Recent from talks
Nothing was collected or created yet.
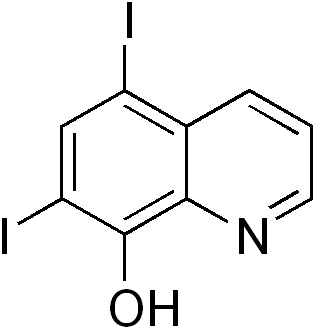
Diiodohydroxyquinoline
View on Wikipedia | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
5,7-Diiodoquinolin-8-ol | |
| Other names
Diquinol, iodoxin, diiodoquin, amebaquin
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.362 |
| KEGG | |
| MeSH | Iodoquinol |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H5I2NO | |
| Molar mass | 396.951 |
| Pharmacology | |
| G01AC01 (WHO) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The quinoline derivative diiodohydroxyquinoline (INN), or iodoquinol (USAN), brand name Diodoquin, can be used in the treatment of amoebiasis.[1]
It is poorly absorbed from the gastrointestinal tract and is used as a luminal amebicide. It acts by chelation of ferrous ions essential for metabolism.[2]
It was discovered by Adco Co. and introduced as diiodohydroxyquinoline.[3]
Susceptibility of Dientamoeba fragilis has been measured.[4]
Iodoquinol is an amebicide used against Entamoeba histolytica, and it is active against both cyst and trophozoites that are localized in the lumen of the intestine. It is considered the drug of choice for treating asymptomatic or moderate forms of amebiasis. The full mechanism of action is unknown. Iodoquinol is used for diseases caused by moderate intestinal amebiasis.
Diiodohydroxyquinoline enhances zinc absorption in the zinc deficiency disorder acrodermatitis enteropathica, probably because it acts as a zinc ionophore.[5]
See also
[edit]References
[edit]- ^ Ghaskadbi S, Vaidya VG (March 1989). "In vivo antimutagenic effect of ascorbic acid against mutagenicity of the common antiamebic drug diiodohydroxyquinoline". Mutat. Res. 222 (3): 219–22. doi:10.1016/0165-1218(89)90137-7. PMID 2493578.
- ^ Nagata, Noriyuki; Marriott, Deborah; Harkness, John; Ellis, John T.; Stark, Damien (2012). "Current treatment options for Dientamoeba fragilis infections". International Journal for Parasitology: Drugs and Drug Resistance. 2: 204–215. doi:10.1016/j.ijpddr.2012.08.002. ISSN 2211-3207. PMC 3862407. PMID 24533282.
- ^ Publishing, William Andrew (2013-01-15). Pharmaceutical Manufacturing Encyclopedia (3rd ed.). Elsevier Science. p. 1312. ISBN 9780080947266.
- ^ Chan FT, Guan MX, Mackenzie AM, Diaz-Mitoma F (May 1994). "Susceptibility testing of Dientamoeba fragilis ATCC 30948 with iodoquinol, paromomycin, tetracycline, and metronidazole". Antimicrob. Agents Chemother. 38 (5): 1157–60. doi:10.1128/aac.38.5.1157. PMC 188168. PMID 8067755.
- ^ Aggett, P.J.; Delves, H.T.; Harries, J.T.; Bangham, A.D. (March 1979). "The possible role of Diodoquin as a zinc ionophore in the treatment of acrodermatitis enteropathica". Biochemical and Biophysical Research Communications. 87 (2): 513–517. doi:10.1016/0006-291X(79)91825-4. PMID 375935.
Diiodohydroxyquinoline
View on GrokipediaChemistry and Properties
Chemical Structure and Nomenclature
Diiodohydroxyquinoline, also known as iodoquinol, is a halogenated derivative of 8-hydroxyquinoline characterized by the molecular formula C₉H₅I₂NO.[2] Its IUPAC name is 5,7-diiodoquinolin-8-ol.[5] The compound is recognized under the United States Adopted Name (USAN) iodoquinol and the International Nonproprietary Name (INN) diiodohydroxyquinoline.[6] The core structure is based on the quinoline ring system, a heterocyclic aromatic compound consisting of a benzene ring fused to a pyridine ring, with nitrogen at position 1. In diiodohydroxyquinoline, a hydroxy group (-OH) is attached at the 8-position adjacent to the nitrogen, while iodine atoms are substituted at the 5- and 7-positions on the benzene portion of the quinoline scaffold. This configuration can be textually represented as follows:- Quinoline numbering: Nitrogen at 1, fusion between 4a and 8a.
- Substituents: I at 5, I at 7, OH at 8.
Physical and Chemical Properties
Diiodohydroxyquinoline appears as a light yellow to light brown powder or fine crystals.[2] The compound contains approximately 63.9% iodine by weight, based on its molecular formula C₉H₅I₂NO with a molecular weight of 396.95 g/mol.[2] It is practically insoluble in water but sparingly soluble in organic solvents such as ethanol, acetone, and ether.[8] Diiodohydroxyquinoline has a melting point of 200–215°C, accompanied by extensive decomposition.[2] The substance is chemically stable under normal conditions of temperature and pressure but is sensitive to light and heat, requiring storage in a cool, dry place protected from sunlight to maintain integrity.[8] It exhibits weakly acidic properties due to the phenolic hydroxy group, with a pKₐ around 8, and demonstrates reactivity in forming chelates with metal ions, a trait shared with other 8-hydroxyquinoline derivatives.[9][10]Medical Uses
Indications and Efficacy
Diiodohydroxyquinoline, commonly known as iodoquinol, is primarily indicated for the treatment of intestinal amebiasis caused by Entamoeba histolytica, including cases in asymptomatic carriers where the goal is to eradicate luminal cysts and trophozoites to prevent transmission and potential progression to invasive disease.[11][12] It acts locally in the intestinal lumen due to its poor systemic absorption, making it suitable for targeting non-invasive forms of the infection without addressing extraintestinal manifestations.[13] Clinical studies have demonstrated its efficacy for luminal amebiasis, particularly in moderate or asymptomatic cases, though its luminal action limits efficacy against systemic or tissue-invasive disease.[14] For invasive amebiasis, iodoquinol is often used in combination with tissue amebicides like metronidazole to achieve comprehensive eradication, with one randomized trial reporting parasitologic cure rates of 97% for the combination versus 87% for metronidazole alone in intestinal amebiasis.[15] Compared to alternatives, iodoquinol is more effective than metronidazole for eliminating luminal forms, as metronidazole primarily targets trophozoites but leaves cysts intact, potentially leading to relapse.[12] Secondary indications include the treatment of infections caused by Balantidium coli, where iodoquinol serves as an alternative agent; Dientamoeba fragilis; and Blastocystis hominis.[16][11][17] It has been used in combination therapies for Giardia lamblia infections, though it is not first-line for giardiasis.[18]Dosage and Administration
Diiodohydroxyquinoline, also known as iodoquinol, is administered exclusively via the oral route in tablet form to target intestinal infections, with no parenteral formulations available.[17][19] For adults, the standard dosage is 650 mg taken orally three times daily after meals for 20 days, not exceeding 2 g per day total.[17][16][19] In pediatric patients, dosing is weight-based at 30–40 mg/kg/day divided into three oral doses after meals for 20 days, with a maximum of 2 g/day and 650 mg per individual dose.[17][16][11] To minimize gastrointestinal upset, tablets should be taken with meals or milk, and the full prescribed course must be completed to ensure efficacy and reduce the risk of treatment failure.[20][21] Post-treatment monitoring, such as stool examinations, is recommended to verify parasite eradication, particularly in cases of amebiasis.Pharmacology
Mechanism of Action
Diiodohydroxyquinoline, also known as iodoquinol, has a mechanism of action that is not fully understood, but is believed to primarily involve chelation of essential metal ions, such as ferrous iron, which are critical for protozoal metabolism.[11] This chelation disrupts enzymatic functions in parasites like Entamoeba histolytica.[22] By binding these ions, the drug impairs the activity of metal-dependent enzymes, leading to halted parasite growth and replication.[18] This action targets both cyst and trophozoite stages of intestinal protozoa, enhancing its efficacy against luminal infections without significant systemic effects on the host.[22] The drug's specificity arises from its localized activity in the intestinal lumen, minimizing broad toxicity while effectively concentrating its effects on parasites residing there, such as Entamoeba histolytica.[11] Diiodohydroxyquinoline also exhibits secondary antibacterial properties through similar metal-binding mechanisms, though these are less potent compared to its antiparasitic activity.[1] This can provide adjunctive inhibition of certain bacterial growth in the gut, but the primary therapeutic focus remains on protozoal targets.[11] The structural hydroxyquinoline core facilitates this chelation, as detailed in the chemical structure section.[13]Pharmacokinetics
Diiodohydroxyquinoline, also known as iodoquinol, exhibits minimal systemic absorption following oral administration, with less than 10% of the dose entering the bloodstream and the majority remaining in the gastrointestinal tract to exert its luminal effects.[23] This poor absorption profile is attributed to its physicochemical properties, resulting in negligible plasma concentrations in humans. Due to the limited absorption, distribution of diiodohydroxyquinoline is primarily confined to the intestines, with no significant penetration into systemic tissues or organs.[19] Animal studies indicate some distribution into tissues for the absorbed fraction, but human data show overall confinement to the GI lumen, supporting its use as a topical intestinal agent.[25] The small absorbed portion undergoes hepatic metabolism, primarily through conjugation with glucuronic acid to form glucuronide conjugates, and to a lesser extent with sulfate.[25] This biotransformation facilitates the elimination of the systemic fraction. Excretion occurs predominantly via the fecal route, with over 90% of the administered dose eliminated unchanged in the feces due to minimal absorption.[23] The absorbed fraction is excreted mainly as metabolites, with approximately 3-5% recovered in the urine.[26] The elimination half-life of the absorbed portion is approximately 11-14 hours, allowing for thrice-daily dosing in clinical practice.[23]Safety and Side Effects
Adverse Effects
Diiodohydroxyquinoline, also known as iodoquinol, is associated with several adverse effects, primarily gastrointestinal disturbances that occur in a notable proportion of users. Common side effects include nausea, vomiting, abdominal cramps, diarrhea, and rectal itching (pruritus ani), based on clinical reports.[27] These symptoms are typically mild to moderate and resolve upon discontinuation of the drug.[20] Hypersensitivity reactions manifest as skin rash, generalized pruritus, or urticaria, occurring infrequently but warranting immediate medical attention if severe.[11] Due to its high iodine content, the drug can exacerbate iodine-related conditions in susceptible individuals, such as acneiform eruptions or goiter (thyroid enlargement).[28] It may also interfere with thyroid function tests for up to six months after treatment, potentially leading to transient thyroid dysfunction.[29] Rare but serious adverse effects include optic neuritis and subacute neurotoxicity, particularly with prolonged or high-dose use. Optic neuritis has been documented in case reports, presenting with blurred vision, eye pain, or decreased vision.[30] Animal studies in rats have demonstrated subacute neurotoxic effects, including motor dysfunction and behavioral alterations, with greater severity in younger animals and females, highlighting potential risks for vulnerable populations.[31] These neurological effects are uncommon in standard therapeutic regimens but underscore the need for monitoring in at-risk groups, as detailed in contraindications. Overall, most adverse effects are reversible following cessation of therapy.[19]Contraindications and Precautions
Diiodohydroxyquinoline, also known as iodoquinol, is contraindicated in patients with known hypersensitivity to iodine, 8-hydroxyquinolines, or the drug itself, as severe allergic reactions may occur.[25] It is also absolutely contraindicated in individuals with hepatic disease, due to the risk of exacerbated liver damage.[19] Additionally, pre-existing optic neuropathy represents an absolute contraindication, given the potential for worsening neuronal damage with use.[19] Relative precautions are advised for patients with thyroid disorders, as the drug's high iodine content can interfere with thyroid function tests for up to 6 months after discontinuation.[25] Use with caution in those with renal insufficiency, where side effect risks may increase, though it is not an absolute contraindication.[32] Pregnancy is classified as category C, with use recommended only if the potential benefit justifies the risk to the fetus, primarily due to iodine-related concerns such as potential thyroid effects on the developing infant.[33] In pediatric patients, dosing should not exceed recommended limits, with heightened caution for long-term or high-dose therapy to avoid increased toxicity risks.[25] Drug interactions include enhanced iodine effects when combined with other iodine-containing medications, potentially leading to iodism or thyroid suppression.[19] Monitoring recommendations include baseline thyroid function tests prior to initiation and periodic assessments during therapy, especially in patients with thyroid conditions.[25] For long-term use, regular eye examinations are advised to detect early signs of optic neuritis or atrophy.[19] In cases of overdose, management is supportive, focusing on symptom relief such as gastrointestinal decontamination if recent ingestion occurred, as no specific antidote exists; monitor for signs of iodism including rash, fever, and neurological symptoms.[25]History and Regulation
Development and Discovery
Diiodohydroxyquinoline, also known as iodoquinol, was developed in the 1930s as a halogenated derivative of 8-hydroxyquinoline to enhance anti-amoebic activity against intestinal infections such as those caused by Entamoeba histolytica.[34] This compound emerged from efforts to improve upon earlier quinoline-based agents like yatren (sodium 5,7-diiodo-8-quinolinol-5-sulfonate), which suffered from poor tolerability and gastrointestinal side effects; the removal of the sulfonate group in diiodohydroxyquinoline resulted in better absorption and reduced toxicity while maintaining efficacy.[34] Initial synthesis was conducted by chemists at G.D. Searle & Company, leading to its commercial introduction as Diodoquin in 1935. The compound was released for clinical evaluation in 1936, with early trials demonstrating its effectiveness in treating amebic colitis and asymptomatic cyst passers.[35] By 1937, reports confirmed cure rates exceeding 80% in small cohorts of patients with intestinal amebiasis, positioning it as a luminal amebicide superior to predecessors in ease of administration and patient compliance.[36] Its use expanded in the post-World War II era amid a surge in international travel, which increased the incidence of imported tropical diseases in non-endemic regions, making Diodoquin a staple for managing amebiasis in travelers and military personnel.[37] Key milestones included widespread adoption in the 1950s for tropical disease prophylaxis, but by the 1970s, growing concerns over neurotoxicity—linked to optic neuropathy and subacute myelo-opticoneuropathy (SMON) in related halogenated hydroxyquinolines—prompted regulatory scrutiny and restrictions on its use, particularly for prolonged or high-dose therapy.[18] Despite these issues, its historical role in anti-amoebic therapy influenced subsequent drug development, emphasizing safer luminal agents.[38]Current Regulatory Status
In the United States, diiodohydroxyquinoline (iodoquinol) is not approved by the Food and Drug Administration (FDA) for any indication as of 2025 and is classified as an unapproved new drug, prohibiting its interstate distribution without a new drug application.[39] Marketed since the 1930s for intestinal amebiasis, commercial oral formulations were discontinued, with all companies agreeing to cease distribution following FDA notification on December 9, 2024, leading to declined use in favor of safer alternatives such as paromomycin.[39][40] It remains available through compounding pharmacies for prescription use in treating luminal infections like amebiasis.[11] Topical formulations combined with hydrocortisone are marketed for dermatological conditions in limited markets but carry FDA disclaimers stating they have not been evaluated for safety or efficacy.[41] Internationally, regulatory status varies, with iodoquinol available in generic or compounded forms in some countries for anti-amebic therapy but restricted or unavailable in others due to historical neurotoxicity concerns associated with related compounds like clioquinol.[18] For instance, it is not used in Japan following the 1970s ban on clioquinol after outbreaks of subacute myelo-optico-neuropathy (SMON), prompting caution for similar halogenated hydroxyquinolines.[42] It is absent from the World Health Organization (WHO) Model List of Essential Medicines, which prioritizes metronidazole and paromomycin for amebiasis.[43] In select markets like Australia, vaginal pessaries containing iodoquinol are approved for specific infections.[44] Current guidelines from the Centers for Disease Control and Prevention (CDC) and WHO recommend iodoquinol only as a second-line luminal agent for amebiasis when first-line therapies like metronidazole fail, emphasizing its role in eradicating intestinal cysts alongside tissue amebicides. No new regulatory approvals for iodoquinol have occurred since 2010, reflecting its niche status amid safer options.[25] Usage has never been fully withdrawn globally, but warnings were issued in the 1970s and 1980s by the FDA and others regarding risks of optic neuropathy and peripheral neuropathy, particularly with prolonged or high-dose administration.[45]References
- https://www.[drugs.com](/page/Drugs.com)/monograph/iodoquinol.html