Recent from talks
Nothing was collected or created yet.
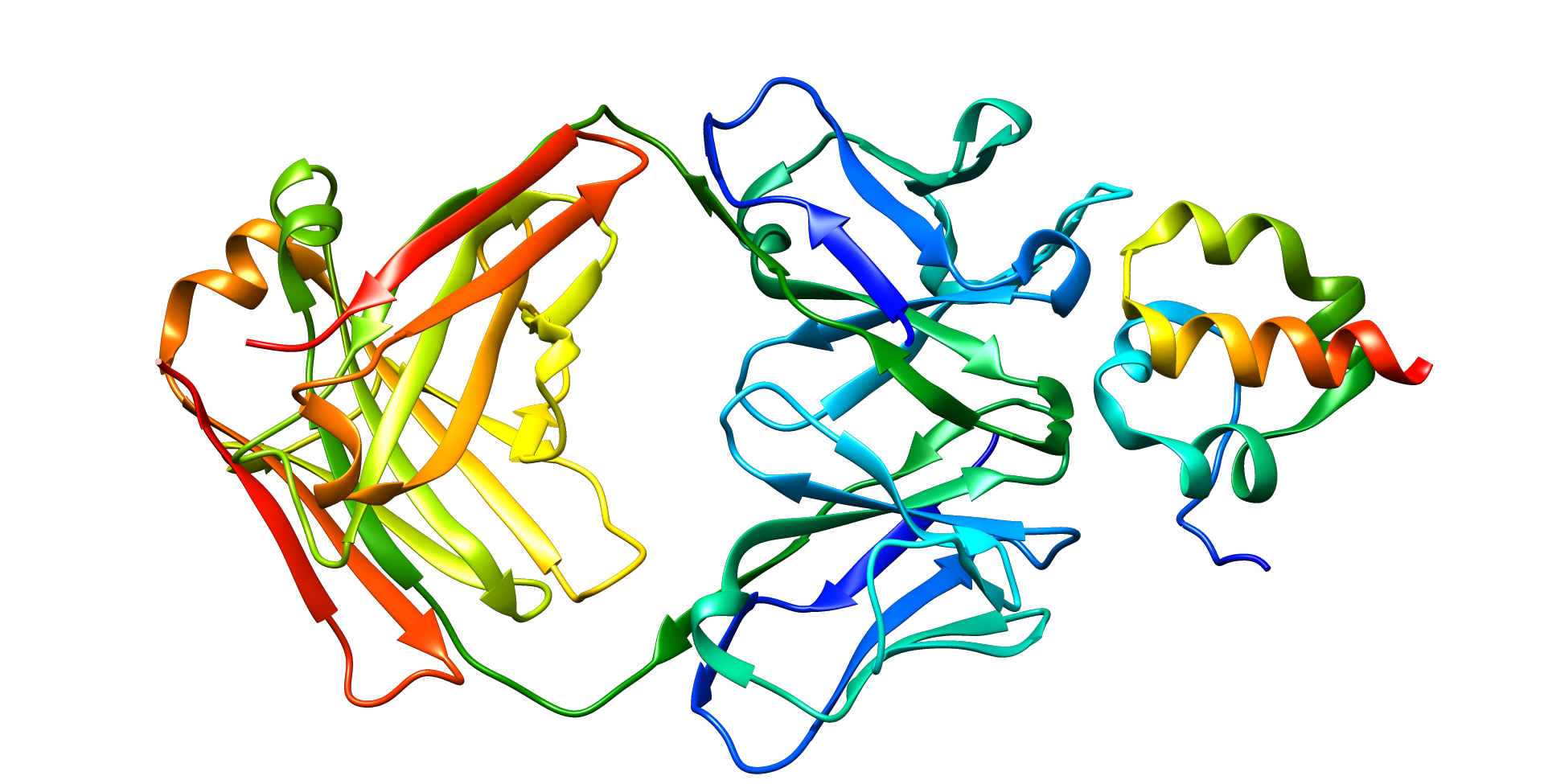
Mesothelin
View on Wikipedia| MSLN | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | MSLN, MPF, SMRP, mesothelin | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 601051; MGI: 1888992; HomoloGene: 4249; GeneCards: MSLN; OMA:MSLN - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Mesothelin, also known as MSLN, is a protein that in humans is encoded by the MSLN gene.[5][6]
Function
[edit]Mesothelin is a 40 kDa protein that is expressed in mesothelial cells.[7] The protein was first identified by its reactivity with monoclonal antibody K1.[8] Subsequent cloning studies showed that the mesothelin gene encodes a precursor protein that is processed to yield mesothelin which is attached to the cell membrane by a glycophosphatidylinositol linkage and a 31-kDa shed fragment named megakaryocyte-potentiating factor (MPF). Although it has been proposed that mesothelin may be involved in cell adhesion, its biological function is not known.[9][10] A knockout mouse line that lacks mesothelin reproduces and develops normally.[11]
Mesothelin is over expressed in several human tumors, including mesothelioma, ovarian cancer, pancreatic adenocarcinoma,[7] lung adenocarcinoma,[12] and cholangiocarcinoma.[13] Mesothelin binds MUC16 (also known as CA125), indicating that the interaction of mesothelin and MUC16 may contribute to the implantation and peritoneal spread of tumors by cell adhesion.[14] The region (residues 296-359) consisting of 64 amino acids at the N-terminus of cell surface mesothelin has been identified as the functional binding domain (named IAB) for MUC16/CA125, suggesting the mechanism of mesothelin acting as a MUC16/CA125 functional partner in cancer development.[15]

Medical applications
[edit]Mesothelin is a tumor differentiation antigen that is normally present on the mesothelial cells lining the pleura,[17] peritoneum and pericardium.[7] Since mesothelin is overexpressed in several cancers and is immunogenic, the protein could be exploited as tumor marker or as the antigenic target of a therapeutic cancer vaccine.[9][18] A 2016 review indicates that some immunotherapeutic strategies have shown encouraging results in early-phase clinical trials.[19] Elevations of serum mesothelin specific to ovarian and other cancer patients may be measured using ELISA assays.[20] Soluble mesothelin is identified as the extracellular domain of membrane-bound mesothelin shed from tumor cells according to the mass spectrometry analysis of soluble mesothelin purified from cell culture supernatant.[21]
Assays for blood-borne mesothelin and MPF for tumor diagnosis, especially applied to asbestos-related mesothelioma have been developed.[22] Elevated serum mesothelin was found in most patients with mesothelioma (71%) and ovarian cancer (67%).[23] Blood MPF and mesothelin levels were correlated, with modest accuracy for malignant pleural mesothelioma and lung cancer (sensitivity 74% and 59%, specificity 90% and 86%, respectively for MPF and mesothelin assays).[24] Circulating mesothelin is reported in nearly all pancreatic cancers,[25] however the levels in healthy persons often exceed 80 ng/mL (using 40 kD molecular weight as the conversion factor) and to widely overlap the values in the pancreatic cancer patients.[26] It was noted that the cutoff levels for normal could differ as much as 10-fold among publications, depending on the assay used[26][24][23] and thus that normal levels must be determined anew when new assays are introduced. Increase of mesothelin-specific antibodies were also detected in the sera of about 40% of patients with mesothelioma and 42% with ovarian cancer, indicating an antibody response to mesothelin was correlated with high expression of mesothelin on tumor cells.[27]
Human monoclonal antibodies HN1 and SD1 targeting mesothelin have been isolated by phage display.[28][29] Mitchell Ho and Ira Pastan at the U.S. National Institutes of Health (NIH) generated rabbit monoclonal antibodies targeting rare and poorly immunogenic epitopes of mesothelin, including the C terminus recognized by the YP218 antibody.[16] The rabbit antibodies have been "humanized" by Ho and Zhang using human immunoglobulin germline framework sequences for CDR grafting based on computational structure modeling.[30] The CAR-T cells derived from the humanized YP218 antibody (hYP218) effectively inhibit the growth of human xenograft tumors in mice.[31][32]
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000102854 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000063011 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Kojima T, Oh-eda M, Hattori K, Taniguchi Y, Tamura M, Ochi N, Yamaguchi N (September 1995). "Molecular cloning and expression of megakaryocyte potentiating factor cDNA". The Journal of Biological Chemistry. 270 (37): 21984–21990. doi:10.1074/jbc.270.37.21984. PMID 7665620.
- ^ Chang K, Pastan I (January 1996). "Molecular cloning of mesothelin, a differentiation antigen present on mesothelium, mesotheliomas, and ovarian cancers". Proceedings of the National Academy of Sciences of the United States of America. 93 (1): 136–140. Bibcode:1996PNAS...93..136C. doi:10.1073/pnas.93.1.136. PMC 40193. PMID 8552591.
- ^ a b c Hassan R, Ho M (January 2008). "Mesothelin targeted cancer immunotherapy". European Journal of Cancer. 44 (1): 46–53. doi:10.1016/j.ejca.2007.08.028. PMC 2265108. PMID 17945478.
- ^ Chang K, Pai LH, Batra JK, Pastan I, Willingham MC (January 1992). "Characterization of the antigen (CAK1) recognized by monoclonal antibody K1 present on ovarian cancers and normal mesothelium". Cancer Research. 52 (1): 181–186. PMID 1727378.
- ^ a b Hassan R, Bera T, Pastan I (June 2004). "Mesothelin: a new target for immunotherapy". Clinical Cancer Research. 10 (12 Pt 1): 3937–3942. doi:10.1158/1078-0432.CCR-03-0801. PMID 15217923.
- ^ Ho M (October 2011). "Advances in liver cancer antibody therapies: a focus on glypican-3 and mesothelin". BioDrugs. 25 (5): 275–284. doi:10.2165/11595360-000000000-00000. PMC 3198870. PMID 21942912.
- ^ Bera TK, Pastan I (April 2000). "Mesothelin is not required for normal mouse development or reproduction". Molecular and Cellular Biology. 20 (8): 2902–2906. doi:10.1128/MCB.20.8.2902-2906.2000. PMC 85523. PMID 10733593.
- ^ Ho M, Bera TK, Willingham MC, Onda M, Hassan R, FitzGerald D, Pastan I (March 2007). "Mesothelin expression in human lung cancer". Clinical Cancer Research. 13 (5): 1571–1575. doi:10.1158/1078-0432.CCR-06-2161. PMID 17332303.
- ^ Yu L, Feng M, Kim H, Phung Y, Kleiner DE, Gores GJ, et al. (October 2010). "Mesothelin as a potential therapeutic target in human cholangiocarcinoma". Journal of Cancer. 1: 141–149. doi:10.7150/jca.1.141. PMC 2948219. PMID 20922056.
- ^ Rump A, Morikawa Y, Tanaka M, Minami S, Umesaki N, Takeuchi M, Miyajima A (March 2004). "Binding of ovarian cancer antigen CA125/MUC16 to mesothelin mediates cell adhesion". The Journal of Biological Chemistry. 279 (10): 9190–9198. doi:10.1074/jbc.M312372200. PMID 14676194.
- ^ Kaneko O, Gong L, Zhang J, Hansen JK, Hassan R, Lee B, Ho M (February 2009). "A binding domain on mesothelin for CA125/MUC16". The Journal of Biological Chemistry. 284 (6): 3739–3749. doi:10.1074/jbc.M806776200. PMC 2635045. PMID 19075018.
- ^ a b Zhang YF, Phung Y, Gao W, Kawa S, Hassan R, Pastan I, Ho M (May 2015). "New high affinity monoclonal antibodies recognize non-overlapping epitopes on mesothelin for monitoring and treating mesothelioma". Scientific Reports. 5: 9928. Bibcode:2015NatSR...5.9928Z. doi:10.1038/srep09928. PMC 4440525. PMID 25996440.
- ^ Grosso F, Mannucci M, Ugo F, Ferro P, Cassinari M, Vigani A, et al. (October 2021). "Pilot Study to Evaluate Serum Soluble Mesothelin-Related Peptide (SMRP) as Marker for Clinical Monitoring of Pleural Mesothelioma (PM): Correlation with Modified RECIST Score". Diagnostics. 11 (11): 2015. doi:10.3390/diagnostics11112015. PMC 8623660. PMID 34829362.
- ^ Hassan R, Ho M (January 2008). "Mesothelin targeted cancer immunotherapy". European Journal of Cancer. 44 (1): 46–53. doi:10.1016/j.ejca.2007.08.028. PMC 2265108. PMID 17945478.
- ^ Morello A, Sadelain M, Adusumilli PS (February 2016). "Mesothelin-Targeted CARs: Driving T Cells to Solid Tumors". Cancer Discovery. 6 (2): 133–146. doi:10.1158/2159-8290.CD-15-0583. PMC 4744527. PMID 26503962.
- ^ Scholler N, Fu N, Yang Y, Ye Z, Goodman GE, Hellström KE, Hellström I (September 1999). "Soluble member(s) of the mesothelin/megakaryocyte potentiating factor family are detectable in sera from patients with ovarian carcinoma". Proceedings of the National Academy of Sciences of the United States of America. 96 (20): 11531–11536. Bibcode:1999PNAS...9611531S. doi:10.1073/pnas.96.20.11531. PMC 18068. PMID 10500211.
- ^ Ho M, Onda M, Wang QC, Hassan R, Pastan I, Lively MO (September 2006). "Mesothelin is shed from tumor cells". Cancer Epidemiology, Biomarkers & Prevention. 15 (9): 1751. doi:10.1158/1055-9965.EPI-06-0479. PMID 16985043.
- ^ Maeda M, Hino O (2006). "Blood tests for asbestos-related mesothelioma". Oncology. 71 (1–2): 26–31. doi:10.1159/000100446. PMID 17344668. S2CID 12953614.
- ^ a b Hassan R, Remaley AT, Sampson ML, Zhang J, Cox DD, Pingpank J, et al. (January 2006). "Detection and quantitation of serum mesothelin, a tumor marker for patients with mesothelioma and ovarian cancer". Clinical Cancer Research. 12 (2): 447–453. doi:10.1158/1078-0432.CCR-05-1477. PMID 16428485.
- ^ a b Iwahori K, Osaki T, Serada S, Fujimoto M, Suzuki H, Kishi Y, et al. (October 2008). "Megakaryocyte potentiating factor as a tumor marker of malignant pleural mesothelioma: evaluation in comparison with mesothelin". Lung Cancer. 62 (1): 45–54. doi:10.1016/j.lungcan.2008.02.012. PMID 18394747.
- ^ Johnston FM, Tan MC, Tan BR, Porembka MR, Brunt EM, Linehan DC, et al. (November 2009). "Circulating mesothelin protein and cellular antimesothelin immunity in patients with pancreatic cancer". Clinical Cancer Research. 15 (21): 6511–6518. doi:10.1158/1078-0432.CCR-09-0565. PMC 2782601. PMID 19843662.
- ^ a b Sharon E, Zhang J, Hollevoet K, Steinberg SM, Pastan I, Onda M, et al. (April 2012). "Serum mesothelin and megakaryocyte potentiating factor in pancreatic and biliary cancers". Clinical Chemistry and Laboratory Medicine. 50 (4): 721–725. doi:10.1515/CCLM.2011.816. PMC 6309905. PMID 22149739.
- ^ Ho M, Hassan R, Zhang J, Wang QC, Onda M, Bera T, Pastan I (May 2005). "Humoral immune response to mesothelin in mesothelioma and ovarian cancer patients". Clinical Cancer Research. 11 (10): 3814–3820. doi:10.1158/1078-0432.CCR-04-2304. PMID 15897581. S2CID 38327063.
- ^ Ho M, Feng M, Fisher RJ, Rader C, Pastan I (May 2011). "A novel high-affinity human monoclonal antibody to mesothelin". International Journal of Cancer. 128 (9): 2020–2030. doi:10.1002/ijc.25557. PMC 2978266. PMID 20635390.
- ^ Tang Z, Feng M, Gao W, Phung Y, Chen W, Chaudhary A, et al. (April 2013). "A human single-domain antibody elicits potent antitumor activity by targeting an epitope in mesothelin close to the cancer cell surface". Molecular Cancer Therapeutics. 12 (4): 416–426. doi:10.1158/1535-7163.MCT-12-0731. PMC 3624043. PMID 23371858.
- ^ Zhang YF, Ho M (April 2017). "Humanization of rabbit monoclonal antibodies via grafting combined Kabat/IMGT/Paratome complementarity-determining regions: Rationale and examples". mAbs. 9 (3): 419–429. doi:10.1080/19420862.2017.1289302. PMC 5384799. PMID 28165915.
- ^ Zhang Z, Jiang D, Yang H, He Z, Liu X, Qin W, et al. (June 2019). "Modified CAR T cells targeting membrane-proximal epitope of mesothelin enhances the antitumor function against large solid tumor". Cell Death & Disease. 10 (7): 476. doi:10.1038/s41419-019-1711-1. PMC 6572851. PMID 31209210.
- ^ Tomar S, Zhang J, Khanal M, Hong J, Venugopalan A, Jiang Q, et al. (July 2022). "Development of Highly Effective Anti-Mesothelin hYP218 Chimeric Antigen Receptor T Cells With Increased Tumor Infiltration and Persistence for Treating Solid Tumors". Molecular Cancer Therapeutics. 21 (7): 1195–1206. doi:10.1158/1535-7163.MCT-22-0073. PMC 9256778. PMID 35499461.
Further reading
[edit]- Alvarez H, Rojas PL, Yong KT, Ding H, Xu G, Prasad PN, et al. (December 2008). "Mesothelin is a specific biomarker of invasive cancer in the Barrett-associated adenocarcinoma progression model: translational implications for diagnosis and therapy". Nanomedicine. 4 (4): 295–301. doi:10.1016/j.nano.2008.06.006. PMC 2606904. PMID 18691948.
- Cristaudo A, Foddis R, Bonotti A, Simonini S, Vivaldi A, Guglielmi G, et al. (April 2010). "Polymorphisms in the putative micro-RNA-binding sites of mesothelin gene are associated with serum levels of mesothelin-related protein". Occupational and Environmental Medicine. 67 (4): 233–236. doi:10.1136/oem.2009.049205. PMID 19858537. S2CID 207017988.
- Pu RT, Pang Y, Michael CW (January 2008). "Utility of WT-1, p63, MOC31, mesothelin, and cytokeratin (K903 and CK5/6) immunostains in differentiating adenocarcinoma, squamous cell carcinoma, and malignant mesothelioma in effusions". Diagnostic Cytopathology. 36 (1): 20–25. doi:10.1002/dc.20747. hdl:2027.42/57547. PMID 18064689. S2CID 10096707.
- Inami K, Kajino K, Abe M, Hagiwara Y, Maeda M, Suyama M, et al. (December 2008). "Secretion of N-ERC/mesothelin and expression of C-ERC/mesothelin in human pancreatic ductal carcinoma". Oncology Reports. 20 (6): 1375–1380. doi:10.3892/or_00000155. PMID 19020717.
- Roe OD, Creaney J, Lundgren S, Larsson E, Sandeck H, Boffetta P, et al. (August 2008). "Mesothelin-related predictive and prognostic factors in malignant mesothelioma: a nested case-control study". Lung Cancer. 61 (2): 235–243. doi:10.1016/j.lungcan.2007.12.025. PMID 18281122.
- Johnson MD, Vito F, O'Connell MJ (June 2008). "Mesothelin expression in the leptomeninges and meningiomas". The Journal of Histochemistry and Cytochemistry. 56 (6): 579–585. doi:10.1369/jhc.2008.950477. PMC 2386771. PMID 18347077.
- Schneider J, Hoffmann H, Dienemann H, Herth FJ, Meister M, Muley T (November 2008). "Diagnostic and prognostic value of soluble mesothelin-related proteins in patients with malignant pleural mesothelioma in comparison with benign asbestosis and lung cancer". Journal of Thoracic Oncology. 3 (11): 1317–1324. doi:10.1097/JTO.0b013e318187491c. PMID 18978568. S2CID 5183602.
- Pass HI, Wali A, Tang N, Ivanova A, Ivanov S, Harbut M, et al. (January 2008). "Soluble mesothelin-related peptide level elevation in mesothelioma serum and pleural effusions". The Annals of Thoracic Surgery. 85 (1): 265–72, discussion 272. doi:10.1016/j.athoracsur.2007.07.042. PMID 18154821.
- Sapede C, Gauvrit A, Barbieux I, Padieu M, Cellerin L, Sagan C, et al. (March 2008). "Aberrant splicing and protease involvement in mesothelin release from epithelioid mesothelioma cells". Cancer Science. 99 (3): 590–594. doi:10.1111/j.1349-7006.2007.00715.x. PMC 11158685. PMID 18167128. S2CID 205233632.
- Li M, Bharadwaj U, Zhang R, Zhang S, Mu H, Fisher WE, et al. (February 2008). "Mesothelin is a malignant factor and therapeutic vaccine target for pancreatic cancer". Molecular Cancer Therapeutics. 7 (2): 286–296. doi:10.1158/1535-7163.MCT-07-0483. PMC 2929838. PMID 18281514.
- Uehara N, Matsuoka Y, Tsubura A (February 2008). "Mesothelin promotes anchorage-independent growth and prevents anoikis via extracellular signal-regulated kinase signaling pathway in human breast cancer cells". Molecular Cancer Research. 6 (2): 186–193. doi:10.1158/1541-7786.MCR-07-0254. PMID 18245228.
- Iwahori K, Osaki T, Serada S, Fujimoto M, Suzuki H, Kishi Y, et al. (October 2008). "Megakaryocyte potentiating factor as a tumor marker of malignant pleural mesothelioma: evaluation in comparison with mesothelin". Lung Cancer. 62 (1): 45–54. doi:10.1016/j.lungcan.2008.02.012. PMID 18394747.
- Creaney J, Yeoman D, Demelker Y, Segal A, Musk AW, Skates SJ, Robinson BW (August 2008). "Comparison of osteopontin, megakaryocyte potentiating factor, and mesothelin proteins as markers in the serum of patients with malignant mesothelioma". Journal of Thoracic Oncology. 3 (8): 851–857. doi:10.1097/JTO.0b013e318180477b. PMID 18670302. S2CID 12405738.
- Shiomi K, Hagiwara Y, Sonoue K, Segawa T, Miyashita K, Maeda M, et al. (March 2008). "Sensitive and specific new enzyme-linked immunosorbent assay for N-ERC/mesothelin increases its potential as a useful serum tumor marker for mesothelioma". Clinical Cancer Research. 14 (5): 1431–1437. doi:10.1158/1078-0432.CCR-07-1613. PMID 18316566.
- Kaneko O, Gong L, Zhang J, Hansen JK, Hassan R, Lee B, Ho M (February 2009). "A binding domain on mesothelin for CA125/MUC16". The Journal of Biological Chemistry. 284 (6): 3739–3749. doi:10.1074/jbc.M806776200. PMC 2635045. PMID 19075018.
- Chang MC, Chen CA, Hsieh CY, Lee CN, Su YN, Hu YH, Cheng WF (December 2009). "Mesothelin inhibits paclitaxel-induced apoptosis through the PI3K pathway". The Biochemical Journal. 424 (3): 449–458. doi:10.1042/BJ20082196. PMID 19747165.
- Cheng WF, Huang CY, Chang MC, Hu YH, Chiang YC, Chen YL, et al. (April 2009). "High mesothelin correlates with chemoresistance and poor survival in epithelial ovarian carcinoma". British Journal of Cancer. 100 (7): 1144–1153. doi:10.1038/sj.bjc.6604964. PMC 2669998. PMID 19293794.
- Hellstrom I, Hellstrom KE (2008). "SMRP and HE4 as Biomarkers for Ovarian Carcinoma when Used Alone and in Combination with CA125 and/Or Each Other". Ovarian Cancer. Advances in Experimental Medicine and Biology. Vol. 622. pp. 15–21. doi:10.1007/978-0-387-68969-2_2. ISBN 978-0-387-68966-1. PMID 18546615.
- Bharadwaj U, Li M, Chen C, Yao Q (November 2008). "Mesothelin-induced pancreatic cancer cell proliferation involves alteration of cyclin E via activation of signal transducer and activator of transcription protein 3". Molecular Cancer Research. 6 (11): 1755–1765. doi:10.1158/1541-7786.MCR-08-0095. PMC 2929833. PMID 19010822.
External links
[edit]- MSLN human gene location in the UCSC Genome Browser.
- MSLN human gene details in the UCSC Genome Browser.
- Overview of all the structural information available in the PDB for UniProt: Q13421 (Mesothelin) at the PDBe-KB.