Recent from talks
Nothing was collected or created yet.
Cystatin C
View on WikipediaCystatin C or cystatin 3 (formerly gamma trace, post-gamma-globulin, or neuroendocrine basic polypeptide),[5] a protein encoded by the CST3 gene, is mainly used as a biomarker of kidney function. Recently, it has been studied for its role in predicting new-onset or deteriorating cardiovascular disease. It also seems to play a role in brain disorders involving amyloid (a specific type of protein deposition), such as Alzheimer's disease. In humans, all cells with a nucleus (cell core containing the DNA) produce cystatin C as a chain of 120 amino acids. It is found in virtually all tissues and body fluids. It is a potent inhibitor of lysosomal proteinases (enzymes from a special subunit of the cell that break down proteins) and probably one of the most important extracellular inhibitors of cysteine proteases (it prevents the breakdown of proteins outside the cell by a specific type of protein degrading enzymes). Cystatin C belongs to the type 2 cystatin gene family.
Role in medicine
[edit]Kidney function
[edit]Glomerular filtration rate (GFR), a marker of kidney health, is most accurately measured by injecting compounds such as inulin, radioisotopes such as 51chromium-EDTA, 125I-iothalamate, 99mTc-DTPA or radiocontrast agents such as iohexol, but these techniques are complicated, costly, time-consuming and have potential side-effects.[6][7] Creatinine is the most widely used biomarker of kidney function. It is inaccurate at detecting mild renal impairment, and levels can vary with muscle mass but not with protein intake. Urea levels might change with protein intake.[8] Formulas such as the Cockcroft and Gault formula and the MDRD formula (see Renal function) try to adjust for these variables.
Cystatin C has a low molecular weight (approximately 13.3 kilodaltons), and it is removed from the bloodstream by glomerular filtration in the kidneys. If kidney function and glomerular filtration rate decline, the blood levels of cystatin C rise. Cross-sectional studies (based on a single point in time) suggest that serum levels of cystatin C are a more precise test of kidney function (as represented by the glomerular filtration rate, GFR) than serum creatinine levels.[7][9] Longitudinal studies (following cystatin C over time) are sparse, but some show promising results.[10][11][12] Although studies are somewhat divergent, most studies find that cystatin C levels are less dependent on age, gender, ethnicity, diet, and muscle mass compared to creatinine,[13][14] and that cystatin C is equal or superior to the other available biomarkers in a range of different patient populations, including diabetic patients, in chronic kidney disease (CKD), and after kidney transplant.[15] It has been suggested that cystatin C might predict the risk of developing CKD, thereby signaling a state of 'preclinical' kidney dysfunction.[16] Additionally, the age-related rise in serum cystatin C is a powerful predictor of adverse age-related health outcomes, including all-cause mortality, death from cardiovascular disease, multimorbidity, and declining physical and cognitive function.[17] The UK's National Institute for Health and Care Excellence (NICE) guideline for the assessment and management of CKD in adults concluded that using serum cystatin C to estimate GFR is more specific for important disease outcomes than use of serum creatinine, and may reduce overdiagnosis in patients with a borderline diagnosis, reducing unnecessary appointments, patient worries, and the overall burden of CKD in the population.[18]
Studies have also investigated cystatin C as a marker of kidney function in the adjustment of medication dosages.[19][20]
Cystatin C levels have been reported to be altered in patients with cancer,[21][22][23] (even subtle) thyroid dysfunction[24][25][26] and glucocorticoid therapy in some[27][28] but not all[29] situations. Other reports have found that levels are influenced by cigarette smoking and levels of C-reactive protein.[30] However, inflammation does not cause an increase in the production of cystatin C, since elective surgical procedures, producing a strong inflammatory response in patients, do not change the plasma concentration of cystatin C.[medical citation needed] Levels seem to be increased in HIV infection, which might or might not reflect actual renal dysfunction.[31][32][33] The role of cystatin C to monitor GFR during pregnancy remains controversial.[34][35] Like creatinine, the elimination of cystatin C via routes other than the kidney increases with worsening GFR.[36]
Death and cardiovascular disease
[edit]Kidney dysfunction increases the risk of death and cardiovascular disease.[37][38] Several studies have found that increased levels of cystatin C are associated with the risk of death, several types of cardiovascular disease (including myocardial infarction, stroke, heart failure, peripheral arterial disease and metabolic syndrome) and healthy aging.[citation needed][clarification needed] Some studies have found cystatin C to be better in this regard than serum creatinine or creatinine-based GFR equations.[39][40][41][42][43][44][45][46][47][48][49][50] Because the association of cystatin C with long term outcomes has appeared stronger than what could be expected for GFR, it has been hypothesized that cystatin C might also be linked to mortality in a way independent of kidney function.[51] In keeping with its housekeeping gene properties, it has been suggested that cystatin C might be influenced by the basal metabolic rate.[52]
Proposed shrunken pore syndrome
[edit]The glomerular sieving coefficients for 10–30 kDa plasma proteins in the human kidney are relatively high with coefficients between 0.9 and 0.07.[medical citation needed][53] These relatively high sieving coefficients, combined with the high production of ultrafiltrate in health, means that proteins less than or equal to 30 kDa in plasma normally are mainly cleared by the kidneys and at least 85% of the clearance of cystatin C occurs in the kidney.[medical citation needed][54] If the pores of the glomerular membrane shrink, the filtration of bigger molecules, e.g. cystatin C, will decrease, whereas the filtration of small molecules, like water and creatinine, will be less affected. In this case, cystatin C-based estimates of GFR, eGFRcystatin C, will be lower than creatinine-based estimates eGFRcreatinine, so that a hypothesized condition, named shrunken pore syndrome, is identified by a low eGFRcystatin C/eGFRcreatinine-ratio.[medical citation needed][55] This syndrome is associated with a very strong increase in mortality.[56]
Neurologic disorders
[edit]Mutations in the cystatin 3 gene are responsible for the Icelandic type of hereditary cerebral amyloid angiopathy, a condition predisposing to intracerebral haemorrhage, stroke and dementia.[57][58] The condition is inherited in a dominant fashion. The monomeric cystatin C forms dimers and oligomers by domain swapping[59] and the structures of both the dimers[60] and oligomers[61] have been determined.
Since cystatin 3 also binds amyloid β and reduces its aggregation and deposition, it is a potential target in Alzheimer's disease.[62][63] Although not all studies have confirmed this, the overall evidence is in favor of a role for CST3 as a susceptibility gene for Alzheimer's disease.[64] Cystatin C levels have been reported to be higher in subjects with Alzheimer's disease.[65]
The role of cystatin C in multiple sclerosis and other demyelinating diseases (characterized by a loss of the myelin nerve sheath) remains controversial.[66]
Other roles
[edit]Cystatin C levels are decreased in atherosclerotic (so-called 'hardening' of the arteries) and aneurysmal (saccular bulging) lesions of the aorta.[67][68][69][70] Genetic and prognostic studies also suggest a role for cystatin C.[71][72] Breakdown of parts of the vessel wall in these conditions is thought to result from an imbalance between proteinases (cysteine proteases and matrix metalloproteinases, increased) and their inhibitors (such as cystatin C, decreased).
A few studies have looked at the role of cystatin C or the CST3 gene in age-related macular degeneration.[73][74] Cystatin C has also been investigated as a prognostic marker in several forms of cancer.[75][76] Its role in pre-eclampsia remains to be confirmed.[77][78][79][80]
Laboratory measurement
[edit]Cystatin C can be measured in a random sample of serum (the fluid in blood from which the red blood cells and clotting factors have been removed) using immunoassays such as nephelometry or particle-enhanced turbidimetry.[81] It is a more expensive test than serum creatinine (around $2 or $3, compared to $0.02 to $0.15), which can be measured with a Jaffe reaction.[82][83][84]
Reference values differ in many populations and with sex and age. Across different studies, the mean reference interval (as defined by the 5th and 95th percentile) was between 0.52 and 0.98 mg/L. For women, the average reference interval is 0.52 to 0.90 mg/L with a mean of 0.71 mg/L. For men, the average reference interval is 0.56 to 0.98 mg/L with a mean of 0.77 mg/L.[81] The normal values decrease until the first year of life, remaining relatively stable before they increase again, especially beyond age 50.[85][86][87] Creatinine levels increase until puberty and differ according to gender from then on, making their interpretation problematic for pediatric patients.[86][88]
In a large study from the United States National Health and Nutrition Examination Survey, the reference interval (as defined by the 1st and 99th percentile) was between 0.57 and 1.12 mg/L. This interval was 0.55 - 1.18 for women and 0.60 - 1.11 for men. Non-Hispanic blacks and Mexican Americans had lower normal cystatin C levels.[85] Other studies have found that in patients with an impaired renal function, women have lower and blacks have higher cystatin C levels for the same GFR.[89] For example, the cut-off values of cystatin C for CKD for a 60-year-old white women would be 1.12 mg/L and 1.27 mg/L in a black man (a 13% increase). For serum creatinine values adjusted with the MDRD equation, these values would be 0.95 mg/dL to 1.46 mg/dL (a 54% increase).[90]
Based on a threshold level of 1.09 mg/L (the 99th percentile in a population of 20- to 39-year-olds without hypertension, diabetes, microalbuminuria or macroalbuminuria or higher than stage 3 chronic kidney disease), the prevalence of increased levels of cystatin C in the United States was 9.6% in subjects of normal weight, increasing in overweight and obese individuals.[91] In Americans aged 60 and 80 and older, serum cystatin is increased in 41% and more than 50%.[85]
Molecular biology
[edit]The cystatin superfamily encompasses proteins that contain multiple cystatin-like sequences. Some of the members are active cysteine protease inhibitors, while others have lost or perhaps never acquired this inhibitory activity. There are three inhibitory families in the superfamily, including the type 1 cystatins (stefins), type 2 cystatins and the kininogens. The type 2 cystatin proteins are a class of cysteine proteinase inhibitors found in a variety of human fluids and secretions, where they appear to provide protective functions. The cystatin locus on the short arm of chromosome 20 contains the majority of the type 2 cystatin genes and pseudogenes.
The CST3 gene is located in the cystatin locus and comprises 3 exons (coding regions, as opposed to introns, non-coding regions within a gene), spanning 4.3 kilo-base pairs. It encodes the most abundant extracellular inhibitor of cysteine proteases. It is found in high concentrations in biological fluids and is expressed in virtually all organs of the body (CST3 is a housekeeping gene). The highest levels are found in semen, followed by breastmilk, tears and saliva. The hydrophobic leader sequence indicates that the protein is normally secreted. There are three polymorphisms in the promoter region of the gene, resulting in two common variants.[92] Several single nucleotide polymorphisms have been associated with altered cystatin C levels.[93]
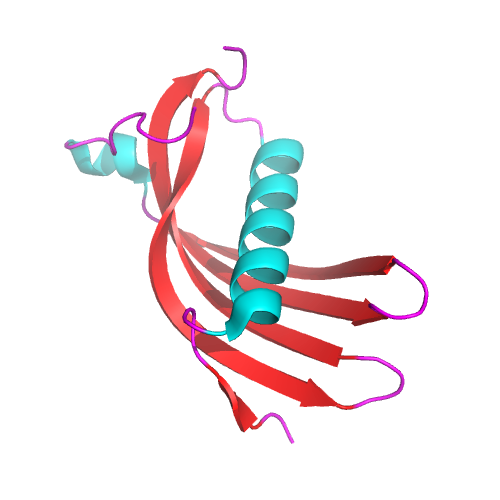
Cystatin C is a non-glycosylated, basic protein (isoelectric point at pH 9.3). The crystal structure of cystatin C is characterized by a short alpha helix and a long alpha helix which lies across a large antiparallel, five-stranded beta sheet. Like other type 2 cystatins, it has two disulfide bonds. Around 50% of the molecules carry a hydroxylated proline. Cystatin C forms dimers (molecule pairs) by exchanging subdomains; in the paired state, each half is made up of the long alpha helix and one beta strand of one partner, and four beta strands of the other partner.[94]
History
[edit]Cystatin C was first described as 'gamma-trace' in 1961 as a trace protein together with other ones (such as beta-trace) in the cerebrospinal fluid and in the urine of people with kidney failure.[95] Grubb and Löfberg first reported its amino acid sequence.[95] They noticed it was increased in patients with advanced kidney failure.[96] It was first proposed as a measure of glomerular filtration rate by Grubb and coworkers in 1985.[97][98]
Use of serum creatinine and cystatin C was found very effective in accurately reflecting the GFR in a study reported in the July 5, 2012, issue of the New England Journal of Medicine.[99]
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000101439 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000027447 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Alzforum: AlzGene". Archived from the original on 2004-12-27.
- ^ Zahran A, El-Husseini A, Shoker A (2007). "Can cystatin C replace creatinine to estimate glomerular filtration rate? A literature review". American Journal of Nephrology. 27 (2): 197–205. doi:10.1159/000100907. PMID 17361076. S2CID 25523216.
- ^ a b Roos JF, Doust J, Tett SE, Kirkpatrick CM (March 2007). "Diagnostic accuracy of cystatin C compared to serum creatinine for the estimation of renal dysfunction in adults and children--a meta-analysis". Clinical Biochemistry. 40 (5–6): 383–91. doi:10.1016/j.clinbiochem.2006.10.026. PMID 17316593.
- ^ King AJ, Levey AS (May 1993). "Dietary protein and renal function". Journal of the American Society of Nephrology. 3 (11): 1723–37. doi:10.1681/ASN.V3111723. PMID 8329667.
- ^ Dharnidharka VR, Kwon C, Stevens G (August 2002). "Serum cystatin C is superior to serum creatinine as a marker of kidney function: a meta-analysis". American Journal of Kidney Diseases. 40 (2): 221–6. doi:10.1053/ajkd.2002.34487. PMID 12148093.
- ^ Premaratne E, MacIsaac RJ, Finch S, Panagiotopoulos S, Ekinci E, Jerums G (May 2008). "Serial measurements of cystatin C are more accurate than creatinine-based methods in detecting declining renal function in type 1 diabetes". Diabetes Care. 31 (5): 971–3. doi:10.2337/dc07-1588. PMID 18319326.
- ^ Perkins BA, Nelson RG, Ostrander BE, Blouch KL, Krolewski AS, Myers BD, Warram JH (May 2005). "Detection of renal function decline in patients with diabetes and normal or elevated GFR by serial measurements of serum cystatin C concentration: results of a 4-year follow-up study". Journal of the American Society of Nephrology. 16 (5): 1404–12. doi:10.1681/ASN.2004100854. PMC 2429917. PMID 15788478.
- ^ Corrao AM, Lisi G, Di Pasqua G, Guizzardi M, Marino N, Ballone E, Chiesa PL (January 2006). "Serum cystatin C as a reliable marker of changes in glomerular filtration rate in children with urinary tract malformations". The Journal of Urology. 175 (1): 303–9. doi:10.1016/S0022-5347(05)00015-7. PMID 16406933.
- ^ Onopiuk A, Tokarzewicz A, Gorodkiewicz E (2015). "Cystatin C: a kidney function biomarker". Adv Clin Chem. 68: 57–69. doi:10.1016/bs.acc.2014.11.007. PMID 25858868.
- ^ Ferguson TW, Komenda P, Tangri N (May 2015). "Cystatin C as a biomarker for estimating glomerular filtration rate". Curr Opin Nephrol Hypertens. 24 (3): 295–300. doi:10.1097/MNH.0000000000000115. PMID 26066476. S2CID 27385.
- ^ Porto JR, Gomes KB, Fernandes AP, Domingueti CP (January 1, 2017). "Cystatin C: a promising biomarker to evaluate renal function". Revista Brasileira de Analises Clinicas. 49 (3). doi:10.21877/2448-3877.201600446. Retrieved 18 November 2020.
- ^ Shlipak MG, Katz R, Sarnak MJ, Fried LF, Newman AB, Stehman-Breen C, et al. (August 2006). "Cystatin C and prognosis for cardiovascular and kidney outcomes in elderly persons without chronic kidney disease". Annals of Internal Medicine. 145 (4): 237–46. doi:10.7326/0003-4819-145-4-200608150-00003. PMID 16908914. S2CID 24679098.
- ^ Justice JN, Ferrucci L, Newman AB, Aroda VR, Bahnson JL, Divers J, Espeland MA, Marcovina S, Pollak MN, Kritchevsky SB, Barzilai N, Kuchel GA (December 2018). "A framework for selection of blood-based biomarkers for geroscience-guided clinical trials: report from the TAME Biomarkers Workgroup". Geroscience. 40 (5–6): 419–436. doi:10.1007/s11357-018-0042-y. PMC 6294728. PMID 30151729.
- ^ "Chronic kidney disease in adults: assessment and management". National Institute for Health and Care Excellence (NICE). 23 July 2014. Retrieved 18 November 2020.
- ^ Hermida J, Tutor JC (June 2006). "Serum cystatin C for the prediction of glomerular filtration rate with regard to the dose adjustment of amikacin, gentamicin, tobramycin, and vancomycin". Therapeutic Drug Monitoring. 28 (3): 326–31. doi:10.1097/01.ftd.0000211805.89440.3d. PMID 16778715. S2CID 27219789.
- ^ Schück O, Teplan V, Sibová J, Stollová M (February 2004). "Predicting the glomerular filtration rate from serum creatinine, serum cystatin C and the Cockcroft and Gault formula with regard to drug dosage adjustment". International Journal of Clinical Pharmacology and Therapeutics. 42 (2): 93–7. doi:10.5414/cpp42093. PMID 15180169.
- ^ Demirtaş S, Akan O, Can M, Elmali E, Akan H (February 2006). "Cystatin C can be affected by nonrenal factors: a preliminary study on leukemia". Clinical Biochemistry. 39 (2): 115–8. doi:10.1016/j.clinbiochem.2005.10.009. PMID 16337174.
- ^ Nakai K, Kikuchi M, Fujimoto K, Kaneko Y, Omori S, Nakai K, Suwabe A (April 2008). "Serum levels of cystatin C in patients with malignancy". Clinical and Experimental Nephrology. 12 (2): 132–139. doi:10.1007/s10157-008-0043-8. PMID 18317874. S2CID 19326312.
- ^ Kos J, Stabuc B, Cimerman N, Brünner N (December 1998). "Serum cystatin C, a new marker of glomerular filtration rate, is increased during malignant progression". Clinical Chemistry. 44 (12): 2556–7. doi:10.1093/clinchem/44.12.2556. PMID 9836733.
- ^ Fricker M, Wiesli P, Brändle M, Schwegler B, Schmid C (May 2003). "Impact of thyroid dysfunction on serum cystatin C". Kidney International. 63 (5): 1944–7. doi:10.1046/j.1523-1755.2003.00925.x. PMID 12675875.
- ^ Manetti L, Pardini E, Genovesi M, Campomori A, Grasso L, Morselli LL, et al. (April 2005). "Thyroid function differently affects serum cystatin C and creatinine concentrations". Journal of Endocrinological Investigation. 28 (4): 346–9. doi:10.1007/bf03347201. PMID 15966508. S2CID 26777752. Archived from the original on 2008-06-08.
- ^ Wiesli P, Schwegler B, Spinas GA, Schmid C (December 2003). "Serum cystatin C is sensitive to small changes in thyroid function". Clinica Chimica Acta; International Journal of Clinical Chemistry. 338 (1–2): 87–90. doi:10.1016/j.cccn.2003.07.022. PMID 14637271.
- ^ Risch L, Herklotz R, Blumberg A, Huber AR (November 2001). "Effects of glucocorticoid immunosuppression on serum cystatin C concentrations in renal transplant patients". Clinical Chemistry. 47 (11): 2055–9. doi:10.1093/clinchem/47.11.2055. PMID 11673383.
- ^ Cimerman N, Brguljan PM, Krasovec M, Suskovic S, Kos J (October 2000). "Serum cystatin C, a potent inhibitor of cysteine proteinases, is elevated in asthmatic patients". Clinica Chimica Acta; International Journal of Clinical Chemistry. 300 (1–2): 83–95. doi:10.1016/S0009-8981(00)00298-9. PMID 10958865.
- ^ Bökenkamp A, van Wijk JA, Lentze MJ, Stoffel-Wagner B (July 2002). "Effect of corticosteroid therapy on serum cystatin C and beta2-microglobulin concentrations". Clinical Chemistry. 48 (7): 1123–6. doi:10.1093/clinchem/48.7.1123. PMID 12089191.
- ^ Knight EL, Verhave JC, Spiegelman D, Hillege HL, de Zeeuw D, Curhan GC, de Jong PE (April 2004). "Factors influencing serum cystatin C levels other than renal function and the impact on renal function measurement". Kidney International. 65 (4): 1416–21. doi:10.1111/j.1523-1755.2004.00517.x. PMID 15086483.
- ^ Odden MC, Scherzer R, Bacchetti P, Szczech LA, Sidney S, Grunfeld C, Shlipak MG (November 2007). "Cystatin C level as a marker of kidney function in human immunodeficiency virus infection: the FRAM study". Archives of Internal Medicine. 167 (20): 2213–9. doi:10.1001/archinte.167.20.2213. PMC 3189482. PMID 17998494. Archived from the original on 2008-06-07.
- ^ Collé A, Tavera C, Prévot D, Leung-Tack J, Thomas Y, Manuel Y, et al. (1992). "Cystatin C levels in sera of patients with human immunodeficiency virus infection. A new avidin-biotin ELISA assay for its measurement". Journal of Immunoassay. 13 (1): 47–60. doi:10.1080/15321819208019824. PMID 1569212.
- ^ Jaroszewicz J, Wiercinska-Drapalo A, Lapinski TW, Prokopowicz D, Rogalska M, Parfieniuk A (2006). "Does HAART improve renal function? An association between serum cystatin C concentration, HIV viral load and HAART duration". Antiviral Therapy. 11 (5): 641–5. doi:10.1177/135965350601100502. PMID 16964834. S2CID 33722022.
- ^ Strevens H, Wide-Swensson D, Torffvit O, Grubb A (2002). "Serum cystatin C for assessment of glomerular filtration rate in pregnant and non-pregnant women. Indications of altered filtration process in pregnancy". Scandinavian Journal of Clinical and Laboratory Investigation. 62 (2): 141–7. doi:10.1080/003655102753611771. PMID 12004930. S2CID 25382382.
- ^ Akbari A, Lepage N, Keely E, Clark HD, Jaffey J, MacKinnon M, Filler G (May 2005). "Cystatin-C and beta trace protein as markers of renal function in pregnancy". BJOG. 112 (5): 575–8. doi:10.1111/j.1471-0528.2004.00492.x. PMID 15842279. S2CID 1851149.
- ^ Sjöström P, Tidman M, Jones I (2005). "Determination of the production rate and non-renal clearance of cystatin C and estimation of the glomerular filtration rate from the serum concentration of cystatin C in humans". Scandinavian Journal of Clinical and Laboratory Investigation. 65 (2): 111–24. doi:10.1080/00365510510013523. PMID 16025834. S2CID 41336736.
- ^ Tonelli M, Wiebe N, Culleton B, House A, Rabbat C, Fok M, et al. (July 2006). "Chronic kidney disease and mortality risk: a systematic review". Journal of the American Society of Nephrology. 17 (7): 2034–47. doi:10.1681/ASN.2005101085. PMID 16738019.
- ^ Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY (September 2004). "Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization". The New England Journal of Medicine. 351 (13): 1296–305. doi:10.1056/NEJMoa041031. PMID 15385656.
- ^ Zethelius B, Berglund L, Sundström J, Ingelsson E, Basu S, Larsson A, et al. (May 2008). "Use of multiple biomarkers to improve the prediction of death from cardiovascular causes". The New England Journal of Medicine. 358 (20): 2107–16. doi:10.1056/NEJMoa0707064. PMID 18480203.
- ^ Shlipak MG, Sarnak MJ, Katz R, Fried LF, Seliger SL, Newman AB, et al. (May 2005). "Cystatin C and the risk of death and cardiovascular events among elderly persons". The New England Journal of Medicine. 352 (20): 2049–60. doi:10.1056/NEJMoa043161. PMID 15901858.
- ^ Ix JH, Shlipak MG, Chertow GM, Whooley MA (January 2007). "Association of cystatin C with mortality, cardiovascular events, and incident heart failure among persons with coronary heart disease: data from the Heart and Soul Study". Circulation. 115 (2): 173–9. doi:10.1161/CIRCULATIONAHA.106.644286. PMC 2771187. PMID 17190862.
- ^ Deo R, Fyr CL, Fried LF, Newman AB, Harris TB, Angleman S, et al. (January 2008). "Kidney dysfunction and fatal cardiovascular disease--an association independent of atherosclerotic events: results from the Health, Aging, and Body Composition (Health ABC) study". American Heart Journal. 155 (1): 62–8. doi:10.1016/j.ahj.2007.08.012. PMID 18082491.
- ^ Koenig W, Twardella D, Brenner H, Rothenbacher D (February 2005). "Plasma concentrations of cystatin C in patients with coronary heart disease and risk for secondary cardiovascular events: more than simply a marker of glomerular filtration rate". Clinical Chemistry. 51 (2): 321–7. doi:10.1373/clinchem.2004.041889. PMID 15563478.
- ^ Jernberg T, Lindahl B, James S, Larsson A, Hansson LO, Wallentin L (October 2004). "Cystatin C: a novel predictor of outcome in suspected or confirmed non-ST-elevation acute coronary syndrome". Circulation. 110 (16): 2342–8. doi:10.1161/01.CIR.0000145166.44942.E0. PMID 15477399.
- ^ Luc G, Bard JM, Lesueur C, Arveiler D, Evans A, Amouyel P, et al. (April 2006). "Plasma cystatin-C and development of coronary heart disease: The PRIME Study" (PDF). Atherosclerosis. 185 (2): 375–80. doi:10.1016/j.atherosclerosis.2005.06.017. PMID 16046222. S2CID 40221090.
- ^ Servais A, Giral P, Bernard M, Bruckert E, Deray G, Isnard Bagnis C (May 2008). "Is serum cystatin-C a reliable marker for metabolic syndrome?". The American Journal of Medicine. 121 (5): 426–32. doi:10.1016/j.amjmed.2008.01.040. PMID 18456039.
- ^ Menon V, Shlipak MG, Wang X, Coresh J, Greene T, Stevens L, et al. (July 2007). "Cystatin C as a risk factor for outcomes in chronic kidney disease". Annals of Internal Medicine. 147 (1): 19–27. doi:10.7326/0003-4819-147-1-200707030-00004. PMID 17606957. S2CID 31964826.
- ^ Sarnak MJ, Katz R, Fried LF, Siscovick D, Kestenbaum B, Seliger S, et al. (January 2008). "Cystatin C and aging success". Archives of Internal Medicine. 168 (2): 147–53. doi:10.1001/archinternmed.2007.40. PMC 2871318. PMID 18227360. Archived from the original on 2008-06-07.
- ^ Djoussé L, Kurth T, Gaziano JM (January 2008). "Cystatin C and risk of heart failure in the Physicians' Health Study (PHS)". American Heart Journal. 155 (1): 82–6. doi:10.1016/j.ahj.2007.08.023. PMC 2179893. PMID 18082494.
- ^ O'Hare AM, Newman AB, Katz R, Fried LF, Stehman-Breen CO, Seliger SL, et al. (2005). "Cystatin C and incident peripheral arterial disease events in the elderly: results from the Cardiovascular Health Study". Archives of Internal Medicine. 165 (22): 2666–70. doi:10.1001/archinte.165.22.2666. PMID 16344426.
- ^ Stevens LA, Levey AS (May 2005). "Chronic kidney disease in the elderly--how to assess risk". The New England Journal of Medicine. 352 (20): 2122–4. doi:10.1056/NEJMe058035. PMID 15901867.
- ^ Delanaye P, Cavalier E, Krzesinski JM (February 2008). "Cystatin C, renal function, and cardiovascular risk". Annals of Internal Medicine. 148 (4): 323. doi:10.7326/0003-4819-148-4-200802190-00023. hdl:2268/4787. PMID 18283218.
- ^ [non-primary source needed] Norden AG, Lapsley M, Lee PJ, Pusey CD, Scheinman SJ, Tam FW, et al. (November 2001). "Glomerular protein sieving and implications for renal failure in Fanconi syndrome". Kidney International. 60 (5): 1885–92. doi:10.1046/j.1523-1755.2001.00016.x. PMID 11703607.
- ^ [non-primary source needed] Tenstad O, Roald AB, Grubb A, Aukland K (August 1996). "Renal handling of radiolabelled human cystatin C in the rat". Scandinavian Journal of Clinical and Laboratory Investigation. 56 (5): 409–14. doi:10.3109/00365519609088795. PMID 8869663.
- ^ [non-primary source needed] Grubb A, Lindström V, Jonsson M, Bäck SE, Åhlund T, Rippe B, Christensson A (July 2015). "Reduction in glomerular pore size is not restricted to pregnant women. Evidence for a new syndrome: 'Shrunken pore syndrome'". Scandinavian Journal of Clinical and Laboratory Investigation. 75 (4): 333–40. doi:10.3109/00365513.2015.1025427. PMC 4487590. PMID 25919022.
- ^ Grubb A (June 2020). "Shrunken pore syndrome - a common kidney disorder with high mortality. Diagnosis, prevalence, pathophysiology and treatment options". Clinical Biochemistry. Online ahead of print: 12–20. doi:10.1016/j.clinbiochem.2020.06.002. PMID 32544475.
- ^ Levy E, Lopez-Otin C, Ghiso J, Geltner D, Frangione B (May 1989). "Stroke in Icelandic patients with hereditary amyloid angiopathy is related to a mutation in the cystatin C gene, an inhibitor of cysteine proteases". The Journal of Experimental Medicine. 169 (5): 1771–8. doi:10.1084/jem.169.5.1771. PMC 2189307. PMID 2541223.
- ^ Levy E, Jaskolski M, Grubb A (January 2006). "The role of cystatin C in cerebral amyloid angiopathy and stroke: cell biology and animal models". Brain Pathology. 16 (1): 60–70. doi:10.1111/j.1750-3639.2006.tb00562.x. PMC 8095742. PMID 16612983. S2CID 21279341.
- ^ Janowski R, Kozak M, Janowska E, Grzonka Z, Grubb A, Abrahamson M, et al. (April 2001). "Human cystatin C, an amyloidogenic protein, dimerizes through three-dimensional domain swapping". Nature Structural Biology. 8 (4): 316–320. doi:10.1038/86188. PMID 11276250. S2CID 28916747.
- ^ Janowski R, Kozak M, Abrahamson M, Grubb A, Jaskolski M (Nov 2005). "Glomerular protein sieving and implications for renal failure in Fanconi syndrome". Proteins. 61 (3): 570–578–92. doi:10.1002/prot.20633. PMID 16170782. S2CID 633348.
- ^ Chrabaszczewska M, Sieradzan AK, Rodziewicz-Motowidło S, Grubb A, Dobson CM, Kunita JR, Kozak M (Aug 2020). "Structural characterization of covalently stabilized human cystatin C oligomers". International Journal of Molecular Sciences. 21 (5860): 5860. doi:10.3390/ijms21165860. PMC 7461555. PMID 32824145.
- ^ Mi W, Pawlik M, Sastre M, Jung SS, Radvinsky DS, Klein AM, et al. (December 2007). "Cystatin C inhibits amyloid-beta deposition in Alzheimer's disease mouse models". Nature Genetics. 39 (12): 1440–2. doi:10.1038/ng.2007.29. PMID 18026100. S2CID 21332698.
- ^ Kaeser SA, Herzig MC, Coomaraswamy J, Kilger E, Selenica ML, Winkler DT, et al. (December 2007). "Cystatin C modulates cerebral beta-amyloidosis". Nature Genetics. 39 (12): 1437–9. doi:10.1038/ng.2007.23. PMID 18026102. S2CID 7001526.
- ^ Bertram L, McQueen MB, Mullin K, Blacker D, Tanzi RE (January 2007). "Systematic meta-analyses of Alzheimer disease genetic association studies: the AlzGene database". Nature Genetics. 39 (1): 17–23. doi:10.1038/ng1934. PMID 17192785. S2CID 452851.
- ^ Chuo LJ, Sheu WH, Pai MC, Kuo YM (2007). "Genotype and plasma concentration of cystatin C in patients with late-onset Alzheimer disease". Dementia and Geriatric Cognitive Disorders. 23 (4): 251–7. doi:10.1159/000100021. PMID 17310123. S2CID 29689590.
- ^ Del Boccio P, Pieragostino D, Lugaresi A, Di Ioia M, Pavone B, Travaglini D, et al. (August 2007). "Cleavage of cystatin C is not associated with multiple sclerosis". Annals of Neurology. 62 (2): 201–4, discussion 205. doi:10.1002/ana.20968. PMID 17006926. S2CID 10687960.
- ^ Shi GP, Sukhova GK, Grubb A, Ducharme A, Rhode LH, Lee RT, et al. (November 1999). "Cystatin C deficiency in human atherosclerosis and aortic aneurysms" (PDF). The Journal of Clinical Investigation. 104 (9): 1191–7. doi:10.1172/JCI7709. PMC 409823. PMID 10545518.
- ^ Abisi S, Burnand KG, Waltham M, Humphries J, Taylor PR, Smith A (December 2007). "Cysteine protease activity in the wall of abdominal aortic aneurysms". Journal of Vascular Surgery. 46 (6): 1260–6. doi:10.1016/j.jvs.2007.08.015. PMID 18155003.
- ^ Abdul-Hussien H, Soekhoe RG, Weber E, von der Thüsen JH, Kleemann R, Mulder A, et al. (March 2007). "Collagen degradation in the abdominal aneurysm: a conspiracy of matrix metalloproteinase and cysteine collagenases". The American Journal of Pathology. 170 (3): 809–17. doi:10.2353/ajpath.2007.060522. PMC 1864891. PMID 17322367.
- ^ Gacko M, Chyczewski L, Chrostek L (1999). "Distribution, activity and concentration of cathepsin B and cystatin C in the wall of aortic aneurysm". Polish Journal of Pathology. 50 (2): 83–6. PMID 10481531.
- ^ Eriksson P, Jones KG, Brown LC, Greenhalgh RM, Hamsten A, Powell JT (January 2004). "Genetic approach to the role of cysteine proteases in the expansion of abdominal aortic aneurysms". The British Journal of Surgery. 91 (1): 86–9. doi:10.1002/bjs.4364. PMC 11439999. PMID 14716800. S2CID 45363107.
- ^ Lindholt JS, Erlandsen EJ, Henneberg EW (November 2001). "Cystatin C deficiency is associated with the progression of small abdominal aortic aneurysms". The British Journal of Surgery. 88 (11): 1472–5. doi:10.1046/j.0007-1323.2001.01911.x. PMID 11683743. S2CID 42364814.
- ^ Zurdel J, Finckh U, Menzer G, Nitsch RM, Richard G (February 2002). "CST3 genotype associated with exudative age related macular degeneration". The British Journal of Ophthalmology. 86 (2): 214–9. doi:10.1136/bjo.86.2.214. PMC 1771004. PMID 11815350.
- ^ Im E, Kazlauskas A (March 2007). "The role of cathepsins in ocular physiology and pathology". Experimental Eye Research. 84 (3): 383–8. doi:10.1016/j.exer.2006.05.017. PMID 16893541.
- ^ Strojan P, Oblak I, Svetic B, Smid L, Kos J (May 2004). "Cysteine proteinase inhibitor cystatin C in squamous cell carcinoma of the head and neck: relation to prognosis". British Journal of Cancer. 90 (10): 1961–8. doi:10.1038/sj.bjc.6601830. PMC 2409457. PMID 15138478.
- ^ Kos J, Krasovec M, Cimerman N, Nielsen HJ, Christensen IJ, Brünner N (February 2000). "Cysteine proteinase inhibitors stefin A, stefin B, and cystatin C in sera from patients with colorectal cancer: relation to prognosis". Clinical Cancer Research. 6 (2): 505–11. PMID 10690531.
- ^ Strevens H, Wide-Swensson D, Grubb A, Hansen A, Horn T, Ingemarsson I, et al. (September 2003). "Serum cystatin C reflects glomerular endotheliosis in normal, hypertensive and pre-eclamptic pregnancies". BJOG. 110 (9): 825–30. doi:10.1111/j.1471-0528.2003.02051.x. PMID 14511964. S2CID 26686328.
- ^ Franceschini N, Qiu C, Barrow DA, Williams MA (2008). "Cystatin C and preeclampsia: a case control study". Renal Failure. 30 (1): 89–95. doi:10.1080/08860220701742229. PMID 18197549. S2CID 205592849.
- ^ Kristensen K, Wide-Swensson D, Schmidt C, Blirup-Jensen S, Lindström V, Strevens H, Grubb A (2007). "Cystatin C, beta-2-microglobulin and beta-trace protein in pre-eclampsia". Acta Obstetricia et Gynecologica Scandinavica. 86 (8): 921–6. doi:10.1080/00016340701318133. PMID 17653875. S2CID 45055267.
- ^ Kristensen K, Larsson I, Hansson SR (March 2007). "Increased cystatin C expression in the pre-eclamptic placenta". Molecular Human Reproduction. 13 (3): 189–95. doi:10.1093/molehr/gal111. PMID 17227816.
- ^ a b Croda-Todd MT, Soto-Montano XJ, Hernández-Cancino PA, Juárez-Aguilar E (September 2007). "Adult cystatin C reference intervals determined by nephelometric immunoassay". Clinical Biochemistry. 40 (13–14): 1084–7. doi:10.1016/j.clinbiochem.2007.05.011. PMID 17624320.
- ^ Lamb EJ, O'Riordan SE, Webb MC, Newman DJ (November 2003). "Serum cystatin C may be a better marker of renal impairment than creatinine". Journal of the American Geriatrics Society. 51 (11): 1674, author reply 1674-5. doi:10.1046/j.1532-5415.2003.515244.x. PMID 14687406. S2CID 34235014.
- ^ Peake M, Whiting M (November 2006). "Measurement of serum creatinine--current status and future goals". The Clinical Biochemist. Reviews. 27 (4): 173–84. PMC 1784008. PMID 17581641.
- ^ Myers GL, Miller WG, Coresh J, Fleming J, Greenberg N, Greene T, et al. (January 2006). "Recommendations for improving serum creatinine measurement: a report from the Laboratory Working Group of the National Kidney Disease Education Program". Clinical Chemistry. 52 (1): 5–18. doi:10.1373/clinchem.2005.0525144. PMID 16332993.
- ^ a b c Köttgen A, Selvin E, Stevens LA, Levey AS, Van Lente F, Coresh J (March 2008). "Serum cystatin C in the United States: the Third National Health and Nutrition Examination Survey (NHANES III)". American Journal of Kidney Diseases. 51 (3): 385–94. doi:10.1053/j.ajkd.2007.11.019. PMID 18295054.
- ^ a b Finney H, Newman DJ, Thakkar H, Fell JM, Price CP (January 2000). "Reference ranges for plasma cystatin C and creatinine measurements in premature infants, neonates, and older children". Archives of Disease in Childhood. 82 (1): 71–5. doi:10.1136/adc.82.1.71. PMC 1718178. PMID 10630919.
- ^ Ognibene A, Mannucci E, Caldini A, Terreni A, Brogi M, Bardini G, et al. (June 2006). "Cystatin C reference values and aging". Clinical Biochemistry. 39 (6): 658–61. doi:10.1016/j.clinbiochem.2006.03.017. PMID 16730690.
- ^ Filler G, Bökenkamp A, Hofmann W, Le Bricon T, Martínez-Brú C, Grubb A (January 2005). "Cystatin C as a marker of GFR--history, indications, and future research". Clinical Biochemistry. 38 (1): 1–8. doi:10.1016/j.clinbiochem.2004.09.025. PMID 15607309.
- ^ Stevens LA, Coresh J, Schmid CH, Feldman HI, Froissart M, Kusek J, et al. (March 2008). "Estimating GFR using serum cystatin C alone and in combination with serum creatinine: a pooled analysis of 3,418 individuals with CKD". American Journal of Kidney Diseases. 51 (3): 395–406. doi:10.1053/j.ajkd.2007.11.018. PMC 2390827. PMID 18295055.
- ^ Shlipak MG (March 2008). "Cystatin C: research priorities targeted to clinical decision making". American Journal of Kidney Diseases. 51 (3): 358–61. doi:10.1053/j.ajkd.2008.01.002. PMID 18295049.
- ^ Muntner P, Winston J, Uribarri J, Mann D, Fox CS (April 2008). "Overweight, obesity, and elevated serum cystatin C levels in adults in the United States". The American Journal of Medicine. 121 (4): 341–8. doi:10.1016/j.amjmed.2008.01.003. PMC 3049932. PMID 18374694.
- ^ "Entrez Gene: CST3 cystatin C (amyloid angiopathy and cerebral hemorrhage)".
- ^ Hwang SJ, Yang Q, Meigs JB, Pearce EN, Fox CS (September 2007). "A genome-wide association for kidney function and endocrine-related traits in the NHLBI's Framingham Heart Study". BMC Medical Genetics. 8 (Suppl 1) S10. doi:10.1186/1471-2350-8-S1-S10. PMC 1995611. PMID 17903292.
- ^ Janowski R, Kozak M, Jankowska E, Grzonka Z, Grubb A, Abrahamson M, Jaskolski M (April 2001). "Human cystatin C, an amyloidogenic protein, dimerizes through three-dimensional domain swapping" (PDF). Nature Structural Biology. 8 (4): 316–20. doi:10.1038/86188. PMID 11276250. S2CID 28916747.
- ^ a b Grubb A, Löfberg H (May 1982). "Human gamma-trace, a basic microprotein: amino acid sequence and presence in the adenohypophysis". Proceedings of the National Academy of Sciences of the United States of America. 79 (9): 3024–7. Bibcode:1982PNAS...79.3024G. doi:10.1073/pnas.79.9.3024. PMC 346341. PMID 6283552.
- ^ Löfberg H, Grubb AO (November 1979). "Quantitation of gamma-trace in human biological fluids: indications for production in the central nervous system". Scandinavian Journal of Clinical and Laboratory Investigation. 39 (7): 619–26. doi:10.3109/00365517909108866. PMID 119302.
- ^ Grubb A, Simonsen O, Sturfelt G, Truedsson L, Thysell H (1985). "Serum concentration of cystatin C, factor D and beta 2-microglobulin as a measure of glomerular filtration rate". Acta Medica Scandinavica. 218 (5): 499–503. doi:10.1111/j.0954-6820.1985.tb08880.x. PMID 3911736.
- ^ Simonsen O, Grubb A, Thysell H (April 1985). "The blood serum concentration of cystatin C (gamma-trace) as a measure of the glomerular filtration rate". Scandinavian Journal of Clinical and Laboratory Investigation. 45 (2): 97–101. doi:10.3109/00365518509160980. PMID 3923607.
- ^ Shlipak MG, Matsushita K, Ärnlöv J, Inker LA, Katz R, Polkinghorne KR, et al. (September 2013). "Cystatin C versus creatinine in determining risk based on kidney function". The New England Journal of Medicine. 369 (10): 932–43. doi:10.1056/NEJMoa1214234. PMC 3993094. PMID 24004120.