Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
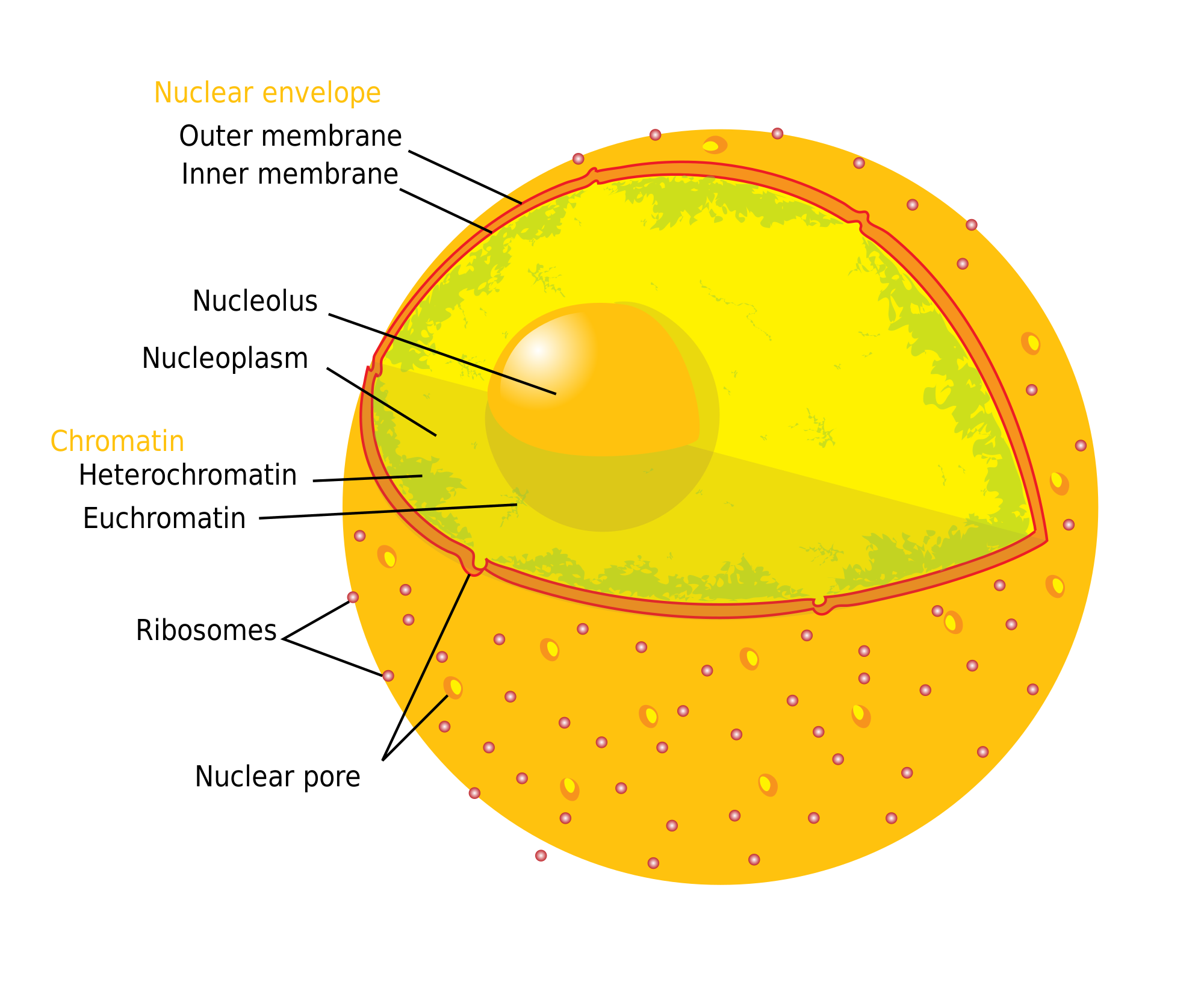
Nuclear pore complex
The nuclear pore complex (NPC), is a large protein complex giving rise to the nuclear pore. A great number of nuclear pores are studded throughout the nuclear envelope that surrounds the eukaryote cell nucleus. The pores enable the nuclear transport of macromolecules between the nucleoplasm of the nucleus and the cytoplasm of the cell. Small molecules can easily diffuse through the pores. Nuclear transport includes the transportation of RNA and ribosomal proteins from the nucleus to the cytoplasm, and the transport of proteins (such as DNA polymerase and lamins), carbohydrates, signaling molecules, and lipids into the nucleus. Each nuclear pore complex can actively mediate up to 1000 translocations per second.
The nuclear pore complex consists predominantly of a family of proteins known as nucleoporins (Nups). Each pore complex in the human cell nucleus is composed of about 1,000 individual protein molecules, from an evolutionarily conserved set of 35 distinct nucleoporins. The conserved sequences that code for nucleoporins regulate molecular transport through the nuclear pore. Nucleoporin-mediated transport does not entail direct energy expenditure but instead relies on concentration gradients associated with the RAN cycle (Ras-related nuclear protein cycle). In 2022 around 90% of the structure of the human NPC was elucidated in an open and a closed conformation, and published in a special issue of Science, featured on the cover. In 2024 the structure of the nuclear basket was solved, finalising the completion of the structure of the nuclear pore complex.
About half of the nucleoporins encompass solenoid protein domains, such as alpha solenoids or beta-propeller folds, and occasionally both as separate structural domains. Conversely, the remaining nucleoporins exhibit characteristics of "natively unfolded" or intrinsically disordered proteins, characterized by high flexibility and a lack of ordered tertiary structure. These disordered proteins, referred to as FG nucleoporins (FG-Nups), contain multiple phenylalanine–glycine repeats (FG repeats) in their amino acid sequences. FG-Nups is one of three main types of nucleoporins found in the NPC. The other two are the transmembrane Nups and the scaffold Nups. The transmembrane Nups are made up of transmembrane alpha helices and play a vital part in anchoring the NPC to the nuclear envelope. The scaffold Nups are made up of alpha solenoid and beta-propeller folds, and create the structural framework of NPCs.
The count of nuclear pore complexes varies across cell types and different stages of the cell's life cycle, with approximately 1,000 NPCs typically found in vertebrate cells. The human nuclear pore complex is a substantial structure, with a molecular weight of 120 megadaltons (MDa). Each NPC comprises eight protein subunits encircling the actual pore, forming the outer ring. Additionally, these subunits project a spoke-shaped protein over the pore channel. The central region of the pore may exhibit a plug-like structure; however, its precise nature remains unknown, and it is yet undetermined whether it represents an actual plug or merely cargo transiently caught in transit.
The nuclear pore complex (NPC) is a crucial cellular structure with a diameter of approximately 120 nanometers in vertebrates. Its channel fuses the inner and outer membranes of the nuclear envelope and varies from 5.2 nanometers in humans to 10.7 nm in the frog Xenopus laevis, with a depth of roughly 45 nm. Additionally, mRNA, being single-stranded, has a thickness ranging from 0.5 to 1 nm. The mammalian NPC has a molecular mass of about 124 MDa, comprising approximately 30 distinct protein components, each in multiple copies. The mammalian NPCs contain about 800 nucleoporins each that are organized into distinct NPC subcomplexes. Conversely, the yeast Saccharomyces cerevisiae possesses a smaller mass, estimated at only 66 MDa.
The nuclear pore complex (NPC) serves as a highly regulated gateway for the transport of molecules between the nucleus and the cytoplasm. This intricate system enables the selective passage for molecules including proteins, RNA, and signaling molecules, ensuring proper cellular function and homeostasis. Small molecules such as proteins water and ions can diffuse through NPCs, but cargoes (>40 KDa) such as RNA and protein require the participation of soluble transport receptors.
The largest family of nuclear transport receptors are karyopherins, that include importins or exportins. These are a superfamily of nuclear transport receptors that facilitate the translocation of proteins, RNAs, and ribonuclear particles across the NPC in a Ran GTP hydrolase-dependent process. This family is further subdivided to the karyopherin-α and the karyopherin-β subfamilies. Other nuclear transport receptors include NTF2 and some NTF2-like proteins.
Three models have been suggested to explain the translocation mechanism:
Hub AI
Nuclear pore complex AI simulator
(@Nuclear pore complex_simulator)
Nuclear pore complex
The nuclear pore complex (NPC), is a large protein complex giving rise to the nuclear pore. A great number of nuclear pores are studded throughout the nuclear envelope that surrounds the eukaryote cell nucleus. The pores enable the nuclear transport of macromolecules between the nucleoplasm of the nucleus and the cytoplasm of the cell. Small molecules can easily diffuse through the pores. Nuclear transport includes the transportation of RNA and ribosomal proteins from the nucleus to the cytoplasm, and the transport of proteins (such as DNA polymerase and lamins), carbohydrates, signaling molecules, and lipids into the nucleus. Each nuclear pore complex can actively mediate up to 1000 translocations per second.
The nuclear pore complex consists predominantly of a family of proteins known as nucleoporins (Nups). Each pore complex in the human cell nucleus is composed of about 1,000 individual protein molecules, from an evolutionarily conserved set of 35 distinct nucleoporins. The conserved sequences that code for nucleoporins regulate molecular transport through the nuclear pore. Nucleoporin-mediated transport does not entail direct energy expenditure but instead relies on concentration gradients associated with the RAN cycle (Ras-related nuclear protein cycle). In 2022 around 90% of the structure of the human NPC was elucidated in an open and a closed conformation, and published in a special issue of Science, featured on the cover. In 2024 the structure of the nuclear basket was solved, finalising the completion of the structure of the nuclear pore complex.
About half of the nucleoporins encompass solenoid protein domains, such as alpha solenoids or beta-propeller folds, and occasionally both as separate structural domains. Conversely, the remaining nucleoporins exhibit characteristics of "natively unfolded" or intrinsically disordered proteins, characterized by high flexibility and a lack of ordered tertiary structure. These disordered proteins, referred to as FG nucleoporins (FG-Nups), contain multiple phenylalanine–glycine repeats (FG repeats) in their amino acid sequences. FG-Nups is one of three main types of nucleoporins found in the NPC. The other two are the transmembrane Nups and the scaffold Nups. The transmembrane Nups are made up of transmembrane alpha helices and play a vital part in anchoring the NPC to the nuclear envelope. The scaffold Nups are made up of alpha solenoid and beta-propeller folds, and create the structural framework of NPCs.
The count of nuclear pore complexes varies across cell types and different stages of the cell's life cycle, with approximately 1,000 NPCs typically found in vertebrate cells. The human nuclear pore complex is a substantial structure, with a molecular weight of 120 megadaltons (MDa). Each NPC comprises eight protein subunits encircling the actual pore, forming the outer ring. Additionally, these subunits project a spoke-shaped protein over the pore channel. The central region of the pore may exhibit a plug-like structure; however, its precise nature remains unknown, and it is yet undetermined whether it represents an actual plug or merely cargo transiently caught in transit.
The nuclear pore complex (NPC) is a crucial cellular structure with a diameter of approximately 120 nanometers in vertebrates. Its channel fuses the inner and outer membranes of the nuclear envelope and varies from 5.2 nanometers in humans to 10.7 nm in the frog Xenopus laevis, with a depth of roughly 45 nm. Additionally, mRNA, being single-stranded, has a thickness ranging from 0.5 to 1 nm. The mammalian NPC has a molecular mass of about 124 MDa, comprising approximately 30 distinct protein components, each in multiple copies. The mammalian NPCs contain about 800 nucleoporins each that are organized into distinct NPC subcomplexes. Conversely, the yeast Saccharomyces cerevisiae possesses a smaller mass, estimated at only 66 MDa.
The nuclear pore complex (NPC) serves as a highly regulated gateway for the transport of molecules between the nucleus and the cytoplasm. This intricate system enables the selective passage for molecules including proteins, RNA, and signaling molecules, ensuring proper cellular function and homeostasis. Small molecules such as proteins water and ions can diffuse through NPCs, but cargoes (>40 KDa) such as RNA and protein require the participation of soluble transport receptors.
The largest family of nuclear transport receptors are karyopherins, that include importins or exportins. These are a superfamily of nuclear transport receptors that facilitate the translocation of proteins, RNAs, and ribonuclear particles across the NPC in a Ran GTP hydrolase-dependent process. This family is further subdivided to the karyopherin-α and the karyopherin-β subfamilies. Other nuclear transport receptors include NTF2 and some NTF2-like proteins.
Three models have been suggested to explain the translocation mechanism: