Recent from talks
Nothing was collected or created yet.
Asymptomatic inflammatory prostatitis
View on Wikipedia| Asymptomatic inflammatory prostatitis | |
|---|---|
 | |
| Specialty | Urology |
Asymptomatic inflammatory prostatitis is a painless inflammation of the prostate gland where there is no evidence of infection.[1] It should be distinguished from the other categories of prostatitis characterised by either pelvic pain or evidence of infection, such as chronic bacterial prostatitis, acute bacterial prostatitis and chronic pelvic pain syndrome (CPPS).[2] It is a common finding in men with benign prostatic hyperplasia.[3]
Signs and symptoms
[edit]These patients have no history of genitourinary pain complaints, but leukocytosis is noted, usually during evaluation for other conditions.[1]
Diagnosis
[edit]Diagnosis is through tests of semen, expressed prostatic secretion (EPS) or prostate tissue that reveal inflammation in the absence of symptoms.[4]
Treatment
[edit]No treatment required. It is standard practice for men with infertility and category IV prostatitis to be given a trial of antibiotics and/or anti-inflammatories, although evidence of efficacy are weak.[5] Since signs of asymptomatic prostatic inflammation may sometimes be associated with prostate cancer, this can be addressed by tests that assess the ratio of free-to-total PSA. The results of these tests were significantly different in prostate cancer and category IV prostatitis in one study.[6]
Additional images
[edit]-
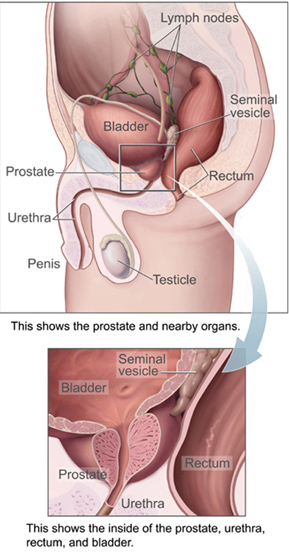
Prostate, urethra, and seminal vesicles.
-
The arteries of the pelvis.
-
Male pelvic organs seen from right side.
References
[edit]- ^ a b "Prostatitis: Benign Prostate Disease: Merck Manual Professional". Archived from the original on 28 April 2010. Retrieved 2010-04-17.
- ^ Holt JD; et al. (2016). "Common Questions About Chronic Prostatitis". Am Fam Physician. 93 (4): 290–6. PMID 26926816.
- ^ Nickel JC; Downey J; Young I; Boag S. (Dec 1999). "Asymptomatic inflammation and/or infection in benign prostatic hyperplasia". BJU International. 84 (9): 976–81. doi:10.1046/j.1464-410x.1999.00352.x. PMID 10571623. S2CID 25119588.
- ^ Clemens JQ, Meenan RT, O'Keeffe Rosetti MC, Gao SY, Calhoun EA (2005). "Incidence and clinical characteristics of National Institutes of Health type III prostatitis in the community". J. Urol. 174 (6): 2319–22. doi:10.1097/01.ju.0000182152.28519.e7. PMID 16280832.
- ^ Weidner W, Krause W, Ludwig M (1999). "Relevance of male accessory gland infection for subsequent fertility with special focus on prostatitis". Hum. Reprod. Update. 5 (5): 421–32. doi:10.1093/humupd/5.5.421. PMID 10582781.
Several inflammatory and reactive alterations of sperm quality seem to be proven; nevertheless, the impact of these findings on male fertility remains in many cases unclear."
- ^ Stancik I, Lüftenegger W, Klimpfinger M, Müller MM, Hoeltl W (2004). "Effect of NIH-IV prostatitis on free and free-to-total PSA". Eur. Urol. 46 (6): 760–4. doi:10.1016/j.eururo.2004.08.003. PMID 15548444.