Recent from talks
All channels
Be the first to start a discussion here.
Be the first to start a discussion here.
Be the first to start a discussion here.
Be the first to start a discussion here.
Welcome to the community hub built to collect knowledge and have discussions related to Tetraselenium tetranitride.
Nothing was collected or created yet.
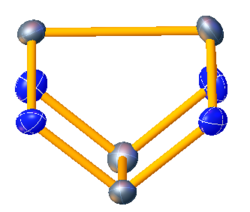
Tetraselenium tetranitride
View on Wikipediafrom Wikipedia
 | |
| Names | |
|---|---|
| IUPAC name
2,4,6,8-tetraselena-1,3,5,7-tetrazatricyclo[3.3.0.03,7]octane
| |
| Other names
nitrogen selenide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Se4N4 | |
| Molar mass | 371.912 g·mol−1 |
| Appearance | orange solid |
| Density | 4.223 g/cm3[1] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
explosive |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tetraselenium tetranitride is the inorganic compound with the formula Se4N4. Like the analogous tetrasulfur tetranitride S4N4, Se4N4 is an orange solid. It is however less soluble and more shock-sensitive than S4N4.[2]
As determined by X-ray crystallography, Se4N4 adopts a cage structure similar to that of S4N4. The Se−Se and Se−N distances are 2.740 and 1.800 Å, respectively. The N−Se−N angles are 90°.[1][3]
Among its many reactions,[4][5] Se4N4 reacts with aluminium bromide to form adducts of Se2N2.[6]
References
[edit]- ^ a b Folkerts, Hella; Neumüller, Bernhard; Dehnicke, Kurt (1994). "Synthese und Kristallstruktur einer neuen Modifikation von Tetraselentetranitrid, Se4N4". Zeitschrift für anorganische und allgemeine Chemie. 620 (6): 1011–1015. doi:10.1002/zaac.19946200610.
- ^ Klapötke, T.M. (1992). "Binary Selenium-Nitrogen Species and Related Compounds". In R. Steudel (ed.). The Chemistry of Inorganic Ring Systems. Studies in Inorganic Chemistry. Vol. 14. Elsevier. p. 409. doi:10.1016/B978-0-444-88933-1.50025-8. ISBN 9780444889331.
- ^ Bärnighausen, H.; von Volkmann, T.; Jander, J. (1966). "Die Kristallstruktur von Tetrastickstofftetraselenid N4Se4". Acta Crystallographica. 21 (4): 571–577. Bibcode:1966AcCry..21..571B. doi:10.1107/S0365110X66003475.
- ^ Kelly, P. F.; Woollins, J. D. (1993). "The Reactivity of Se4N4 in Liquid Ammonia". Polyhedron. 12 (10): 1129–1133. doi:10.1016/S0277-5387(00)88201-7.
- ^ Kelly, P. F.; Slawin, A. M. Z.; Soriano-Rama, A. (1997). "Use of Se4N4 and Se(NSO)2 in the Preparation of Palladium Adducts of Diselenium Dinitride, Se2N2; Crystal Structure of [PPh4]2[Pd2Br6(Se2N2)]". Dalton Transactions. 1997 (4): 559–562. doi:10.1039/a606311j.
- ^ Kelly, P. F.; Slawin, A. M. Z. (1996). "Preparation and Crystal Structure of [(AlBr3)2(Se2N2)], the First Example of a Main-Group Element Adduct of Diselenium Dinitride". Dalton Transactions. 1996 (21): 4029–4030. doi:10.1039/DT9960004029.