Iodoform
View on Wikipedia
 | |||
| |||
 | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Triiodomethane | |||
| Other names | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 1697010 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.795 | ||
| EC Number |
| ||
| KEGG | |||
| MeSH | iodoform | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CHI3 | |||
| Molar mass | 393.732 g·mol−1 | ||
| Appearance | Pale, light yellow, opaque crystals | ||
| Odor | Saffron-like[3] | ||
| Density | 4.008 g/cm3[3] | ||
| Melting point | 119 °C (246 °F; 392 K)[3] | ||
| Boiling point | 218 °C (424 °F; 491 K)[3] | ||
| 100 mg/L[3] | |||
| Solubility in diethyl ether | 136 g/L | ||
| Solubility in acetone | 120 g/L | ||
| Solubility in ethanol | 78 g/L | ||
| log P | 3.118 | ||
Henry's law
constant (kH) |
3.4 μmol·Pa−1·kg−1 | ||
| −117.1·10−6 cm3/mol | |||
| Structure | |||
| Hexagonal | |||
| Tetrahedral at C | |||
| Thermochemistry | |||
Heat capacity (C)
|
157.5 J/(K·mol) | ||
Std enthalpy of
formation (ΔfH⦵298) |
180.1 – 182.1 kJ/mol | ||
Std enthalpy of
combustion (ΔcH⦵298) |
−716.9 – −718.1 kJ/mol | ||
| Pharmacology | |||
| D09AA13 (WHO) | |||
| Hazards | |||
| GHS labelling: | |||

| |||
| Warning | |||
| H315, H319, H335 | |||
| P261, P280, P305+P351+P338 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 204 °C (399 °F; 477 K) | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
|||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
none[4] | ||
REL (Recommended)
|
0.6 ppm (10 mg/m3)[4] | ||
IDLH (Immediate danger)
|
N.D.[4] | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||

Iodoform (also known as triiodomethane) is the organoiodine compound with the chemical formula CHI3. It is a pale yellow, crystalline, volatile substance, with a penetrating and distinctive odor (in older chemistry texts, the smell is sometimes referred to as that of hospitals, where the compound is still commonly used) and, analogous to chloroform, sweetish taste. It is occasionally used as a disinfectant.
Naming
[edit]The name iodoform originates with the "formyle radical," an archaic term for the HC moiety,[citation needed] and is retained for historical consistency. A full, modern name is triiodomethane. Another possible name is "carbon hydride triiodide". The "hydride" in the latter is sometimes omitted,[2] though the IUPAC recommends against doing so, as "carbon triiodide" could also mean C2I6 (hexaiodoethane, a highly unstable compound).
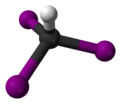
Structure
[edit]The molecule adopts a tetrahedral geometry with C3v symmetry.
Synthesis and reactions
[edit]The synthesis of iodoform was first described by Georges-Simon Serullas in 1822, by reactions of iodine vapour with steam over red-hot coals, and also by reaction of potassium with ethanolic iodine in the presence of water;[6] and at much the same time independently by John Thomas Cooper.[7] It is synthesized in the haloform reaction by the reaction of iodine and sodium hydroxide with any one of these four kinds of organic compounds: a methyl ketone (CH3COR), acetaldehyde (CH3CHO), ethanol (CH3CH2OH), and certain secondary alcohols (CH3CHROH, where R is an alkyl or aryl group).
The reaction of iodine and base with methyl ketones is so reliable that the iodoform test (the appearance of a yellow precipitate) is used to probe the presence of a methyl ketone. This is also the case when testing for specific secondary alcohols containing at least one methyl group in alpha-position.
Some reagents (e.g. hydrogen iodide) convert iodoform to diiodomethane. Conversion to carbon dioxide is also possible.[8] Iodoform reacts with aqueous silver nitrate to produce carbon monoxide. When treated with powdered elemental silver the iodoform is reduced, producing acetylene. Upon heating iodoform decomposes to produce diatomic iodine, hydrogen iodide gas, and carbon.
Natural occurrence
[edit]The angel's bonnet mushroom contains iodoform, and shows its characteristic odor.
Applications
[edit]The compound finds small-scale use as a disinfectant.[5][9] Around the beginning of the 20th century, it was used in medicine as a healing and antiseptic dressing for wounds and sores and, although this use is now largely superseded by superior antiseptics, it is still used in otolaryngology in the form of bismuth subnitrate iodoform paraffin paste (BIPP) as an antiseptic packing for cavities.[10] It is the active ingredient in many ear powders for dogs and cats, along with zinc oxide and propionic acid, which are used to prevent infection and facilitate removal of ear hair.[citation needed]
See also
[edit]References
[edit]- ^ "Front Matter". Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 661. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
The retained names 'bromoform' for HCBr3, 'chloroform' for HCCl3, and 'iodoform' for HCI3 are acceptable in general nomenclature. Preferred IUPAC names are substitutive names.
- ^ a b "Iodoform".
- ^ a b c d e f g Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0343". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b Merck Index, 12 Edition, 5054
- ^ Surellas, Georges-Simon (1822), Notes sur l'Hydriodate de potasse et l'Acide hydriodique. -- Hydriodure de carbone; moyen d'obtenir, à l'instant, ce composé triple [Notes on the hydroiodide of potassium and on hydroiodic acid -- hydroiodide of carbon; means of obtaining instantly this compound of three elements] (in French), Metz, France: Antoine, pp. 17–20, 28–29
- ^ James, Frank A. J. L. (2004). "Cooper, John Thomas". Oxford Dictionary of National Biography (online ed.). Oxford University Press. doi:10.1093/ref:odnb/39361. Retrieved 26 January 2012. (Subscription, Wikipedia Library access or UK public library membership required.)
- ^ Shreeve, W. W.; Leaver, F.; Siegel, I. (1952). "A Method for the Specific Conversion of Iodoform to Carbon Dioxide". J. Am. Chem. Soc. 74 (9): 2404. Bibcode:1952JAChS..74.2404S. doi:10.1021/ja01129a067.
- ^ Lyday, Phyllis A. (2005), "Iodine and Iodine Compounds", Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, pp. 1–13, doi:10.1002/14356007.a14_381.pub2, ISBN 9783527306732
- ^ Randhawa, G. K.; Graham, R.; Matharu, K. S. (2019). "Bismuth Iodoform Paraffin Paste: History and uses". British Journal of Oral and Maxillofacial Surgery. 57 (10): E53 – E54. doi:10.1016/j.bjoms.2019.10.153.
External links
[edit]- NIOSH Pocket Guide to Chemical Hazards. "#0343". National Institute for Occupational Safety and Health (NIOSH).
- Preparation
- . Encyclopædia Britannica. Vol. 14 (11th ed.). 1911. p. 726.
Iodoform
View on GrokipediaNomenclature and structure
Nomenclature
Iodoform is the retained trivial name for the organoiodine compound with the molecular formula CHI3.[4] The term "iodoform" originates from the prefix "iodo-" denoting iodine combined with "-form," derived from "formyl," referring to the formyl group (HCO–), reflecting its historical association with formyl derivatives.[5] This naming convention parallels the haloform series, which includes chloroform (retained name for trichloromethane, CHCl3), bromoform (retained name for tribromomethane, CHBr3), and iodoform as the trihalogenated methane analogs where chlorine, bromine, or iodine replaces hydrogen atoms.[4] The preferred IUPAC name for CHI3 is triiodomethane, emphasizing the systematic substitution of three iodine atoms on a methane backbone.[4] Other synonymous names include carbon triiodide and methyl triiodide, which highlight the carbon-iodine composition or the methyl group's implied structure.[1] These alternative designations are used in various chemical contexts but do not supersede the retained or preferred IUPAC nomenclature for formal identification.[6]Molecular structure
Iodoform has the chemical formula .[1] The molecule features a central carbon atom covalently bonded to one hydrogen atom and three iodine atoms via single bonds. In its Lewis structure, the carbon atom serves as the central atom with four sigma bonds and no lone pairs, while each iodine atom possesses three lone pairs of electrons to satisfy the octet rule.[1][7] The three-dimensional arrangement adopts a tetrahedral molecular geometry with point group symmetry, where the central carbon is at the tetrahedron's center, the hydrogen occupies one vertex, and the three iodine atoms occupy the remaining vertices.[1][8] The C–I bond lengths are approximately 213 pm, and the H–C–I bond angle is about 109.5°, consistent with the idealized tetrahedral configuration.[9]Properties
Physical properties
Iodoform is a pale yellow to bright yellow crystalline solid or powder with a pungent, disagreeable odor often described as saffron-like.[1][10][11] The compound exhibits noticeable volatility at room temperature due to its vapor pressure of approximately 0.04 mmHg, resulting in a detectable vapor.[1][12] Key physical properties of iodoform under standard conditions are summarized below:| Property | Value |
|---|---|
| Molecular weight | 393.73 g/mol |
| Melting point | 119–122 °C |
| Boiling point | 218 °C (sublimes) |
| Density | 4.008 g/cm³ at 25 °C |
| Solubility in water | 0.01 g/100 mL at room temperature |
| Solubility in organic solvents | Soluble in ethanol (7.8 g/100 mL), diethyl ether (13.6 g/100 mL), and chloroform |