Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
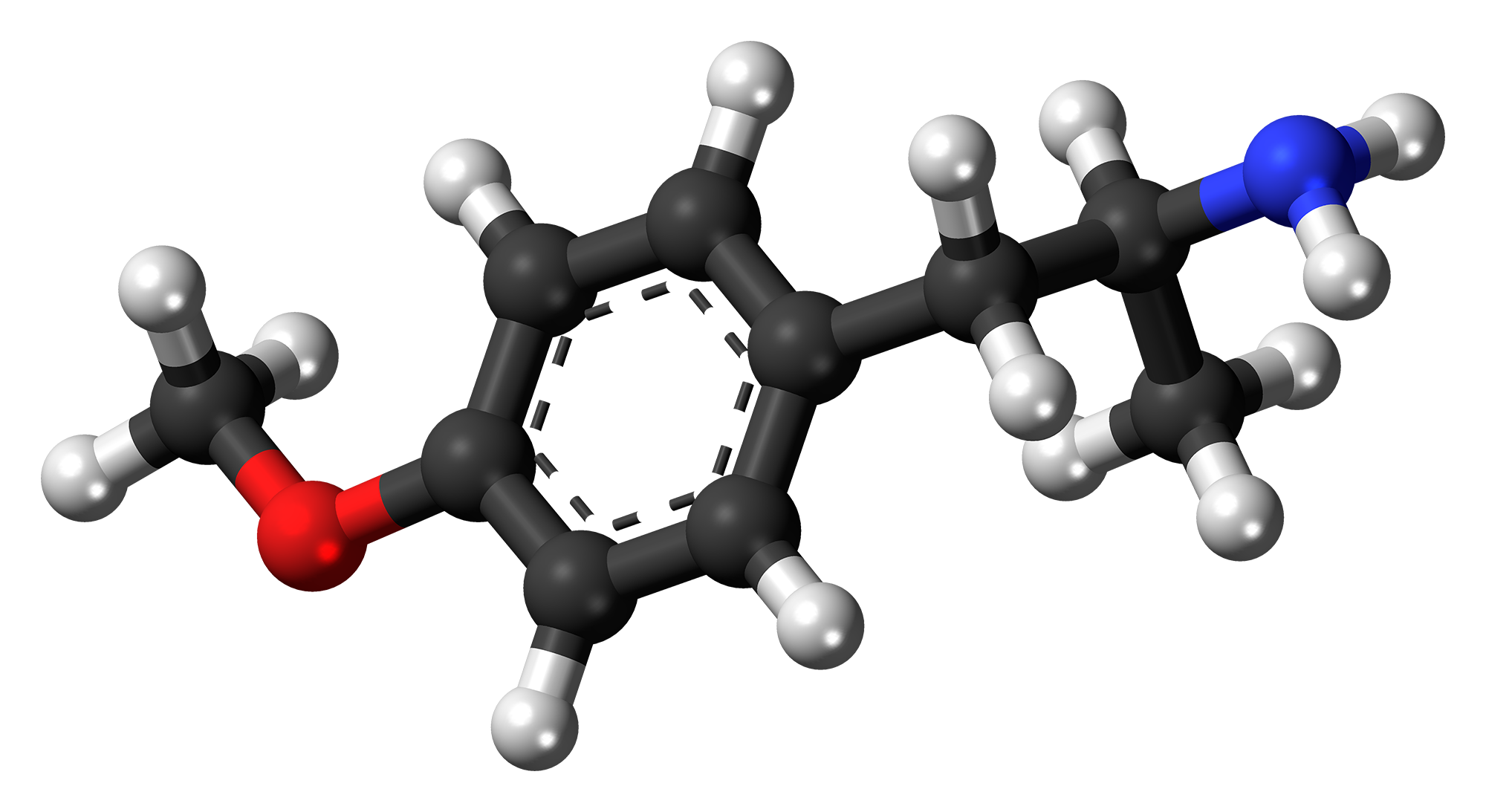
Para-Methoxyamphetamine
para-Methoxyamphetamine (PMA), also known as 4-methoxyamphetamine (4-MA), is a designer drug of the amphetamine class with serotonergic effects. Unlike other similar drugs of this family, PMA does not produce stimulant, euphoriant, or entactogenic effects, and behaves more like an antidepressant in comparison, though it does have some psychedelic properties.
PMA has been found in tablets touted as MDMA (ecstasy) although its effects are markedly different compared to those of MDMA. The consequences of such deception have often included hospitalization and death for unwitting users. PMA is commonly synthesized from anethole, the flavor compound of anise and fennel, mainly because the starting material for MDMA, safrole, has become less available due to law enforcement action, causing illicit drug manufacturers to use anethole as an alternative.
According to Alexander Shulgin in PiHKAL (Phenethylamines I Have Known and Loved), the effects of PMA at doses of 50 to 80 mg orally included hypertension, diethyltryptamine (DET)-reminiscent effects, distinct after-images, and some paresthesia, "intoxication" or alcohol-like intoxication, and no psychedelic effects. In clinical studies, PMA produced "excitation, other central effects, and sympathomimetic effects, but similarly no psychotomimetic effects. Animal studies suggested that PMA would have partial psychedelic effects, but this did not seem to prove true in humans. PMA only partially substituted for the psychostimulant amphetamine in drug discrimination tests. It did not substitute for MDMA in rodents, suggesting lack of entactogenic effects.
PMA has been associated with numerous adverse reactions including death. Effects of PMA ingestion include many effects of the hallucinogenic amphetamines including accelerated and irregular heartbeat, blurred vision, and a strong feeling of intoxication that is often unpleasant. At high doses unpleasant effects such as nausea and vomiting, severe hyperthermia and hallucinations may occur. The effects of PMA also seem to be much more unpredictable and variable between individuals than those of MDMA, and sensitive individuals may die from a dose of PMA by which a less susceptible person might only be mildly affected. While PMA alone may cause significant toxicity, the combination of PMA with MDMA has a synergistic effect that seems to be particularly hazardous. Since PMA has a slow onset of effects, several deaths have occurred where individuals have taken a pill containing PMA, followed by a pill containing MDMA some time afterwards due to thinking that the first pill was not active.
PMA overdose can be a serious medical emergency that may occur at only slightly above the usual recreational dose range, especially if PMA is mixed with other stimulant drugs such as cocaine or MDMA. Characteristic symptoms are pronounced hyperthermia, tachycardia, and hypertension, along with agitation, confusion, and convulsions. PMA overdose also tends to cause hypoglycaemia and hyperkalaemia, which can help to distinguish it from MDMA overdose. Complications can sometimes include more serious symptoms such as rhabdomyolysis and cerebral hemorrhage, requiring emergency surgery. There is no specific antidote, so treatment is symptomatic, and usually includes both external cooling, and internal cooling via IV infusion of cooled saline. Benzodiazepines are used initially to control convulsions, with stronger anticonvulsants such as phenytoin or thiopental used if convulsions continue. Blood pressure can be lowered either with a combination of alpha blockers and beta blockers (or a mixed alpha/beta blocker), or with other drugs such as nifedipine or nitroprusside. Serotonin antagonists and dantrolene may be used as required. Despite the seriousness of the condition, the majority of patients survive if treatment is given in time, however, patients with a core body temperature over 40 °C at presentation tend to have a poor prognosis.
PMA acts as a selective serotonin releasing agent (SSRA) with weak effects on dopamine and norepinephrine transporters. Its EC50 values for induction of monoamine release are 166 nM for dopamine and 867 nM for norepinephrine in rat brain synaptosomes, whereas serotonin was not reported. The drug has been found to robustly increase brain serotonin levels and to weakly increase brain dopamine levels in rodents in vivo. Relative to MDMA, PMA appears to be considerably less effective as a releaser of serotonin, with properties more akin to a serotonin reuptake inhibitor in comparison.
PMA has also been shown to act as a potent monoamine oxidase inhibitor (MAOI), specifically as a reversible inhibitor of the enzyme monoamine oxidase A (MAO-A) with no significant effects on monoamine oxidase B (MAO-B). The IC50 of PMA for MAO-A inhibition has been reported to be 300 to 600 nM.
PMA shows very low affinities for the serotonin 5-HT1A, 5-HT2A, and 5-HT2C receptors. Its affinities (Ki) for these receptors have been reported to be >20,000 nM, 11,200 nM, and >13,000 nM, respectively. In another earlier study, PMA similarly showed very weak affinity for serotonin receptors, including the serotonin 5-HT1 and 5-HT2 receptors (Ki = 79,400 nM and 33,600 nM, respectively). On the other hand, PMA shows much higher affinities for the mouse and rat trace amine-associated receptor 1 (TAAR1).
Hub AI
Para-Methoxyamphetamine AI simulator
(@Para-Methoxyamphetamine_simulator)
Para-Methoxyamphetamine
para-Methoxyamphetamine (PMA), also known as 4-methoxyamphetamine (4-MA), is a designer drug of the amphetamine class with serotonergic effects. Unlike other similar drugs of this family, PMA does not produce stimulant, euphoriant, or entactogenic effects, and behaves more like an antidepressant in comparison, though it does have some psychedelic properties.
PMA has been found in tablets touted as MDMA (ecstasy) although its effects are markedly different compared to those of MDMA. The consequences of such deception have often included hospitalization and death for unwitting users. PMA is commonly synthesized from anethole, the flavor compound of anise and fennel, mainly because the starting material for MDMA, safrole, has become less available due to law enforcement action, causing illicit drug manufacturers to use anethole as an alternative.
According to Alexander Shulgin in PiHKAL (Phenethylamines I Have Known and Loved), the effects of PMA at doses of 50 to 80 mg orally included hypertension, diethyltryptamine (DET)-reminiscent effects, distinct after-images, and some paresthesia, "intoxication" or alcohol-like intoxication, and no psychedelic effects. In clinical studies, PMA produced "excitation, other central effects, and sympathomimetic effects, but similarly no psychotomimetic effects. Animal studies suggested that PMA would have partial psychedelic effects, but this did not seem to prove true in humans. PMA only partially substituted for the psychostimulant amphetamine in drug discrimination tests. It did not substitute for MDMA in rodents, suggesting lack of entactogenic effects.
PMA has been associated with numerous adverse reactions including death. Effects of PMA ingestion include many effects of the hallucinogenic amphetamines including accelerated and irregular heartbeat, blurred vision, and a strong feeling of intoxication that is often unpleasant. At high doses unpleasant effects such as nausea and vomiting, severe hyperthermia and hallucinations may occur. The effects of PMA also seem to be much more unpredictable and variable between individuals than those of MDMA, and sensitive individuals may die from a dose of PMA by which a less susceptible person might only be mildly affected. While PMA alone may cause significant toxicity, the combination of PMA with MDMA has a synergistic effect that seems to be particularly hazardous. Since PMA has a slow onset of effects, several deaths have occurred where individuals have taken a pill containing PMA, followed by a pill containing MDMA some time afterwards due to thinking that the first pill was not active.
PMA overdose can be a serious medical emergency that may occur at only slightly above the usual recreational dose range, especially if PMA is mixed with other stimulant drugs such as cocaine or MDMA. Characteristic symptoms are pronounced hyperthermia, tachycardia, and hypertension, along with agitation, confusion, and convulsions. PMA overdose also tends to cause hypoglycaemia and hyperkalaemia, which can help to distinguish it from MDMA overdose. Complications can sometimes include more serious symptoms such as rhabdomyolysis and cerebral hemorrhage, requiring emergency surgery. There is no specific antidote, so treatment is symptomatic, and usually includes both external cooling, and internal cooling via IV infusion of cooled saline. Benzodiazepines are used initially to control convulsions, with stronger anticonvulsants such as phenytoin or thiopental used if convulsions continue. Blood pressure can be lowered either with a combination of alpha blockers and beta blockers (or a mixed alpha/beta blocker), or with other drugs such as nifedipine or nitroprusside. Serotonin antagonists and dantrolene may be used as required. Despite the seriousness of the condition, the majority of patients survive if treatment is given in time, however, patients with a core body temperature over 40 °C at presentation tend to have a poor prognosis.
PMA acts as a selective serotonin releasing agent (SSRA) with weak effects on dopamine and norepinephrine transporters. Its EC50 values for induction of monoamine release are 166 nM for dopamine and 867 nM for norepinephrine in rat brain synaptosomes, whereas serotonin was not reported. The drug has been found to robustly increase brain serotonin levels and to weakly increase brain dopamine levels in rodents in vivo. Relative to MDMA, PMA appears to be considerably less effective as a releaser of serotonin, with properties more akin to a serotonin reuptake inhibitor in comparison.
PMA has also been shown to act as a potent monoamine oxidase inhibitor (MAOI), specifically as a reversible inhibitor of the enzyme monoamine oxidase A (MAO-A) with no significant effects on monoamine oxidase B (MAO-B). The IC50 of PMA for MAO-A inhibition has been reported to be 300 to 600 nM.
PMA shows very low affinities for the serotonin 5-HT1A, 5-HT2A, and 5-HT2C receptors. Its affinities (Ki) for these receptors have been reported to be >20,000 nM, 11,200 nM, and >13,000 nM, respectively. In another earlier study, PMA similarly showed very weak affinity for serotonin receptors, including the serotonin 5-HT1 and 5-HT2 receptors (Ki = 79,400 nM and 33,600 nM, respectively). On the other hand, PMA shows much higher affinities for the mouse and rat trace amine-associated receptor 1 (TAAR1).