrieved 29 December 2009.
- ^ Kolata G (27 June 2009). "Grant System Leads Cancer Researchers to Play It Safe". The New York Times. Archived from the original on 8 June 2011. Retrieved 29 December 2009.
- ^ Leaf C (22 March 2004). "Why We're Losing The War on Cancer". Fortune Magazine (CNN Money). Archived from the original on 2 May 2014.
- ^ Begley CG, Ellis LM (March 2012). "Drug development: Raise standards for preclinical cancer research". Nature (Comment article). 483 (7391): 531–533. Bibcode:2012Natur.483..531B. doi:10.1038/483531a. PMID 22460880. S2CID 4326966. (Erratum: doi:10.1038/485041e)
- ^ Haelle T (7 December 2021). "Dozens of major cancer studies can't be replicated". Science News. Retrieved 19 January 2022.
- ^ "Reproducibility Project: Cancer Biology". www.cos.io. Center for Open Science. Retrieved 19 January 2022.
- ^ Mobley A, Linder SK, Braeuer R, Ellis LM, Zwelling L (2013). Arakawa H (ed.). "A survey on data reproducibility in cancer research provides insights into our limited ability to translate findings from the laboratory to the clinic". PLOS ONE. 8 (5) e63221. Bibcode:2013PLoSO...863221M. doi:10.1371/journal.pone.0063221. PMC 3655010. PMID 23691000.
- ^ Van Noorden R (July 2023). "Medicine is plagued by untrustworthy clinical trials. How many studies are faked or flawed?". Nature. 619 (7970): 454–458. Bibcode:2023Natur.619..454V. doi:10.1038/d41586-023-02299-w. PMID 37464079.
- ^ "Help Conquer Cancer". 19 November 2007. Archived from the original on 16 November 2007. Retrieved 19 November 2007.
- ^ a b Klein, Harry; Mazor, Tali; Siegel, Ethan; Trukhanov, Pavel; Ovalle, Andrea; Vecchio Fitz, Catherine Del; Zwiesler, Zachary; Kumari, Priti; Van Der Veen, Bernd; Marriott, Eric; Hansel, Jason; Yu, Joyce; Albayrak, Adem; Barry, Susan; Keller, Rachel B.; MacConaill, Laura E.; Lindeman, Neal; Johnson, Bruce E.; Rollins, Barrett J.; Do, Khanh T.; Beardslee, Brian; Shapiro, Geoffrey; Hector-Barry, Suzanne; Methot, John; Sholl, Lynette; Lindsay, James; Hassett, Michael J.; Cerami, Ethan (6 October 2022). "MatchMiner: an open-source platform for cancer precision medicine". npj Precision Oncology. 6 (1): 69. doi:10.1038/s41698-022-00312-5. PMC 9537311. PMID 36202909.
- ^ "Researchers report genomic profiling from more than 110,000 tumors". News-Medical.net. 19 July 2022. Retrieved 20 November 2022.
- ^ "Cancer Awareness Dates". cancer.net. 19 December 2013. Archived from the original on 9 December 2017. Retrieved 9 May 2018.
External links
[edit]Cancer research
View on GrokipediaHistorical Development
Pre-20th Century Foundations
The earliest documented observations of cancer date to ancient Egypt around 3000 BC, as recorded in the Edwin Smith Papyrus, which describes a case of breast cancer with bulging tumors but offers no effective treatment beyond palliative measures like cauterization.[8] Additional Egyptian papyri from the period, numbering seven and deciphered in the late 19th century, provide further descriptions of tumors, attributing them to supernatural causes or recommending surgical excision when possible, though outcomes were limited by rudimentary tools and lack of anesthesia.[9] In classical antiquity, Hippocrates (c. 460–370 BC), often called the Father of Medicine, advanced understanding by rejecting divine origins and proposing a humoral theory, positing that cancer arose from imbalances in the body's four humors—blood, phlegm, yellow bile, and black bile—leading to excess black bile in tumors.[10] He coined the term carcinoma (from Greek karkinos, meaning crab) to describe malignant tumors, noting their crab-like veins extending outward, and distinguished between benign and malignant growths while advocating cautious surgery for localized cases, avoiding intervention in advanced or ulcerated ones due to poor prognosis.[9] Galen (c. 129–216 AD) built on this framework, reinforcing the humoral imbalance as causal and influencing medical thought for over a millennium, though empirical evidence remained anecdotal, derived from dissections and clinical observations without systematic pathology.[11] Medieval and Renaissance periods saw minimal progress, with humoral theory persisting amid religious prohibitions on dissection, though figures like Andreas Vesalius (1514–1564) improved anatomical accuracy through autopsies, occasionally noting cancerous growths without altering etiological views.[8] By the 18th century, Percivall Pott's 1775 report linked chronic exposure to chimney soot with scrotal cancer in young chimney sweeps, marking the first recognition of an environmental carcinogen and shifting focus toward external causes over purely internal imbalances.[4] The 19th century introduced microscopy and cellular insights, enabling pathologists to examine tissues at the microscopic level and replace gross descriptions with cellular analysis.[12] Rudolf Virchow's 1858 work Cellular Pathology, based on lectures in Berlin, established that all cells arise from pre-existing cells (omnis cellula e cellula) and that diseases, including cancer, originate from abnormal cellular proliferation rather than systemic fluid imbalances, laying the groundwork for modern oncology by emphasizing localized cellular dysfunction as the primary mechanism.[13][14] This cellular paradigm, supported by histological evidence from autopsies and biopsies, refuted earlier holistic theories and enabled classification of cancers by tissue origin, though causation remained elusive without genetic knowledge.[15]20th Century Breakthroughs
Early 20th-century research established key etiological mechanisms of cancer. In 1902, Theodor Boveri hypothesized that malignant tumors originate from a single cell with chromosomal damage leading to uncontrolled proliferation.[4] Peyton Rous identified in 1911 a virus capable of inducing sarcomas in chickens, providing the first evidence of viral oncogenesis and earning him the Nobel Prize in Physiology or Medicine in 1966.[4] In 1915, Katsusaburo Yamagiwa and Koichi Ichikawa experimentally induced skin cancer in rabbits using coal tar, confirming the role of chemical carcinogens.[4] Mid-century advances shifted focus to therapeutic interventions and cytogenetics. During World War II, nitrogen mustard derivatives from mustard gas observations were first administered in 1942 to a patient with lymphoma, initiating modern chemotherapy by demonstrating cytotoxic effects on cancer cells.[16] Sidney Farber reported in 1948 temporary remissions in children with acute lymphoblastic leukemia using aminopterin, a folic acid antagonist, marking the first chemotherapeutic success against leukemia.[17] Epidemiological studies by Richard Doll and others in 1950 established cigarette smoking as a primary cause of lung cancer through cohort analyses showing dose-dependent risk.[4] The 1960 discovery of the Philadelphia chromosome by Peter Nowell and David Hungerford in chronic myeloid leukemia cells revealed the first consistent cytogenetic abnormality in a human cancer.[4] Later decades unveiled molecular foundations. Researchers in 1976 identified proto-oncogenes in normal cells analogous to viral oncogenes, demonstrating how mutations in cellular genes drive tumorigenesis, as recognized by the 1989 Nobel Prize to J. Michael Bishop and Harold Varmus.[4] The TP53 gene, a critical tumor suppressor frequently mutated in cancers, was cloned in 1979.[4] In 1984, the HER2 oncogene was discovered and linked to aggressive breast cancers, enabling subsequent targeted therapies, while human papillomavirus types 16 and 18 were associated with cervical cancer.[4] These findings transitioned cancer research toward genetic and molecular paradigms.Post-1971 War on Cancer Era
The National Cancer Act of 1971, signed into law by President Richard Nixon on December 23, dramatically expanded the National Cancer Institute's (NCI) authority, budget, and infrastructure, marking the formal declaration of the "War on Cancer." This legislation increased federal cancer research funding from approximately $500 million in fiscal year 1972 to over $6.5 billion by 2021 (adjusted for inflation in later estimates), enabling the establishment of a network of comprehensive cancer centers, cooperative clinical trial groups, and enhanced data-sharing systems.[18][19] The act prioritized basic research into cancer causation alongside applied efforts in treatment and prevention, fostering interdisciplinary collaborations that accelerated discoveries in molecular oncology.[20] Post-1971 research yielded pivotal insights into cancer's genetic underpinnings, building on earlier viral studies to identify cellular oncogenes in the late 1970s and tumor suppressor genes like RB1 in retinoblastoma by 1986. The 1980s and 1990s saw the elucidation of key signaling pathways, such as RAS and TP53 mutations, which underpin uncontrolled proliferation, informing the development of the first targeted therapies like trastuzumab for HER2-positive breast cancer, approved by the FDA in 1998.[4] The Human Genome Project's completion in 2003 facilitated large-scale genomic initiatives, including the NCI's Cancer Genome Atlas (TCGA) launched in 2006, which cataloged somatic mutations across dozens of cancer types, revealing actionable drivers in over 30% of cases for precision medicine.[21] Immunotherapy emerged as a transformative paradigm in the 2010s, with FDA approvals for ipilimumab (a CTLA-4 inhibitor) in 2011 for melanoma and pembrolizumab (a PD-1 inhibitor) in 2014 for advanced cancers, harnessing T-cell responses to achieve durable remissions in subsets of patients previously unresponsive to chemotherapy. These advances, alongside refined chemotherapy regimens and radiation techniques, contributed to empirical gains: U.S. age-adjusted cancer mortality declined by 32% from 1991 to 2019, with five-year survival rates rising from 49% in 1975 to 68% by 2018, driven largely by reductions in smoking-related lung cancer and improved early detection for breast, prostate, and colorectal cancers. Childhood leukemia survival exceeded 90% by the 2010s, up from near-zero fatality pre-1970s.[22][23][24] Despite these metrics, the era has faced scrutiny for underwhelming progress against common epithelial cancers like pancreatic and lung adenocarcinoma, where five-year survival remains below 20% for metastatic disease, reflecting high drug development failure rates (over 90% in phase III trials) and tumor heterogeneity evading single-target interventions. Critics, including analyses from NCI data, argue the "war" metaphor fostered overpromising of imminent cures, diverting resources from prevention—responsible for up to 50% of reductions—toward high-cost treatments amid stagnant overall incidence rises due to aging populations. Reproducibility challenges surfaced prominently in the 2010s, with studies showing only 46% of landmark preclinical cancer papers replicable, attributed to selective reporting and insufficient statistical rigor, prompting initiatives like the Reproducibility Project: Cancer Biology. Mainstream academic sources often underemphasize these systemic issues, potentially due to funding dependencies, while independent reviews highlight how entrenched incentives prioritize novel therapies over rigorous validation.[25][26][27] ![Graphic of results and barriers. 193 experiments were designed, 87 were initiated, and 50 were completed.][center]This figure illustrates replication barriers in cancer biology experiments, underscoring methodological hurdles in post-1971 research validation.[4]
Underlying Mechanisms of Cancer
Genetic Mutations and Oncogenes
Genetic mutations constitute a primary driver of cancer initiation and progression, with somatic alterations in proto-oncogenes converting them into oncogenes that confer a growth advantage to affected cells. Proto-oncogenes encode proteins critical for normal cellular processes such as signal transduction, transcription, and cell cycle regulation; their dysregulation typically requires only a single "gain-of-function" hit, unlike the biallelic inactivation needed for most tumor suppressor genes. Mechanisms of activation include point mutations that lock proteins in an active state, gene amplification increasing dosage, chromosomal translocations creating fusion proteins, or viral insertions, as initially observed in retroviral oncogenesis. These changes enable oncogenes to sustain proliferative signaling, a core hallmark of cancer, independent of external growth factors.[28][29] The RAS gene family exemplifies oncogenic activation via point mutations, particularly at codons 12, 13, or 61, which impair GTPase activity and result in persistent downstream signaling through MAPK and PI3K pathways. KRAS mutations occur in roughly 90% of pancreatic ductal adenocarcinomas, 40% of colorectal cancers, and 30% of lung adenocarcinomas, making it one of the most prevalent oncogenic drivers across epithelial malignancies. Similarly, BRAF V600E mutations, activating the RAF kinase in the MAPK cascade, are found in over 50% of melanomas and 10-15% of colorectal cancers, often amenable to targeted inhibition. MYC amplification or translocation, as in Burkitt lymphoma via t(8;14), drives transcriptional upregulation of proliferation genes, contributing to 10-20% of high-grade lymphomas and various solid tumors. These examples illustrate how specific mutations confer tissue-specific oncogenic potency, with pan-cancer analyses identifying fewer than 100 recurrently mutated oncogenes amid thousands of passenger variants.[30][31][29] The discovery of cellular oncogenes stemmed from retroviral research, with J. Michael Bishop and Harold E. Varmus demonstrating in 1976 that the src oncogene in Rous sarcoma virus derives from a normal cellular proto-oncogene (c-src), challenging the prevailing view of cancer as purely viral. This work, extended to human cancers through transfection assays identifying activated RAS in 1980s bladder and lung tumors, established the somatic mutation theory and earned the 1989 Nobel Prize in Physiology or Medicine. Subsequent genomic sequencing has revealed that advanced cancers harbor an average of two to three driver mutations in oncogenes or related pathways, underscoring the multi-hit nature of tumorigenesis while highlighting oncogene "addiction," wherein cancer cells become dependent on sustained oncogene activity for survival, a vulnerability exploited in therapies like EGFR inhibitors in non-small cell lung cancer. Despite these advances, challenges persist, as many oncogenes exhibit context-dependent essentiality, functioning as drivers only in specific tumor subtypes rather than universally.[29][32][33]Epigenetic and Non-Genetic Factors
Epigenetic modifications regulate gene expression through mechanisms independent of DNA sequence alterations, including DNA methylation at cytosine residues, post-translational histone modifications such as acetylation and methylation, and non-coding RNA-mediated silencing. These processes maintain cellular identity and respond to environmental cues but become dysregulated in cancer, cooperating with genetic mutations to promote tumorigenesis. Aberrant epigenetics contribute to all hallmarks of cancer, from sustained proliferation to metastasis, by enabling reversible switches in gene activity that confer adaptive advantages to tumor cells.[34] DNA methylation patterns in cancer exhibit paradoxical features: genome-wide hypomethylation, which activates oncogenes and transposable elements leading to chromosomal instability, alongside hypermethylation of CpG islands in promoter regions, which silences tumor suppressor genes. For instance, hypermethylation of the MLH1 promoter in colorectal cancers results in microsatellite instability and defective DNA mismatch repair, observed in approximately 15% of sporadic cases. Similarly, hypermethylation of MGMT in gliomas impairs DNA repair and sensitizes cells to alkylating agents, with prevalence rates up to 40-50% in glioblastoma. These changes arise cumulatively during carcinogenesis, often initiated by environmental exposures like tobacco smoke or dietary factors that alter methyl donor availability.[35][36] Histone modifications further disrupt chromatin architecture in cancer, with reduced acetylation (via histone deacetylase overexpression) compacting chromatin and repressing transcription of antiproliferative genes, while aberrant methylation (e.g., H3K27me3 enrichment) maintains stem-like states in tumor-initiating cells. In breast cancer, enhancer of zeste homolog 2 (EZH2), a histone methyltransferase, is upregulated in over 50% of cases, driving polycomb repressive complex activity that silences developmental regulators and promotes invasion. Non-coding RNAs, particularly microRNAs, fine-tune these networks; downregulation of miR-200 family members in epithelial-mesenchymal transition enhances metastasis by derepressing ZEB1/2 transcription factors.[37][38] Non-genetic factors beyond core epigenetics, such as phenotypic plasticity and microenvironmental influences, enable cancer cell adaptation without mutational change. Stochastic fluctuations in gene expression generate epigenetic heterogeneity within clones, allowing rapid selection for resistant subpopulations under therapy, as evidenced by single-cell analyses showing non-genetic state transitions driving dormancy or senescence escape in leukemia models. Tumor microenvironment components, including hypoxia-inducible factors and inflammatory cytokines, induce epigenetic reprogramming; chronic inflammation via NF-κB signaling promotes histone acetylation at oncogenes in hepatocellular carcinoma. Exposomal elements like diet-derived metabolites (e.g., folate deficiency altering methylation) or viral infections (e.g., EBV altering host epigenomes) further mediate these effects, underscoring causality from external perturbations to heritable cellular states. While genetic lesions provide initiating hits, non-genetic mechanisms amplify progression and resistance, with empirical models demonstrating their sufficiency in mutation-bearing cells to initiate tumors under selective pressure.[39][40][41]Hallmarks of Cancer Biology
The hallmarks of cancer encapsulate the core biological capabilities that distinguish malignant cells from normal ones, enabling tumor initiation, progression, and metastasis. Proposed by Douglas Hanahan and Robert A. Weinberg in their 2000 Cell paper, the original framework outlined six essential traits acquired through genetic and epigenetic alterations during multistep carcinogenesis.[42] This conceptualization has guided cancer biology research by focusing on functional outcomes rather than isolated molecular events, emphasizing how these capabilities interact to drive neoplastic transformation.[43] The six founding hallmarks are:- Sustaining proliferative signaling: Cancer cells reprogram signaling circuits to maintain persistent mitogenic stimulation, often via oncogenic mutations in receptor tyrosine kinases or downstream effectors like RAS, decoupling growth from external cues.[42]
- Evading growth suppressors: Tumors counteract antigrowth signals from tumor suppressor genes such as TP53 and RB1, which normally enforce cell cycle arrest or senescence in response to stress.[42]
- Resisting cell death: Neoplastic cells acquire resistance to apoptosis through inactivation of pro-apoptotic pathways (e.g., BCL-2 family dysregulation) or overexpression of anti-apoptotic proteins, allowing survival despite oncogenic stress.[42]
- Enabling replicative immortality: Cancer cells overcome telomere shortening via telomerase reactivation or alternative lengthening mechanisms, permitting indefinite proliferation without senescence.[42]
- Inducing angiogenesis: Tumors stimulate new blood vessel formation by secreting pro-angiogenic factors like VEGF, ensuring nutrient and oxygen supply for expansive growth.[42]
- Activating invasion and metastasis: Malignant cells gain motility and invasiveness, breaching tissue barriers and disseminating to distant sites via epithelial-mesenchymal transition and extracellular matrix remodeling.[42]
Methodological Approaches
Basic Laboratory Research
Basic laboratory research in cancer biology investigates the molecular and cellular distinctions between normal and malignant cells to uncover mechanisms driving uncontrolled proliferation, oncogenic transformation, and metastatic dissemination. These studies, typically conducted in vitro or in vivo under controlled conditions, inform hypotheses about carcinogenesis and potential therapeutic vulnerabilities without direct patient involvement.[1] Core techniques encompass cell culture systems, ranging from monolayer cultures of established lines like HeLa or MCF-7 to advanced three-dimensional organoids that better mimic tumor microenvironments and enable examination of invasion and drug responses.[1] Tissue-engineered platforms and ex vivo models further replicate spatial organization and stromal interactions observed in tumors.[1] Genetically engineered mouse models (GEMMs) integrate human-relevant mutations to simulate spontaneous tumor development, facilitating analysis of initiation events, immune evasion, angiogenesis, and metastasis in a physiological context. Since their refinement in the 1990s and 2000s, GEMMs have validated oncogenes and tumor suppressors, assessed microenvironmental influences, and tested preclinical interventions.[48][49][50] CRISPR-Cas9, adapted for mammalian cells around 2013, enables precise gene editing to probe causal roles of specific alterations in cancer phenotypes, with applications in functional validation and high-complexity pooled screens identifying synthetic lethalities or resistance drivers. By 2020, CRISPR had integrated into routine cancer studies for modeling dependencies and editing therapy-resistant clones.[51][52][53] High-throughput screening (HTS) platforms automate testing of vast compound libraries or RNAi/CRISPR libraries against cancer cell panels to pinpoint modulators of proliferation, apoptosis, or pathway activity. In colorectal cancer models, targeted HTS has yielded pathway-specific inhibitors, accelerating hit-to-lead processes in early discovery.[54][55] Reproducibility challenges persist, as evidenced by the Reproducibility Project: Cancer Biology (2013–2021), which planned 193 experiments from prominent publications but initiated only 87 and completed 50 due to methodological gaps, reagent variability, and logistical hurdles.[56][6] Independent assessments indicate low replication rates for many preclinical findings, prompting calls for standardized protocols and preregistration to enhance reliability.[57][7]Translational and Preclinical Studies
Preclinical studies in cancer research involve systematic testing of therapeutic candidates in controlled laboratory settings, including in vitro cell-based assays and in vivo animal models, to assess pharmacokinetics, toxicology, efficacy, and potential mechanisms of action prior to advancing to human clinical trials.[58] These investigations prioritize identifying agents that demonstrate antitumor activity while minimizing toxicity, thereby informing the design of interventional trials.[59] Translational efforts integrate preclinical findings with clinical observations, such as through reverse translation where patient data refines model selection, to enhance the predictive accuracy of outcomes in human disease contexts.[60] In vitro models, traditionally reliant on two-dimensional monolayer cultures of immortalized cancer cell lines, provide initial high-throughput screening for drug sensitivity but often fail to recapitulate tumor microenvironment interactions or heterogeneity observed in patients.[61] Advances include three-dimensional organoid cultures derived from patient tumors, which better mimic tissue architecture, stromal influences, and drug penetration, enabling more physiologically relevant efficacy predictions as demonstrated in colorectal and pancreatic cancer studies since the mid-2010s.[62] In vivo preclinical models encompass subcutaneous or orthotopic xenografts, where human cancer cells are engrafted into immunocompromised mice, allowing evaluation of tumor progression and therapeutic responses in a host environment.[63] Patient-derived xenografts (PDX), established by direct implantation of fresh patient tumor fragments, preserve histopathological, genomic, and epigenetic features of the primary malignancy, with engraftment rates of 20-40% across solid tumors and improved correlation to clinical drug responses compared to cell line xenografts.[64] [65] Genetically engineered mouse models (GEMMs) introduce targeted mutations, such as oncogene activations or tumor suppressor knockouts, to replicate human cancer initiation and progression; the first such models emerged in 1984 with transgenic mice expressing activated oncogenes like Myc or Ras, enabling longitudinal studies of multistep carcinogenesis.[66] These models, refined through Cre-loxP recombination systems in the 1990s, facilitate dissection of genetic drivers and microenvironmental roles but require validation against human tumors due to species-specific differences in immune responses and metabolism.[67] Despite these tools, preclinical cancer research faces a reproducibility crisis, with pharmaceutical attempts revealing low replication rates: Amgen confirmed only 6 of 53 landmark studies (11%) in 2012, attributing failures to selective reporting and experimental variability in academic settings.[68] Bayer similarly replicated 20-25% of preclinical projects in 2011, highlighting issues like insufficient statistical power and unblinded analyses.[6] The Reproducibility Project: Cancer Biology, launched in 2013, designed 193 experiments from high-impact papers but completed only 50 by 2021 due to resource constraints and cooperation barriers, underscoring systemic challenges in validating preclinical claims.[69] Such discrepancies contribute to high attrition, with approximately 95% of oncology candidates failing in clinical phases despite preclinical promise, often because models inadequately capture human tumor heterogeneity, immune dynamics, or adaptive resistance.[70] Efforts to improve translation include humanized mouse models incorporating patient immune components and co-clinical trials pairing GEMM or PDX responses with parallel patient monitoring, as in precision oncology initiatives since 2015, which have refined target selection for therapies like PARP inhibitors in BRCA-mutated cancers.[71] These approaches emphasize rigorous controls, multi-omics validation, and prospective testing to mitigate biases toward positive results prevalent in peer-reviewed literature.[72]Clinical and Interventional Trials
Clinical and interventional trials represent the critical translational bridge from preclinical research to approved cancer therapies, involving human participants to assess safety, efficacy, and optimal dosing of interventions such as drugs, radiation, surgery, or immunotherapies. These trials are structured in phases: Phase I focuses on safety and pharmacokinetics in small cohorts (typically 20-100 patients), Phase II evaluates preliminary efficacy and side effects in larger groups (100-300), Phase III conducts randomized comparisons against standard care in thousands to confirm benefits and risks, and Phase IV monitors long-term effects post-approval. Interventional trials specifically test causal effects of treatments, often using randomized controlled designs to minimize bias, contrasting with observational studies that merely track outcomes. Key historical milestones include the 1942 trial of nitrogen mustard (mechlorethamine), the first chemotherapy agent tested systematically on lymphoma patients, yielding temporary remissions and establishing cytotoxic drugs as a paradigm.[4] The 1970s saw large-scale adjuvant chemotherapy trials for breast cancer, such as the NSABP B-01 study (1972), which demonstrated improved survival with combination regimens like CMF (cyclophosphamide, methotrexate, fluorouracil). Targeted therapy advanced with imatinib's Phase II/III trials for chronic myeloid leukemia (1999-2001), achieving 90% response rates by inhibiting BCR-ABL kinase, marking the shift to molecularly guided interventions. Immunotherapy breakthroughs followed, including ipilimumab's Phase III trial (2010) for melanoma, which extended median survival from 6.4 to 10 months via CTLA-4 blockade, despite immune-related toxicities. Success rates remain low, with only 13.4% of investigational cancer drugs achieving regulatory approval from Phase I onward, compared to higher rates (around 20-25%) for non-oncology indications, reflecting tumor heterogeneity, adaptive resistance, and stringent endpoints like overall survival.[73] From Phase II to approval, advancement occurs in just 7-11% of cases, exacerbated by discrepancies between preclinical models and human physiology, where 95% of candidates fail due to inefficacy or toxicity.[74] [75] Positive Phase III results show partial reproducibility, with modeling estimating replication probabilities below 50% for statistically significant trials, attributable to underpowered studies, endpoint variability, and publication bias favoring positives.[76] Major barriers impede trial conduct and enrollment, including patient-level factors like logistical burdens (travel, frequent visits), financial strain, and distrust in research—cited by up to 30% of eligible patients declining participation.[77] [78] Provider-side issues encompass inadequate trial awareness, staffing shortages, and prolonged regulatory approvals, limiting community site involvement where 80% of patients receive care.[79] Disparities amplify challenges, with underrepresented minorities facing implicit biases and geographic barriers, resulting in trials skewed toward white, urban demographics despite diverse disease burdens.[80] Recent advances (2023-2025) emphasize precision and adaptive designs, such as basket trials matching therapies to biomarkers regardless of tumor site, with FDA approvals for tissue-agnostic agents like larotrectinib (NTRK inhibitors) expanding since 2018.[81] Neoadjuvant immunotherapy trials, like KEYNOTE-522 (ongoing refinements post-2023), have boosted pathologic complete response rates to 60% in triple-negative breast cancer when combined with chemotherapy.[82] Antibody-drug conjugates (ADCs) and bispecific antibodies showed Phase III successes, e.g., sacituzumab govitecan extending survival in metastatic triple-negative breast cancer by 5 months (2023 data).[83] These innovations, while promising, underscore persistent hurdles in scalability and equity, with calls for streamlined regulations to enhance accrual without compromising rigor.[84]Prevention Strategies
Epidemiological Identification of Risk Factors
Epidemiological research employs observational study designs, such as prospective cohort studies and retrospective case-control studies, to identify associations between exposures and cancer incidence by comparing rates in exposed versus unexposed groups, often adjusting for confounders like age and socioeconomic status.[85] These methods have established causality for several risk factors using criteria including dose-response relationships, biological plausibility, and consistency across studies.[86] Landmark examples include the 1950 case-control study by Richard Doll and Austin Bradford Hill, which linked tobacco smoking to lung cancer with an odds ratio of approximately 14 for heavy smokers, later corroborated by the 1954 British Doctors cohort study showing a 24-fold increased risk for lung cancer mortality among persistent smokers.[86] Tobacco remains the leading identifiable cause, attributable to about 20-25% of all cancer deaths globally, with strongest evidence for lung, larynx, and bladder cancers classified as Group 1 carcinogens by the International Agency for Research on Cancer (IARC) based on sufficient human epidemiological data.[87]01438-6/fulltext) Alcohol consumption has been identified as a risk factor through large cohort studies, such as those pooling data from over 100,000 participants, demonstrating dose-dependent increases in risks for cancers of the mouth, pharynx, esophagus, liver, and breast, with even light-to-moderate intake elevating breast cancer risk by 5-10%.[88] IARC classifies ethanol in alcoholic beverages as a Group 1 carcinogen, supported by meta-analyses of epidemiological evidence showing relative risks rising from 1.1 for low intake to over 5 for heavy drinking in upper aerodigestive tract cancers.[87] In 2019, alcohol contributed to 4.1% of global cancer cases, with behavioral risks like this dominating over metabolic factors in attributable burden.01438-6/fulltext) Obesity, measured by body mass index (BMI) ≥30 kg/m², emerged as a risk factor via cohort studies like the Nurses' Health Study and Health Professionals Follow-up Study, linking it to 13 cancer types including colorectal, postmenopausal breast, and endometrial, with hazard ratios of 1.2-2.0 depending on site and adiposity duration.[88] Epidemiological data indicate obesity accounts for 4-8% of cancers in high-income countries, with risks mediated by insulin resistance, inflammation, and sex hormones, though confounding by physical inactivity complicates isolation.01438-6/fulltext)[89] Combined with alcohol, these factors outweighed smoking's role in some female cancers in European cohorts.[90] Infectious agents were pinpointed through serological and cohort studies, such as those establishing human papillomavirus (HPV) types 16 and 18 as causes of cervical cancer (relative risk >100 in persistent infections) and hepatitis B/C viruses for hepatocellular carcinoma (odds ratios 20-50).[87] IARC's evaluations, drawing on global incidence patterns and intervention trials like HPV vaccination reducing precancerous lesions by 90%, confirm these as Group 1 carcinogens responsible for 15-20% of cancers worldwide, predominantly in developing regions.[87] Environmental and occupational exposures, including asbestos (mesothelioma risk elevated 100-fold in exposed workers) and ionizing radiation (linear no-threshold model from atomic bomb survivor cohorts showing excess leukemia at 0.1-1 Gy doses), were identified via occupational registries and atomic/nuclear studies.[91] These findings underscore epidemiology's role in quantifying population-attributable fractions, guiding prevention despite challenges like recall bias in case-control designs and residual confounding in observational data.[88]Public Health Interventions
Public health interventions for cancer prevention involve population-wide strategies, including policy regulations, vaccination drives, and educational campaigns, aimed at curtailing exposure to established carcinogens and infectious agents. These measures target modifiable risk factors identified through epidemiological research, such as tobacco use, viral infections, ultraviolet radiation, and excessive alcohol consumption, with evidence from longitudinal studies demonstrating causal reductions in cancer incidence attributable to decreased exposure prevalence.[92] For instance, interventions like tobacco taxation and restrictions have yielded the largest averted cancer deaths, primarily from lung cancer, underscoring the dose-response relationship between smoking and oncogenesis.[93] Tobacco control policies exemplify successful public health action, with comprehensive implementations—including excise taxes, smoke-free laws, and advertising prohibitions—correlating with sharp declines in smoking rates and subsequent lung cancer mortality. In the United States, such strategies averted approximately 3.9 million lung cancer deaths from 1975 to 2020, reflecting a lag of decades between reduced initiation and peak incidence drops.[94] Globally, modeling indicates that a tobacco-free generation policy for those born after 2006 could prevent over 1.1 million lung cancer deaths by mid-century, based on projections from current prevalence trends and historical policy impacts.[95] These outcomes align with causal evidence from cohort studies tracking policy-exposed cohorts against unexposed ones, confirming that higher policy stringency indices reduce not only lung but also other tobacco-attributable cancers like those of the oral cavity and bladder.[96] Human papillomavirus (HPV) vaccination programs represent a targeted intervention against virus-driven cancers, particularly cervical cancer, with routine immunization of adolescents since 2006 leading to herd immunity effects and plummeting disease burdens. In Sweden, nationwide HPV vaccination with coverage exceeding 80% resulted in an 88% reduction in invasive cervical cancer incidence among women vaccinated before age 17, as evidenced by a registry-linked cohort study.[97][98] Similarly, U.S. data from nearly 3.5 million individuals showed HPV vaccination conferred up to 90% protection against vaccine-targeted strains, translating to fewer precancerous lesions and invasive cases.[99] Single-dose regimens, tested in trials like KEN SHE, achieved 97.5% efficacy against persistent high-risk HPV infections, supporting scalable public programs in resource-limited settings to avert an estimated 40% of cervical cancers through early vaccination.[100] These reductions persist across socioeconomic groups, with stronger effects in fully implemented national schedules.[101] Alcohol policy interventions, such as minimum unit pricing (MUP) and volumetric taxation, address ethanol's role as a Group 1 carcinogen, particularly for hepatocellular carcinoma and upper aerodigestive tract cancers. Scotland's 2018 MUP of £0.50 per unit, combined with taxes, projected a 4.3-4.7% drop in cumulative chronic liver disease incidence over a decade, including alcohol-related liver cancers, via consumption modeling validated against sales data.[102] International Agency for Research on Cancer analyses link stricter policies to dose-dependent risk reductions, with cessation or moderation yielding inverse associations for liver cancer in cohort studies.[103] [104] Ultraviolet radiation awareness campaigns promote behavioral shifts like sunscreen use and shade-seeking to prevent skin cancers, with WHO estimating 80-90% preventability through reduced exposure. Programs such as Australia's SunSmart initiative, launched in 1988, increased sun protection compliance by 20-30% in targeted populations, correlating with stabilized melanoma rates post-peak rises, though attribution requires disentangling from ozone recovery.[105] U.S. efforts like CDC's Sun Sense have educated outdoor workers, yielding modest incidence plateaus in high-risk groups per surveillance data.[106] Overall, these interventions' efficacy hinges on enforcement and cultural uptake, with meta-analyses affirming causal pathways from policy exposure to lowered carcinogen dose and tumor formation.[107] Despite these advances, significant global disparities exist in access to and research on cancer prevention strategies. Low- and middle-income countries (LMICs), despite bearing a disproportionate share of the global cancer burden, receive only an estimated 0.5%–5% of global cancer research funding.[108] Additionally, cancer clinical trials often underrepresent minorities and elderly patients, limiting the generalizability of evidence to diverse populations.[109] In high-resource settings, comprehensive prevention efforts are exemplified by the German Cancer Research Center (DKFZ), which, through its National Cancer Prevention Center established in 2019 in partnership with German Cancer Aid, integrates research, education, training, public outreach, and policy advice to systematically advance cancer prevention across Germany.[110]Lifestyle Modifications and Their Evidence
Lifestyle modifications, including smoking cessation, maintaining a healthy body weight, regular physical activity, balanced diet, and limited alcohol intake, have been associated with reduced cancer incidence and mortality in multiple epidemiological studies. The World Health Organization estimates that between 30% and 50% of cancer cases can currently be prevented by avoiding risk factors and implementing existing evidence-based prevention strategies, including lifestyle changes.[111] A 2020 analysis of combined lifestyle factors indicated that adherence to healthy behaviors—such as not smoking, moderate alcohol consumption, physical activity, healthy diet, and normal body weight—correlates with up to 30-50% lower risk of cancer morbidity and mortality across populations.[112] The World Cancer Research Fund (WCRF) Third Expert Report, based on systematic reviews of over 100 million people, concludes that these factors causally influence cancer risk through mechanisms like reduced inflammation, improved insulin sensitivity, and lower exposure to carcinogens.[113] Smoking cessation substantially mitigates cancer risks, particularly for lung cancer, which accounts for about 85% of smoking-attributable cases. Quitting before age 40 averts approximately 90% of the excess lifetime mortality risk from smoking-related diseases, including cancer, according to a 2024 prospective cohort study of over 1 million adults.[114] Within 10-15 years post-cessation, lung cancer risk halves compared to continued smokers, as evidenced by CDC surveillance data from U.S. cohorts.[115] Even after a lung cancer diagnosis, cessation reduces recurrence and improves survival, with meta-analyses showing 15-50% lower mortality in quitters versus persistent smokers.[116] Physical activity consistently lowers risk for at least 13 cancer types, including breast, colon, and endometrial cancers. A 2023 dose-response meta-analysis of cohort studies found that meeting recommended levels (7.5-15 MET-hours/week) reduces overall cancer incidence by 6-29%, with greater benefits for vigorous activity; for instance, high activity levels correlate with 20% lower endometrial cancer risk in women.[117][118] Mechanisms include enhanced DNA repair, reduced circulating estrogens, and anti-inflammatory effects, supported by prospective data from the NCI. Leisure-time vigorous intermittent activity, even in short bouts (e.g., 4.5 minutes daily median), associates with 31-32% lower incidence of activity-related cancers in UK Biobank participants.[119] Maintaining a healthy body weight is linked to lower risk for 13-15 cancers, comprising about 40% of U.S. diagnoses, per CDC and NCI estimates from large-scale epidemiology. Excess adiposity promotes carcinogenesis via hyperinsulinemia, chronic inflammation, and sex hormone dysregulation; a 2023 review identified elevated risks for breast (postmenopausal), colorectal, and endometrial cancers with BMI ≥30 kg/m².[120][121] Intentional weight loss of >5 kg in overweight individuals reduces overall cancer risk, as shown in longitudinal studies, though evidence for post-diagnosis survival benefits remains observational and confounded by reverse causation.[122] Dietary patterns emphasizing whole grains, vegetables, fruits, and limited processed meats and sugars align with WCRF recommendations, showing probabilistic limited-suggestive evidence for reduced risks of colorectal and breast cancers. The 2025 WCRF report on dietary-lifestyle patterns reinforces that plant-based diets lower overall cancer risk by 10-20% in adherent populations, contrasting with high red meat intake, which increases colorectal cancer odds by 17% per 100g daily.[123][124] No single nutrient or supplement consistently prevents cancer; for example, multivitamins show null effects in RCTs like the Physicians' Health Study II. Alcohol consumption exhibits a dose-response relationship with cancer risk, with no threshold for safety; even low intake (<1 drink/day) elevates risks for breast and colorectal cancers by 5-10%, per systematic reviews.[125] Heavy drinking (>3 drinks/day) markedly increases oral, esophageal, and liver cancer incidences, as quantified in a 2023 meta-analysis across 100+ studies.[126] Reduction or abstinence post-diagnosis improves treatment outcomes, with increased consumption linking to higher all-cancer mortality in cohort data.[127] Combined adherence to these modifications yields synergistic effects; a 2023 cohort study found that following WCRF/AICR guidelines before and after diagnosis associates with 20-40% lower recurrence and mortality across cancers.[128] However, individual responses vary due to genetic factors, underscoring that while lifestyle accounts for 30-50% of modifiable cancer burden, non-modifiable risks persist. Evidence derives primarily from observational cohorts, with causation inferred from consistency, temporality, and biological plausibility, though RCTs for prevention remain ethically challenging.[112]Detection and Early Diagnosis
Screening Technologies
Screening technologies for cancer detection target asymptomatic individuals to identify malignancies at stages amenable to curative intervention, thereby reducing mortality through early treatment. Population-based programs have demonstrated varying degrees of efficacy, with randomized controlled trials (RCTs) and meta-analyses providing the strongest evidence. For instance, combined screening adherence in the United States has saved an estimated 12.2–16.2 million life-years since the introduction of U.S. Preventive Services Task Force (USPSTF) recommendations.[129] However, benefits must be weighed against harms such as false positives, overdiagnosis of indolent lesions, and unnecessary interventions, which can lead to complications without improving survival.[130] Mammography remains the primary screening modality for breast cancer in women aged 40–74, involving low-dose X-ray imaging to detect microcalcifications and masses. Meta-analyses of RCTs indicate a 20–31% reduction in breast cancer mortality for women invited to screening, with greater effects (38–48%) among those who attend.[131] A 2024 systematic review of cohort studies reported a 45% lower risk of breast cancer death among attendees compared to non-attendees, though no impact on all-cause mortality was observed.00181-1/fulltext) The USPSTF recommends biennial screening starting at age 40, but controversies persist regarding overdiagnosis rates of 10–20%, potentially leading to overtreatment of non-progressive tumors.[132] Prostate-specific antigen (PSA) testing measures serum levels of a prostate-derived protein to screen for prostate cancer in men aged 55–69, often prompting biopsy if elevated. The European Randomized Study of Screening for Prostate Cancer (ERSPC) trial showed a 20% reduction in prostate cancer mortality with PSA screening, but at the cost of substantial overdiagnosis—up to 50% of detected cases may represent indolent disease unlikely to cause harm.[133] USPSTF advises shared decision-making due to these trade-offs, as opportunistic testing has not demonstrably reduced overall mortality while increasing biopsies and treatments with risks like incontinence.[134] Emerging refinements, such as PSA density or multiparametric MRI, aim to mitigate false positives but lack widespread validation.[130] For colorectal cancer, colonoscopy enables direct visualization and polypectomy, recommended every 10 years for average-risk adults aged 45–75. Observational data link it to a 69% reduction in incidence and 88% in mortality for screened segments, with RCTs confirming lower colorectal cancer risk at 10 years post-invitation.[135][136] Non-invasive alternatives like fecal immunochemical testing (FIT) detect occult blood, achieving 10–68% mortality reductions in modeling studies, though less effective for proximal lesions.00174-4/fulltext) Harms include perforation (0.1–0.2% risk) and incomplete protection against interval cancers.[137] Low-dose computed tomography (LDCT) scans screen high-risk individuals for lung cancer, targeting current or former heavy smokers aged 50–80 with at least 20 pack-years. The National Lung Screening Trial demonstrated a 20% mortality reduction, prompting USPSTF endorsement for annual LDCT.[138] False-positive rates exceed 20%, necessitating follow-up imaging or biopsies, but net benefits accrue in selected populations.[139] Cervical cancer screening relies on Papanicolaou (Pap) cytology to identify dysplastic cells, supplemented by high-risk human papillomavirus (HPV) DNA testing for women aged 30–65. Co-testing every 5 years or HPV alone reduces incidence by detecting persistent infections causal to nearly all cases; guidelines recommend starting at age 21 with Pap alone every 3 years.[140] HPV vaccination has further diminished prevalence, enhancing screening efficacy.[141] Overdiagnosis is minimal given the precancerous focus, with harms limited to colposcopy referrals.[142]Biomarker and Liquid Biopsy Advances
Biomarkers in cancer research encompass measurable indicators such as circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), exosomes, and proteins that reflect tumor presence, progression, or therapeutic response.[143] Advances have focused on enhancing their sensitivity and specificity through next-generation sequencing (NGS) and multi-omics approaches, enabling non-invasive detection via liquid biopsies—analyses of blood, urine, or other fluids.[144] These methods address limitations of tissue biopsies, such as invasiveness and sampling bias, by capturing tumor heterogeneity in real-time.[145] Key milestones include the FDA approval of Guardant360 CDx in August 2020 as the first comprehensive liquid biopsy for solid tumors, analyzing over 70 genes for mutations and fusions to guide therapy selection.[146] In 2021, FoundationOne Liquid CDx received approval for similar NGS-based profiling across 300+ genes, expanding companion diagnostic uses for targeted therapies in cancers like non-small cell lung cancer.[147] A significant 2024 advancement was the FDA approval of Guardant Health's Shield blood test on July 29 as the first liquid biopsy for primary colorectal cancer screening, detecting 83% of stage II-IV cases and 13% of stage I cases in the ECLIPSE study of 20,000+ participants at average risk.[148] These approvals underscore liquid biopsy's shift toward clinical utility, though early-stage detection remains challenged by low ctDNA shedding rates, often below 0.1% of total cell-free DNA.[143] Recent technical progress integrates epigenomic profiling, such as methylation patterns, with genomic alterations for improved accuracy; for instance, Guardant360 Liquid combines both to monitor minimal residual disease (MRD) post-treatment, detecting recurrence earlier than imaging in trials for lung and breast cancers.[149] Mass spectrometry-based proteomics has emerged for protein biomarkers, identifying novel panels in CTCs and exosomes with higher specificity than ctDNA alone, as shown in 2024 studies correlating plasma proteome changes with therapeutic resistance.[150] CRISPR-associated enzymes have been adapted for rapid, point-of-care detection of ctDNA mutations, achieving sensitivities down to 0.01% variant allele frequency in 2024 assays for liquid biopsy samples.[151] Despite these gains, validation trials emphasize the need for larger cohorts to confirm prognostic value, with false positives from clonal hematopoiesis posing ongoing hurdles.[144] Clinical adoption is growing, particularly for monitoring immunotherapy response via CTC phenotypic markers, but cost and standardization remain barriers to widespread use.[152]Emerging AI and Computational Tools
Artificial intelligence (AI) algorithms, particularly deep learning models, have advanced cancer detection by automating the analysis of medical images, achieving sensitivities and specificities often surpassing human radiologists in controlled settings. For breast cancer screening, convolutional neural networks trained on large datasets of mammograms can detect invasive ductal carcinoma with an area under the curve (AUC) exceeding 0.90, enabling earlier identification than traditional methods alone.[153] In June 2025, the U.S. Food and Drug Administration (FDA) authorized Clairity Breast, the first AI tool specifically designed to predict a woman's five-year breast cancer risk directly from mammogram images, integrating pixel-level features to stratify patients for targeted follow-up.[154] [155] Similarly, AI systems applied to low-dose computed tomography (CT) scans for lung cancer screening identify pulmonary nodules as small as 3-5 mm with detection rates up to 94%, facilitating intervention when tumors are most curable.[156] Computational tools enhance biomarker analysis in liquid biopsies by processing circulating tumor DNA (ctDNA) and other analytes from blood samples. Machine learning models integrate multi-omics data—such as methylation patterns and protein biomarkers—to boost early detection sensitivity for gastrointestinal cancers to over 85% in pilot studies, outperforming single-marker assays through feature selection and pattern recognition.[157] [158] For colorectal cancer, AI-driven analysis of plasma cfDNA mutations combined with radiomics from imaging yields precision classification with AUC values around 0.95, allowing non-invasive monitoring and risk assessment without tissue biopsies.[159] These tools employ algorithms like random forests or neural networks to handle noisy, high-dimensional data, reducing false positives that plague conventional thresholds.[160] Beyond imaging and biopsies, predictive computational models leverage electronic health records (EHRs) and genomic data for population-level risk stratification. Ensemble learning approaches, incorporating variables like age, family history, and polygenic risk scores, forecast cancer onset with hazard ratios indicating 20-30% improved accuracy over standard models like Gail or Tyrer-Cuzick for breast cancer.[161] In 2025, AI frameworks for DNA methylation profiling in peripheral blood detected colorectal cancer precursors with 89% specificity, using supervised classifiers trained on thousands of samples to identify causal epigenetic signatures.[162] Such tools, while promising, require validation across diverse populations to mitigate overfitting and algorithmic biases observed in datasets skewed toward certain demographics.[163] Overall, these emerging applications underscore AI's potential to shift detection paradigms toward proactive, data-driven paradigms, though clinical integration demands rigorous prospective trials to confirm real-world efficacy.[164]Treatment Research
Pharmacological and Targeted Therapies
![Signal transduction pathways in cancer cells][float-right] Pharmacological therapies in cancer treatment primarily encompass cytotoxic chemotherapy agents that interfere with cell division and proliferation, affecting both malignant and normal rapidly dividing cells. Nitrogen mustard, the first chemotherapeutic agent, was introduced in the 1940s following observations of its effects on lymphoid tissue during World War II, establishing the foundation for modern regimens.[4] Advances since then include the development of platinum-based drugs like cisplatin, approved in 1978, which cross-links DNA to induce apoptosis and has demonstrated response rates up to 90% in testicular cancer when combined with etoposide and bleomycin.[165] Recent evidence supports optimized dosing and supportive care to mitigate toxicity, with studies showing that dose-dense chemotherapy improves progression-free survival in breast cancer by 20-30% compared to conventional schedules.[166] Targeted therapies exploit specific molecular alterations in cancer cells, such as oncogenic mutations or overexpressed proteins, to achieve greater selectivity and reduced off-target effects relative to traditional chemotherapy. Tyrosine kinase inhibitors (TKIs), a cornerstone of this approach, block aberrant signaling pathways; for instance, imatinib, approved by the FDA in 2001 for chronic myeloid leukemia (CML), targets the BCR-ABL fusion protein and yields complete cytogenetic responses in over 80% of chronic-phase patients, transforming CML from a fatal disease to a manageable condition with median survival exceeding 10 years.[167] Monoclonal antibodies, another key class, include rituximab, approved in 1997 for non-Hodgkin lymphoma, which binds CD20 to trigger immune-mediated cytotoxicity and improves overall survival by 20-30% in combination with chemotherapy.[168] By 2023, the FDA had approved 79 therapeutic monoclonal antibodies, with at least 48 designated for oncology indications.[168] Emerging targeted modalities include antibody-drug conjugates (ADCs) and small-molecule inhibitors for novel targets. ADCs like datopotamab deruxtecan, under investigation in 2024 trials, deliver payloads directly to tumor cells expressing specific antigens, showing objective response rates of 30-40% in non-small cell lung cancer refractory to prior therapies.[169] PARP inhibitors, such as olaparib approved in 2014 for BRCA-mutated ovarian cancer, exploit synthetic lethality in DNA repair-deficient tumors, extending progression-free survival by 3-7 months in phase III trials.[170] In 2025, FDA approvals included 13 new oncology agents, many targeted, expanding options for rare cancers and head/neck squamous cell carcinoma.[171] Despite these gains, resistance remains a primary challenge, arising through target mutations, pathway reactivation, or tumor heterogeneity. For EGFR inhibitors in lung cancer, secondary T790M mutations confer resistance in 50-60% of cases, necessitating next-generation agents like osimertinib, approved in 2015.[172] Non-genetic mechanisms, including epigenetic adaptations and cancer stem cell persistence, further complicate efficacy, with preclinical models indicating bypass signaling in up to 70% of resistant tumors.[173] Ongoing research emphasizes combination strategies, such as TKIs with chemotherapy, to delay resistance and improve durable responses, as evidenced by hazard ratios for progression reduced by 20-40% in randomized trials.[174] Clinical data from 2001-2018 approvals show that while 35% of targeted drugs extend overall survival, median benefits are modest at 2-3 months, underscoring the need for biomarkers to predict responders.[175]Immunotherapies and Vaccine Development
Immunotherapies harness the patient's immune system to target cancer cells, primarily through strategies that enhance T-cell recognition and activation against tumor antigens. Checkpoint inhibitors, such as anti-PD-1 (e.g., pembrolizumab) and anti-CTLA-4 (e.g., ipilimumab) antibodies, block inhibitory signals that tumors exploit to evade immunity, leading to durable responses in immunogenic cancers. In metastatic melanoma, combination therapy with nivolumab and ipilimumab has achieved a 52% rate of cancer-free survival at 10 years among treated patients, a marked improvement over historical chemotherapy outcomes of less than 10%.[176] For non-small cell lung cancer, PD-1 inhibitors combined with chemotherapy extend median overall survival by approximately 22 months compared to chemotherapy alone in some cohorts, though response rates vary by tumor mutation burden and PD-L1 expression levels, with only 20-40% of patients achieving long-term benefit.[177] These agents, first approved by the FDA in 2011 for melanoma, have revolutionized treatment for subsets of patients but fail in "cold" tumors lacking sufficient neoantigens or immune infiltration, highlighting the need for biomarkers to predict efficacy.[178] Adoptive cell therapies, including chimeric antigen receptor (CAR) T-cell therapies, involve engineering patient T cells to express receptors targeting specific tumor antigens, followed by reinfusion. Approved since 2017 for relapsed B-cell acute lymphoblastic leukemia and certain non-Hodgkin lymphomas, CAR-T products like axicabtagene ciloleucel yield complete remission rates of 40-80% in aggressive lymphomas, with 2-year progression-free survival around 40% in follicular lymphoma.[179][180] Efficacy remains confined largely to hematologic malignancies due to challenges in solid tumors, such as antigen heterogeneity, immunosuppressive microenvironments, and poor T-cell trafficking, resulting in response rates below 20% in trials for cancers like glioblastoma or pancreatic adenocarcinoma as of 2025.[181] Cytokine release syndrome and neurotoxicity affect up to 90% of recipients, necessitating intensive management, yet these therapies demonstrate proof-of-principle for personalized immune redirection.[182] Vaccine development in cancer spans preventive and therapeutic approaches, with preventive vaccines targeting oncogenic viruses showing robust success. The human papillomavirus (HPV) vaccine, introduced in 2006, prevents nearly 100% of persistent infections by high-risk HPV types 16 and 18, reducing cervical precancer incidence by 80-90% in vaccinated cohorts and averting an estimated 90% of HPV-attributable cervical cancers.[183][184] Population-level studies confirm a 87% drop in cervical cancer rates among women vaccinated before age 17, underscoring causal efficacy against viral integration-driven oncogenesis, though uptake remains suboptimal at 60% globally due to access and hesitancy barriers.[98] Therapeutic cancer vaccines aim to elicit de novo immune responses against established tumors, often using tumor-associated antigens, neoantigens, or viral vectors, but have faced high failure rates in phase III trials due to weak immunogenicity, tumor immune escape, and inadequate trial endpoints focused on survival over immune correlates. Sipuleucel-T, an autologous dendritic cell vaccine targeting prostatic acid phosphatase, received FDA approval in April 2010 for metastatic castration-resistant prostate cancer, extending median overall survival by 4.1 months (HR 0.78) without tumor regression, relying instead on immune activation metrics.[185][186] Despite this milestone, subsequent vaccines like those for melanoma (e.g., gvax) or breast cancer (e.g., E75) failed to meet primary survival endpoints in large trials, with recurrence reductions not translating to overall survival gains, attributed to regulatory T-cell suppression and antigen loss variants.[187] Recent advances incorporate mRNA platforms and personalization, as in neoantigen vaccines showing 30-50% immune response rates in phase I trials for melanoma and pancreatic cancer by 2025, with ongoing phase II studies combining them with checkpoint inhibitors to overcome tolerance; however, scalability and cost limit broad adoption, and long-term efficacy data remain pending.[188][189] These efforts underscore the causal role of antigen-specific T cells in tumor control but reveal persistent hurdles in achieving consistent clinical impact across heterogeneous cancers.Novel Modalities Including Gene Editing
Gene editing technologies, such as CRISPR-Cas9, represent a paradigm shift in cancer therapeutics by allowing targeted alterations to genomic sequences in tumor cells or immune effectors, potentially disrupting oncogenic pathways or enhancing antitumor immunity.[190] These approaches address limitations of traditional therapies by enabling precision modifications that spare healthy tissue, with clinical translation accelerating since the first CRISPR-based trials in 2016.[191] As of February 2025, approximately 250 gene-editing trials are underway globally, with over 150 involving CRISPR candidates, including multiple for hematologic and solid tumors.[192] CRISPR-Cas9 has been applied ex vivo to edit patient-derived hematopoietic stem cells or T cells, as in CRISPR Therapeutics' CTX131, an allogeneic therapy targeting solid tumors and lymphomas by knocking out immune checkpoints like PD-1 and inserting tumor-specific receptors; phase 1 trials reported initial safety data in 2025, with efficacy updates anticipated later that year.[193] In a first-in-human trial completed in May 2025, University of Minnesota researchers used CRISPR-Cas9 to edit T cells for advanced gastrointestinal cancers, achieving partial responses in early patients by disrupting TGF-β signaling in tumors.[194] For solid tumors, ongoing trials explore in vivo delivery via lipid nanoparticles or viral vectors to directly edit cancer genomes, though off-target effects and delivery efficiency remain hurdles requiring further validation.[195] Chimeric antigen receptor (CAR) T-cell therapies exemplify gene-edited modalities, where autologous or allogeneic T cells are engineered to express synthetic receptors targeting cancer antigens; seven FDA approvals exist as of 2025, primarily for B-cell malignancies, with complete remissions in up to 80% of refractory cases for products like axicabtagene ciloleucel.[179] Recent advances include "triple-threat" CAR-T designs incorporating multiple antigens and armored cytokines, debuting in phase 1 trials at institutions like the University of Kansas Cancer Center in August 2025 to combat antigen escape in aggressive tumors.[196] In vivo CAR-T generation, bypassing ex vivo manipulation, uses systemic vectors to reprogram circulating T cells directly; preclinical data from 2025 show feasibility in reducing manufacturing costs by over 90% while expanding access beyond specialized centers.[197] Allogeneic "off-the-shelf" CAR-T variants, edited for universal compatibility via HLA and TCR knockouts, entered phase 2 trials in 2025, demonstrating persistence comparable to autologous cells in lymphoma models.[198] Advanced variants like base editing and prime editing offer higher fidelity by enabling single-nucleotide changes without double-strand breaks, minimizing unintended mutations. Base editors have modeled cancer-driving point mutations, such as TP53 variants, in organoids to validate causality and test reversion strategies, with applications in engineering CAR-T cells resistant to tumor microenvironments.[199] Prime editing, introduced in 2019, facilitates insertions, deletions, and transitions with up to 90% efficiency in cell lines; by 2025, it corrected cancer-associated mutations in organoid models of colorectal and breast cancers, paving the way for therapeutic reversion of driver lesions like KRAS G12D.[200] Clinical pipelines incorporate these for precision oncology, though long-term genomic stability data from large cohorts are pending, underscoring the need for rigorous preclinical causal modeling before broad adoption.[201]Modifiable Risk Factors
Tobacco and Environmental Exposures
Tobacco use, particularly cigarette smoking, accounts for approximately one-third of all cancer deaths in the United States, with an estimated 160,848 smoking-attributable cancer deaths annually among adults over age 35 as of 2008 data.[202][203] Globally, tobacco kills over 7 million people yearly, including through carcinogenesis, with smokers facing up to 15-30 times higher risk of lung cancer compared to nonsmokers.[204] In the U.S., cigarette smoking links to 80-90% of lung cancer deaths, alongside elevated risks for cancers of the larynx, oral cavity, esophagus, pancreas, bladder, kidney, cervix, and acute myeloid leukemia.[205] Carcinogens in tobacco smoke, such as polycyclic aromatic hydrocarbons and nitrosamines, induce DNA adducts and mutations, establishing a direct causal pathway via epidemiological cohort studies and animal models.[203] Cessation reduces risk progressively; quitting for 10-15 years lowers lung cancer mortality by 30-50% relative to continued smokers.[203] Environmental exposures to known carcinogens contribute smaller but significant fractions of cancer incidence, often synergizing with tobacco. Radon, a naturally occurring radioactive gas classified as a Group 1 carcinogen by the International Agency for Research on Cancer (IARC), attributes 3-14% of lung cancer cases worldwide, with population-attributable fractions reaching 12-29% in high-exposure regions based on residential measurements and miner cohorts.[206] Asbestos fibers, another IARC Group 1 agent, cause nearly all mesotheliomas, with lifetime risk scaling with cumulative exposure; occupational cohorts show mesothelioma incidence rising 0.02% per 1,000 fibers per gram of lung tissue, while low-level residential exposure elevates odds by 2.5-fold.[207][208] Ambient fine particulate matter (PM2.5) from air pollution, deemed carcinogenic by IARC, associates with 8-11% increased lung cancer risk per 10 μg/m³ increment, driving 4.2 million premature deaths yearly including via non-lung cancers like those of the digestive tract.[209][210] Other Group 1 environmental carcinogens, including benzene and arsenic in water or air, elevate leukemia and skin/lung cancer risks through genotoxic mechanisms confirmed in prospective studies.[211] Mitigation through exposure reduction yields measurable benefits; U.S. radon testing and ventilation programs have averted thousands of lung cancers, while asbestos bans since the 1980s correlate with declining mesothelioma rates in regulated cohorts.[212] Combined tobacco-environmental effects amplify risks multiplicatively, as seen in smokers with radon or asbestos exposure facing 10-25 times higher lung cancer odds than unexposed nonsmokers.[207] Public health interventions prioritizing these modifiable factors, supported by longitudinal data from agencies like the CDC and WHO, underscore causal realism over correlative associations.[205][210]Dietary Patterns and Nutritional Evidence
Large-scale meta-analyses of cohort studies indicate that adherence to healthy dietary patterns, characterized by high consumption of fruits, vegetables, whole grains, and legumes, correlates with reduced risk of multiple cancers, including colorectal and breast cancers.[213][214] Conversely, Western-style patterns rich in red and processed meats, refined grains, and sugars show positive associations with increased cancer incidence, particularly for gastrointestinal malignancies.[215] These findings derive primarily from observational epidemiology, where confounding factors such as lifestyle and socioeconomic status complicate causal inference, though consistent patterns across diverse populations strengthen the evidence.[216] The Mediterranean dietary pattern, emphasizing olive oil, fish, nuts, and moderate alcohol alongside plant foods, demonstrates inverse associations with overall cancer risk and specific sites like breast and colorectal in prospective cohorts.[217] A 2024 analysis of older adults found higher adherence linked to lower cancer incidence, though effects on mortality were less clear, highlighting potential benefits in prevention rather than progression.[218] Plant-based diets, including vegetarian variants, exhibit 10-12% reductions in overall cancer risk in meta-analyses of prospective studies, with stronger effects for digestive cancers due to elevated fiber and phytochemical intake.[219][220] In contrast, unhealthy plant-based patterns incorporating refined carbs and sugars may elevate digestive cancer risks.[221] Consumption of red and processed meats specifically elevates colorectal cancer risk, with the International Agency for Research on Cancer classifying processed meat as carcinogenic (Group 1) based on sufficient evidence from mechanistic and epidemiological data; each 50 grams daily increases relative risk by about 18%.[222][223] Red meat falls in Group 2A (probable carcinogen), supported by cohort studies showing dose-dependent associations.[224] Randomized trials of nutritional supplements for cancer prevention largely fail to demonstrate benefits and occasionally reveal harms. Antioxidant supplements show no overall preventive effect in meta-analyses of 22 trials, with relative risk near 1.0.[225] The Vitamin D and Omega-3 Trial (VITAL) reported no reduction in invasive cancer incidence from vitamin D supplementation.[226] Similarly, the Selenium and Vitamin E Cancer Prevention Trial (SELECT) found no prostate cancer prevention from these agents, and broader reviews confirm little to no benefit from vitamins and minerals against cancer onset.[227][228] These results underscore that isolated nutrients do not replicate the protective effects observed in whole-food dietary patterns, likely due to synergistic interactions absent in supplements.Obesity, Physical Activity, and Metabolic Influences
Obesity, defined by body mass index (BMI) ≥30 kg/m², is associated with increased risk for at least 13 cancer types, including endometrial, esophageal adenocarcinoma, gastric cardia, liver, kidney, multiple myeloma, meningioma, colorectal, gallbladder, postmenopausal breast, ovarian, pancreatic, and thyroid cancers.[229] A systematic review of cohort studies indicates that for every 5 kg/m² increase in BMI, cancer mortality rises by approximately 10%.[229] Globally, high BMI accounted for 3.6% of new adult cancer cases in 2012, totaling about 481,000 cases, with higher fractions in women (4.1%) than men (3.1%).[230] More recent estimates from 2022 suggest obesity-attributable cancer mortality at 5.69 per 100,000 people annually, with over 462,000 deaths worldwide.[231] Epidemiological evidence links higher BMI to elevated colorectal cancer risk, with overweight and obesity conferring 18% and 32% greater odds, respectively, compared to normal weight.[232] In younger adults, obesity raises early-onset colorectal cancer risk by up to 88%.[233] Intentional weight loss has shown potential to reduce overall cancer incidence in 71.4% of examined studies, though evidence remains observational and requires causal confirmation.[234] Physical activity exerts a protective effect against several cancers, with strong evidence for reduced risk of breast, colon, endometrium, bladder, stomach, and esophageal adenocarcinoma.[235] Observational studies consistently demonstrate that higher activity levels lower cancer risk by 10-20%, potentially through modulation of insulin sensitivity, inflammation, and sex hormones.[236][118] Systematic reviews support these associations across tumor types, with benefits graded as strong or highly suggestive for multiple outcomes.[237] Metabolic influences, including metabolic syndrome (MetS) and type 2 diabetes, independently elevate cancer risk beyond obesity alone. MetS, characterized by central obesity, hypertension, dyslipidemia, and hyperglycemia, is linked to higher incidence of colorectal, breast, and endometrial cancers.[238] Type 2 diabetes associates with increased risk for liver, pancreas, endometrium, colorectal, breast, and bladder cancers, with epidemiological data showing site-specific hazard ratios up to 2-3 fold.[239] A pooled analysis of cohorts found a metabolic risk score (encompassing BMI, blood pressure, and plasma markers) predicting higher overall cancer incidence, underscoring shared pathways like hyperinsulinemia and chronic inflammation.[240] These factors often cluster, amplifying risk, as evidenced by stronger associations in new-onset MetS cases.[241]Funding and Resource Allocation
Major Funding Sources and Trends
The National Cancer Institute (NCI), established under the National Institutes of Health (NIH), serves as the largest single source of public funding for cancer research globally, with its fiscal year 2025 appropriation reaching $7.22 billion, primarily allocated to extramural grants supporting university and hospital-based studies.[242] This funding has historically trended upward nominally from $3.31 billion in 2000 to over $6 billion by the early 2020s, driven by legislative initiatives like the Cancer Moonshot, though real-term growth has stagnated amid inflation and competing priorities, with NCI's share of the overall NIH budget declining from 18% in the 1990s to around 12% recently.[243] Internationally, government agencies such as Cancer Research UK (annual budget exceeding £500 million) and the European Commission's Horizon Europe program contribute significantly, but U.S. federal funding accounts for over 40% of worldwide cancer research investment, exacerbating disparities where low- and middle-income countries receive less than 5% of total global allocations.[244] Pharmaceutical industry investment represents the fastest-growing funding stream, with oncology R&D comprising a disproportionate share of biopharma pipelines due to high potential returns from targeted therapies and biologics. In 2024, global venture funding for cancer initiatives totaled $10.3 billion across 169 deals, up from prior years, fueled by antibody-drug conjugates and cell/gene therapies projected to drive oncology spending from $252 billion in treatment costs to $441 billion by 2029.[245] [246] Leading firms like Merck & Co. topped R&D spenders with billions directed toward immuno-oncology, reflecting a trend where industry outpaces public sources in late-stage development but focuses on commercially viable areas, often sidelining basic research or rare cancers with lower profitability.[247] Philanthropic organizations provide targeted, flexible funding complementary to public and industry sources, emphasizing early-stage and prevention-oriented work. The American Cancer Society (ACS), the largest non-governmental funder, invested $145 million in research grants in 2022 alone, contributing to breakthroughs since 1946 through peer-reviewed awards prioritizing innovative hypotheses over institutional prestige.[248] Entities like the Cancer Research Institute and World Cancer Research Fund International have sustained annual grants in the tens of millions, with recent calls for 2025/2026 focusing on diet, immunotherapy, and global inequities, though their scale remains dwarfed by governmental and corporate inputs.[249] [250] Overall trends indicate nominal increases in total funding but a slowdown in public sector growth, with U.S. NCI facing proposed 2026 cuts to $4.53 billion amid broader federal austerity, while private investments accelerate in precision medicine amid declining global research activity since 2016.[251] [252] This shift heightens reliance on industry, which prioritizes high-revenue modalities, and reveals persistent geographic skews, as high-income nations capture 90% of funds despite bearing only 55% of the global cancer burden.[253] Such patterns underscore causal drivers like economic incentives favoring late-stage trials over foundational science, potentially hindering long-term progress in underfunded areas like etiology and prevention.[244]Allocation Disparities by Cancer Type
The National Cancer Institute (NCI), the primary federal funder of cancer research in the United States, allocates extramural and intramural resources across cancer types based on peer-reviewed grants, with fiscal year 2023 estimates indicating breast cancer received $542 million, lung cancer $435 million, prostate cancer $258.6 million, pancreatic cancer $246 million, and brain and central nervous system cancers $236.1 million out of a total NCI budget of $7.22 billion.[254] Updated fiscal year 2024 projections show similar distributions, with breast cancer at $545.1 million and colorectal cancer at $208.4 million, reflecting priorities in prevalence-driven and advocacy-supported areas.[254] These allocations encompass basic, translational, and clinical research but exclude cross-cutting programs like clinical trials, which totaled $806.6 million in fiscal year 2023.[254] Disparities arise when funding deviates from objective disease burden metrics such as incidence, mortality, or potential years of life lost (PYLL), with high-mortality cancers like pancreatic (5-year survival rate of approximately 12%) and lung often underfunded relative to their lethality compared to breast cancer (5-year survival rate of 91%).[255][256] For example, a 2012 analysis of NCI data found breast and prostate cancers overfunded by factors exceeding their proportional mortality contributions, while pancreatic and liver cancers were underfunded by similar margins, a pattern persisting in later reviews.[255][257] Nonprofit contributions exacerbate this, as breast cancer garners substantial private support uncorrelated with its relative death toll (about 42,000 U.S. deaths annually versus 125,000 for lung cancer), whereas pancreatic (50,000 deaths) and esophageal cancers receive disproportionately less.[258][259]| Cancer Type | NCI Funding (FY 2023, $M) | Approx. U.S. Annual Deaths (2024 est.) | Funding-to-Mortality Ratio Insight |
|---|---|---|---|
| Breast | 542 | 42,000 | Overfunded relative to deaths; strong advocacy influence.[255] |
| Lung | 435 | 125,000 | Underfunded given highest mortality; tobacco links may reduce priority.[256][259] |
| Pancreatic | 246 | 50,000 | Underfunded despite low survival; limited advocacy.[257][259] |
| Brain/CNS | 236.1 | 19,000 | Moderate funding but high PYLL due to younger onset; overfunding in some metrics.[254][260] |
