Recent from talks
Nothing was collected or created yet.
Nasolacrimal duct
View on Wikipedia| Nasolacrimal duct | |
|---|---|
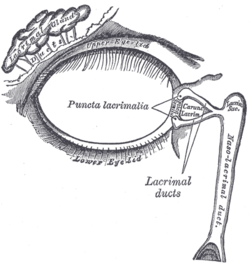
 The lacrimal apparatus. Right side. | |
| Details | |
| Identifiers | |
| Latin | ductus nasolacrimalis |
| MeSH | D009301 |
| TA98 | A15.2.07.070 |
| TA2 | 6859 |
| FMA | 9703 |
| Anatomical terminology | |
The nasolacrimal duct (also called the tear duct) carries tears from the lacrimal sac of the eye into the nasal cavity.[1][2] The duct begins in the eye socket between the maxillary and lacrimal bones, from where it passes downwards and backwards. The opening of the nasolacrimal duct into the inferior nasal meatus of the nasal cavity is partially covered by a mucosal fold (valve of Hasner or plica lacrimalis).[3]
Excess tears flow through the nasolacrimal duct which drains into the inferior nasal meatus. This is the reason the nose starts to run when a person is crying or has watery eyes from an allergy, and why one can sometimes taste eye drops. This is for the same reason when applying some eye drops it is often advised to close the nasolacrimal duct by pressing it with a finger to prevent the medicine from escaping the eye and having unwanted side effects elsewhere in the body as it will proceed through the canal to the nasal cavity.
Like the lacrimal sac, the duct is lined by stratified columnar epithelium containing mucus-secreting goblet cells, and is surrounded by connective tissue.
Nasolacrimal canal
[edit]| Nasolacrimal canal | |
|---|---|
 Tear system. a = lacrimal gland b = superior lacrimal punctum c = superior lacrimal canal d = lacrimal sac e = inferior lacrimal punctum f = inferior lacrimal canal g = nasolacrimal canal | |
| Details | |
| Identifiers | |
| Latin | canalis nasolacrimalis |
| MeSH | D009301 |
| TA98 | A15.2.07.070 |
| TA2 | 6859 |
| FMA | 9703 |
| Anatomical terms of bone | |
The canal containing the duct is called the nasolacrimal canal. It is formed by indentations in the inferior nasal conchae, maxilla and lacrimal bone. The canal drains into the nasal cavity through the anterior portion of the inferior meatus, which is between the inferior concha and the floor of the nasal cavity.
Clinical significance
[edit]Obstruction of the nasolacrimal duct may occur.[4][5][6] This leads to the excess overflow of tears called epiphora (chronic low-grade nasolacrimal duct occlusion).[7] A congenital obstruction can cause cystic expansion of the duct and is called a dacryocystocele or Timo cyst. Persons with dry eye conditions can be fitted with punctal plugs that seal the ducts to limit the amount of fluid drainage and retain moisture.
During an ear infection, excess mucus may drain through the nasolacrimal duct in the opposite way tears drain. [citation needed]
In humans, the tear ducts in males tend to be larger than the ones in females.[8]
Additional image
[edit]-
Roof, floor, and lateral wall of left nasal cavity
-
The seven bones which articulate to form the orbit.
-
Outline of bones of face, showing position of air sinuses.
See also
[edit]References
[edit]- ^ "Nasolacrimal System Anatomy: Embryology, Puncta, Canaliculi". 2020-02-19.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Herbert, Ronald A.; Janardhan, Kyathanahalli S.; Pandiri, Arun R.; Cesta, Mark F.; Miller, Rodney A. (2018). "Nose, Larynx, and Trachea". Boorman's Pathology of the Rat. Elsevier. pp. 391–435. doi:10.1016/b978-0-12-391448-4.00022-8. ISBN 978-0-12-391448-4. S2CID 80517730.
The paired nasolacrimal ducts carry lacrimal secretions from the eye to the nasal cavity, and originate as oval openings near the edge of the medial canthus of the eyelids. Initially the duct is small and circular, but in the middle portion, the diameter increases and the appearance is more oblong and saccular. The diameter again decreases before the duct enters the ventrolateral nasal vestibule medial to the root of the incisor tooth approximately 2 mm caudal to the nares.
- ^ "Medical Definition of PLICA LACRIMALIS". Merriam-Webster Medical Dictionary. 2020-05-08. Retrieved 2020-05-08.
an imperfect valve at the opening of the nasolacrimal duct into the inferior meatus of the nose
- ^ CARREIRO, J (2009). "Head and neck". Pediatric Manual Medicine. Elsevier. pp. 13–97. doi:10.1016/b978-0-443-10308-7.00002-8. ISBN 978-0-443-10308-7.
Dacryostenosis is a condition whereby the nasolacrimal duct is narrowed or blocked and the glandular secretions into the eye are prevented from draining properly. The secretions collect around the orifice of the duct and in the corner of the eye, where they thicken, resulting in a gooey, sticky substance that further clogs any drainage route.
- ^ Stagner, Anna M.; Jakobiec, Frederick A.; Eagle, Ralph C.; Charles, Norman C. (2018). "Infections of the Eye and Its Adnexa". Diagnostic Pathology of Infectious Disease. Elsevier. pp. 648–685. doi:10.1016/b978-0-323-44585-6.00021-7. ISBN 978-0-323-44585-6.
Dacryocystitis is produced by obstruction of the nasolacrimal duct with resultant tear stasis. Clinically, patients display erythema and swelling of the lacrimal sac, creating a mass in the medial canthal area, centered below the medial canthal tendon.
- ^ "Blocked tear ducts in infants" Archived 2012-07-08 at the Wayback Machine, Pediatric Views, June 2006.
- ^ Nerad, Jeffrey A.; Carter, Keith D.; Alford, Mark (2008). "Disorders of the Lacrimal System: Congenital Obstruction". Oculoplastic and Reconstructive Surgery. Elsevier. pp. 131–137. doi:10.1016/b978-0-323-05386-0.50010-7. ISBN 978-0-323-05386-0.
- ^ "Tears of Men and Women Are Different", Wall Street Journal, mirrored at "Why Men and Women Shed Different Tears", Fox News, May 5, 2011.
External links
[edit]- Anatomy figure: 33:04-09 at Human Anatomy Online, SUNY Downstate Medical Center
- Anatomy photo:29:os-0510 at the SUNY Downstate Medical Center - "Orbits and Eye: Orbital openings and related anatomical features"
- lesson9 at The Anatomy Lesson by Wesley Norman (Georgetown University)