Recent from talks
Nothing was collected or created yet.
Leukotriene B4
View on Wikipedia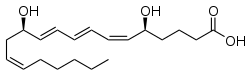 | |
| Names | |
|---|---|
| Preferred IUPAC name
(5S,6Z,8E,10E,12R,14Z)-5,12-Dihydroxyicosa-6,8,10,14-tetraenoic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H32O4 | |
| Molar mass | 336.472 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
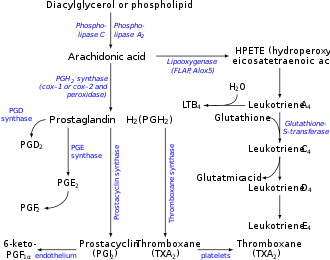
Leukotriene B4 (LTB4) is a leukotriene involved in inflammation. It has been shown to promote insulin resistance in obese mice.
Biochemistry
[edit]LTB4 is a leukotriene involved in inflammation. It is produced from leukocytes in response to inflammatory mediators and is able to induce the adhesion and activation of leukocytes on the endothelium, allowing them to bind to and cross it into the tissue.[1] In neutrophils, it is also a potent chemoattractant, and is able to induce the formation of reactive oxygen species and the release of lysosomal enzymes by these cells.[1] It is synthesized by leukotriene-A4 hydrolase from leukotriene A4.[2]
Diabetes
[edit]A study at the University of California, San Diego School of Medicine has shown that LTB4 promotes insulin resistance in obese mice.[3] Obesity is the major cause of insulin resistance in type 2 diabetes.[4]
References
[edit]- ^ a b Cotran; Kumar, Collins (1999). Robbins Pathologic Basis of Disease. Philadelphia: W.B Saunders Company. ISBN 0-7216-7335-X.
- ^ "LTA4H". uniprot. Retrieved 9 April 2013.
- ^ "Molecular Link between Obesity and Type 2 Diabetes Reveals Potential Therapy". UC San Diego Health. Archived from the original on 2022-02-18.
- ^ Li, P; Oh, DY; Bandyopadhyay, G; Lagakos, WS; Talukdar, S; Osborn, O; Johnson, A; Chung, H; Maris, M; Ofrecio, JM; Taguchi, S; Lu, M; Olefsky, JM (2015). "LTB4 promotes insulin resistance in obese mice by acting on macrophages, hepatocytes and myocytes". Nature Medicine. 21 (3): 239–247. doi:10.1038/nm.3800. PMC 4429798. PMID 25706874.
