Recent from talks
Nothing was collected or created yet.
Thyroid disease
View on Wikipedia| Thyroid disease | |
|---|---|
 | |
| An illustration of goiter, a type of thyroid disease | |
| Specialty | Endocrinology, medical genetics |
Thyroid disease is a medical condition that affects the structure and/or function of the thyroid gland. The thyroid gland is located at the front of the neck and produces thyroid hormones[1] that travel through the blood to help regulate many other organs, meaning that it is an endocrine organ. These hormones normally act in the body to regulate energy use, infant development, and childhood development.[2]
There are five general types of thyroid disease, each with their own symptoms. A person may have one or several different types at the same time. The five groups are:
- Hypothyroidism (low function) caused by not having enough free thyroid hormones[2]
- Hyperthyroidism (high function) caused by having too many free thyroid hormones[2]
- Structural abnormalities, most commonly a goiter (enlargement of the thyroid gland)[2]
- Tumors which can be benign (not cancerous) or cancerous[2]
- Abnormal thyroid function tests without any clinical symptoms (subclinical hypothyroidism or subclinical hyperthyroidism).[2]
In the US, hypothyroidism and hyperthyroidism were respectively found in 4.6 and 1.3% of the >12y old population (2002).[3]
In some types, such as subacute thyroiditis or postpartum thyroiditis, symptoms may go away after a few months and laboratory tests may return to normal.[4] However, most types of thyroid disease do not resolve on their own. Common hypothyroid symptoms include fatigue, low energy, weight gain, inability to tolerate the cold, slow heart rate, dry skin and constipation.[5] Common hyperthyroid symptoms include irritability, anxiety, weight loss, fast heartbeat, inability to tolerate the heat, diarrhea, and enlargement of the thyroid.[6] Structural abnormalities may not produce symptoms; however, some people may have hyperthyroid or hypothyroid symptoms related to the structural abnormality or notice swelling of the neck.[7] Rarely goiters can cause compression of the airway, compression of the vessels in the neck, or difficulty swallowing.[7] Tumors, often called thyroid nodules, can also have many different symptoms ranging from hyperthyroidism to hypothyroidism to swelling in the neck and compression of the structures in the neck.[7]
Diagnosis starts with a history and physical examination. Screening for thyroid disease in patients without symptoms is a debated topic although commonly practiced in the United States.[8] If dysfunction of the thyroid is suspected, laboratory tests can help support or rule out thyroid disease. Initial blood tests often include thyroid-stimulating hormone (TSH) and free thyroxine (T4).[9] Total and free triiodothyronine (T3) levels are less commonly used.[9] If autoimmune disease of the thyroid is suspected, blood tests looking for Anti-thyroid autoantibodies can also be obtained. Procedures such as ultrasound, biopsy and a radioiodine scanning and uptake study may also be used to help with the diagnosis, particularly if a nodule is suspected.[2]
Thyroid diseases are highly prevalent worldwide,[10][11][12] and treatment varies based on the disorder. Levothyroxine is the mainstay of treatment for people with hypothyroidism,[13] while people with hyperthyroidism caused by Graves' disease can be managed with iodine therapy, antithyroid medication, or surgical removal of the thyroid gland.[14] Thyroid surgery may also be performed to remove a thyroid nodule or to reduce the size of a goiter if it obstructs nearby structures or for cosmetic reasons.[14]
Signs and symptoms
[edit]Symptoms of the condition vary with type: hypo- vs. hyperthyroidism, which are further described below.
Possible symptoms of hypothyroidism are:[15][16]
- Tiredness
- Unexplained weight gain
- Slow movement
- Muscle cramps
- Slow heart rate (bradycardia)
- Sensitivity to cold temperatures
- Constipation
- Depressed mood
- Memory difficulty
Possible symptoms of hyperthyroidism are:[17]
- Difficulty sleeping (insomnia)
- Unexplained weight loss
- Tremors
- Fast heart rate (tachycardia) or palpitations
- Sensitivity to hot temperatures, excess sweating
- Diarrhea
- Anxiety, irritability
Note: certain symptoms and physical changes can be seen in both hypothyroidism and hyperthyroidism —fatigue, fine / thinning hair, menstrual cycle irregularities, muscle weakness / aches (myalgia), and different forms of myxedema.[18][19]
Diseases
[edit]Low function
[edit]Hypothyroidism is a state in which the body is not producing enough thyroid hormones, or is not able to respond to / utilize existing thyroid hormones properly. The main categories are:
- Thyroiditis: an inflammation of the thyroid gland
- Hashimoto's thyroiditis / Hashimoto's disease
- Ord's thyroiditis
- Postpartum thyroiditis
- Silent thyroiditis
- Acute thyroiditis
- Riedel's thyroiditis (the majority of cases do not affect thyroid function, but approximately 30% of cases lead to hypothyroidism)
- Iatrogenic hypothyroidism[20]
- Postoperative hypothyroidism
- Medication- or radiation-induced hypothyroidism
- Thyroid hormone resistance
- Euthyroid sick syndrome
- Congenital hypothyroidism: a deficiency of thyroid hormone from birth, which untreated can lead to cretinism
High function
[edit]
Hyperthyroidism is a state in which the body is producing too much thyroid hormone. The main hyperthyroid conditions are:
- Graves' disease
- Toxic thyroid nodule
- Thyroid storm
- Toxic nodular struma (Plummer's disease)
- Hashitoxicosis: transient hyperthyroidism that can occur in Hashimoto's thyroiditis
Structural abnormalities
[edit]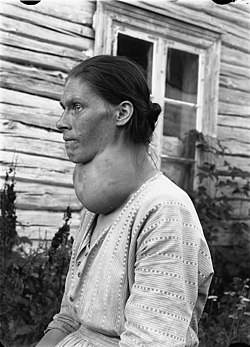
- Goiter: an abnormal enlargement of the thyroid gland
- Lingual thyroid
- Thyroglossal duct cyst
Tumors
[edit]- Thyroid cancer
- Lymphomas are usually malignant
- Thyroid adenomas are benign tumors
Medication side effects
[edit]Certain medications can have the unintended side effect of affecting thyroid function. While some medications can lead to significant hypothyroidism or hyperthyroidism and those at risk will need to be carefully monitored, some medications may affect thyroid hormone lab tests without causing any symptoms or clinical changes, and may not require treatment.[citation needed] The following medications have been linked to various forms of thyroid disease:
- Amiodarone (more commonly can lead to hypothyroidism, but can be associated with some types of hyperthyroidism)[21]
- Lithium salts (hypothyroidism)
- Some types of interferon and IL-2 (thyroiditis)[22]
- Glucocorticoids, dopamine agonists, and somatostatin analogs (block TSH, which can lead to hypothyroidism)[22]
Pathophysiology
[edit]Most thyroid disease in the United States stems from a condition where the body's immune system attacks itself. In other instances, thyroid disease comes from the body trying to adapt to environmental conditions like iodine deficiency or to new physiologic conditions like pregnancy.
Autoimmune Thyroid Disease
[edit]Autoimmune thyroid disease is a general category of disease that occurs due to the immune system targeting its own body. It is not fully understood why this occurs, but it is thought to be partially genetic as these diseases tend to run in families.[9] In one of the most common types, Graves' Disease, the body produces antibodies against the TSH receptor on thyroid cells.[4] This causes the receptor to activate even without TSH being present and causes the thyroid to produce and release excess thyroid hormone (hyperthyroidism).[4] Another common form of autoimmune thyroid disease is Hashimoto's thyroiditis where the body produces antibodies against different normal components of the thyroid gland, most commonly thyroglobulin, thyroid peroxidase, and the TSH receptor.[9] These antibodies cause the immune system to attack the thyroid cells and cause inflammation (lymphocytic infiltration) and destruction (fibrosis) of the gland.[9]
Goiter
[edit]Goiter is the general enlargement of the thyroid that can be associated with many thyroid diseases. The main reason this happens is because of increased signaling to the thyroid by way of TSH receptors to try to make it produce more thyroid hormone.[9] This causes increased vascularity and increase in size (hypertrophy) of the gland.[9] In hypothyroid states or iodine deficiency, the body recognizes that it is not producing enough thyroid hormone and starts to produce more TSH to help stimulate the thyroid to produce more thyroid hormone.[9] This stimulation causes the gland to increase in size to increase production of thyroid hormone. In hyperthyroidism caused by Graves' Disease or toxic multinodular goiter, there is excess stimulation of the TSH receptor even when thyroid hormone levels are normal.[4] In Graves' Disease this is because of an autoantibodies (Thyroid Stimulating Immunoglobulins) which bind to and activate the TSH receptors in place of TSH while in toxic multinodular goiter this is often because of a mutation in the TSH receptor that causes it to activate without receiving a signal from TSH.[4] In more rare cases, the thyroid may become enlarged because it becomes filled with thyroid hormone or thyroid hormone precursors that it is unable to release or because of congenital abnormalities or because of increased intake of iodine from supplementation or medication.[9]
Pregnancy
[edit]There are many changes to the body during pregnancy. One of the major changes to help with the development of the fetus is the production of human chorionic gonadotropin (hCG). This hormone, produced by the placenta, has similar structure to TSH and can bind to the maternal TSH receptor to produce thyroid hormone.[23] During pregnancy, there is also an increase in estrogen which causes the mother to produce more thyroxine binding globulin, which is what carries most of the thyroid hormone in the blood.[24] These normal hormonal changes often make pregnancy look like a hyperthyroid state but may be within the normal range for pregnancy, so it necessary to use trimester specific ranges for TSH and free T4.[23][24] True hyperthyroidism in pregnancy is most often caused by an autoimmune mechanism from Graves' Disease.[23] New diagnosis of hypothyroidism in pregnancy is rare because hypothyroidism often makes it difficult to become pregnant in the first place.[23] When hypothyroidism is seen in pregnancy, it is often because an individual already has hypothyroidism and needs to increase their levothyroxine dose to account for the increased thyroxine binding globulin present in pregnancy.[23]
Diagnosis
[edit]Diagnosis of thyroid disease depends on symptoms and whether or not a thyroid nodule is present. Most patients will receive a blood test. Others might need an ultrasound, biopsy or a radioiodine scanning and uptake study.
Blood tests
[edit]
Thyroid function tests
[edit]There are several hormones that can be measured in the blood to determine how the thyroid gland is functioning. These include the thyroid hormones triiodothyronine (T3) and its precursor thyroxine (T4), which are produced by the thyroid gland. Thyroid-stimulating hormone (TSH) is another important hormone that is secreted by the anterior pituitary cells in the brain. Its primary function is to increase the production of T3 and T4 by the thyroid gland.
The most useful marker of thyroid gland function is serum thyroid-stimulating hormone (TSH) levels. TSH levels are determined by a classic negative feedback system in which high levels of T3 and T4 suppress the production of TSH, and low levels of T3 and T4 increase the production of TSH. TSH levels are thus often used by doctors as a screening test, where the first approach is to determine whether TSH is elevated, suppressed, or normal.[25]
- Elevated TSH levels can signify inadequate thyroid hormone production (hypothyroidism)
- Suppressed TSH levels can point to excessive thyroid hormone production (hyperthyroidism)
Because a single abnormal TSH level can be misleading, T3 and T4 levels must be measured in the blood to further confirm the diagnosis. When circulating in the body, T3 and T4 are bound to transport proteins. Only a small fraction of the circulating thyroid hormones are unbound or free, and thus biologically active. T3 and T4 levels can thus be measured as free T3 and T4, or total T3 and T4, which takes into consideration the free hormones in addition to the protein-bound hormones. Free T3 and T4 measurements are important because certain drugs and illnesses can affect the concentrations of transport proteins, resulting in differing total and free thyroid hormone levels. There are differing guidelines for T3 and T4 measurements.
- Free T4 levels should be measured in the evaluation of hypothyroidism, and low free T4 establishes the diagnosis. T3 levels are generally not measured in the evaluation of hypothyroidism.[13]
- Free T4 and total T3 can be measured when hyperthyroidism is of high suspicion as it will improve the accuracy of the diagnosis. Free T4, total T3 or both are elevated and serum TSH is below normal in hyperthyroidism. If the hyperthyroidism is mild, only serum T3 may be elevated and serum TSH can be low or may not be detected in the blood.[14]
- Free T4 levels may also be tested in patients who have convincing symptoms of hyper- and hypothyroidism, despite a normal TSH.
Antithyroid antibodies
[edit]Autoantibodies to the thyroid gland may be detected in various disease states. There are several anti-thyroid antibodies, including anti-thyroglobulin antibodies (TgAb), anti-microsomal/anti-thyroid peroxidase antibodies (TPOAb), and TSH receptor antibodies (TSHRAb).[13]
- Elevated anti-thryoglobulin (TgAb) and anti-thyroid peroxidase antibodies (TPOAb) can be found in patients with Hashimoto's thyroiditis, the most common autoimmune type of hypothyroidism. TPOAb levels have also been found to be elevated in patients who present with subclinical hypothyroidism (where TSH is elevated, but free T4 is normal), and can help predict progression to overt hypothyroidism. The American Association Thyroid Association thus recommends measuring TPOAb levels when evaluating subclinical hypothyroidism or when trying to identify whether nodular thyroid disease is due to autoimmune thyroid disease.[19]
- When the etiology of hyperthyroidism is not clear after initial clinical and biochemical evaluation, measurement of TSH receptor antibodies (TSHRAb) can help make the diagnosis. In Graves' disease, TSHRAb levels are elevated as they are responsible for activating the TSH receptor and causing increased thyroid hormone production.[18]
Other markers
[edit]- There are two markers for thyroid-derived cancers.
- Thyroglobulin (TG) levels can be elevated in well-differentiated papillary or follicular adenocarcinoma. It is often used to provide information on residual, recurrent or metastatic disease in patients with differentiated thyroid cancer. However, serum TG levels can be elevated in most thyroid diseases. Routine measurement of serum TG for evaluation of thyroid nodules is therefore currently not recommended by the American Thyroid Association.[26]
- Elevated calcitonin levels in the blood have been shown to be associated with the rare medullary thyroid cancer. However, the measurement of calcitonin levels as a diagnostic tool is currently controversial due to falsely high or low calcitonin levels in a variety of diseases other than medullary thyroid cancer.[26][27]
- Very infrequently, TBG and transthyretin levels may be abnormal; these are not routinely tested.
- To differentiate between different types of hypothyroidism, a specific test may be used. Thyrotropin-releasing hormone (TRH) is injected into the body through a vein. This hormone is naturally secreted by the hypothalamus and stimulates the pituitary gland. The pituitary responds by releasing thyroid-stimulating hormone (TSH). Large amounts of externally administered TRH can suppress the subsequent release of TSH. This amount of release-suppression is exaggerated in primary hypothyroidism, major depression, cocaine dependence, amphetamine dependence and chronic phencyclidine abuse. There is a failure to suppress in the manic phase of bipolar disorder.[28]
Ultrasound
[edit]Many people may develop a thyroid nodule at some point in their lives. Although many who experience this worry that it is thyroid cancer, there are many causes of nodules that are benign and not cancerous. If a possible nodule is present, a doctor may order thyroid function tests to determine if the thyroid gland's activity is being affected. If more information is needed after a clinical exam and lab tests, medical ultrasonography can help determine the nature of thyroid nodule(s). There are some notable differences in typical benign vs. cancerous thyroid nodules that can particularly be detected by the high-frequency sound waves in an ultrasound scan. The ultrasound may also locate nodules that are too small for a doctor to feel on a physical exam, and can demonstrate whether a nodule is primarily solid, liquid (cystic), or a mixture of both. It is an imaging process that can often be done in a doctor's office, is painless, and does not expose the individual to any radiation.[29]
The main characteristics that can help distinguish a benign vs. malignant (cancerous) thyroid nodule on ultrasound are as follows:[30]
| Possible thyroid cancer | More likely benign |
|---|---|
| irregular borders | smooth borders |
| hypoechoic (less echogenic than the surrounding tissue) | hyperechoic |
| incomplete "halo" | spongiform appearance |
| significant intranodular / central blood flow by power Doppler | marked peripheral blood flow |
| microcalcifications | larger, broad calcifications (note: these can be seen in medullary thyroid cancer) |
| nodule appears more tall than wide on transverse study | "comet tail" artifact as sound waves bounce off intranodular colloid |
| documented progressive increase in size of nodule on ultrasound |
Although ultrasonography is a very important diagnostic tool, this method is not always able to separate benign from malignant nodules with certainty. In suspicious cases, a tissue sample is often obtained by biopsy for microscopic examination.
Radioiodine scanning and uptake
[edit]
Thyroid scintigraphy, in which the thyroid is imaged with the aid of radioactive iodine (usually iodine-123, which does not harm thyroid cells, or rarely, iodine-131),[31] is performed in the nuclear medicine department of a hospital or clinic. Radioiodine collects in the thyroid gland before being excreted in the urine. While in the thyroid, the radioactive emissions can be detected by a camera, producing a rough image of the shape (a radioiodine scan) and tissue activity (a radioiodine uptake) of the thyroid gland.
A normal radioiodine scan shows even uptake and activity throughout the gland. Irregular uptake can reflect an abnormally shaped or abnormally located gland, or it can indicate that a portion of the gland is overactive or underactive. For example, a nodule that is overactive ("hot") -- to the point of suppressing the activity of the rest of the gland—is usually a thyrotoxic adenoma, a surgically curable form of hyperthyroidism that is rarely malignant. In contrast, finding that a substantial section of the thyroid is inactive ("cold") may indicate an area of non-functioning tissue, such as thyroid cancer.
The amount of radioactivity can be quantified and serves as an indicator of the metabolic activity of the gland. A normal quantitation of radioiodine uptake demonstrates that about 8-35% of the administered dose can be detected in the thyroid 24 hours later. Overactivity or underactivity of the gland, as may occur with hyperthyroidism or hypothyroidism, is usually reflected in increased or decreased radioiodine uptake. Different patterns may occur with different causes of hypo- or hyperthyroidism.
Biopsy
[edit]A medical biopsy refers to the obtaining of a tissue sample for examination under the microscope or other testing, usually to distinguish cancer from noncancerous conditions. Thyroid tissue may be obtained for biopsy by fine needle aspiration (FNA) or by surgery.[citation needed]
Fine needle aspiration has the advantage of being a brief, safe, outpatient procedure that is safer and less expensive than surgery and does not leave a visible scar. Needle biopsies became widely used in the 1980s, though it was recognized that the accuracy of identification of cancer was good, but not perfect. The accuracy of the diagnosis depends on obtaining tissue from all of the suspicious areas of an abnormal thyroid gland. The reliability of fine needle aspiration is increased when sampling can be guided by ultrasound, and over the last 15 years, this has become the preferred method for thyroid biopsy in North America.[32][citation needed]
Treatment
[edit]Medication
[edit]Levothyroxine is a stereoisomer of thyroxine (T4) which is degraded much more slowly and can be administered once daily in patients with hypothyroidism.[13] Natural thyroid hormone from pigs is sometimes also used, especially for people who cannot tolerate the synthetic version. Hyperthyroidism caused by Graves' disease may be treated with the thioamide drugs propylthiouracil, carbimazole or methimazole, or rarely with Lugol's solution. Additionally, hyperthyroidism and thyroid tumors may be treated with radioactive iodine. Ethanol injections for the treatment of recurrent thyroid cysts and metastatic thyroid cancer in lymph nodes can also be an alternative to surgery.[citation needed]
Surgery
[edit]Thyroid surgery is performed for a variety of reasons. A nodule or lobe of the thyroid is sometimes removed for biopsy or because of the presence of an autonomously functioning adenoma causing hyperthyroidism. A large majority of the thyroid may be removed (subtotal thyroidectomy) to treat the hyperthyroidism of Graves' disease, or to remove a goiter that is unsightly or impinges on vital structures.[citation needed]
A complete thyroidectomy of the entire thyroid, including associated lymph nodes, is the preferred treatment for thyroid cancer. Removal of the bulk of the thyroid gland usually produces hypothyroidism unless the person takes thyroid hormone replacement. Consequently, individuals who have undergone a total thyroidectomy are typically placed on thyroid hormone replacement (e.g. levothyroxine) for the remainder of their lives. Higher than normal doses are often administered to prevent recurrence.[citation needed]
If the thyroid gland must be removed surgically, care must be taken to avoid damage to adjacent structures, the parathyroid glands and the recurrent laryngeal nerve. Both are susceptible to accidental removal and/or injury during thyroid surgery.[citation needed]
The parathyroid glands produce parathyroid hormone (PTH), a hormone needed to maintain adequate amounts of calcium in the blood. Removal results in hypoparathyroidism and a need for supplemental calcium and vitamin D each day. In the event that the blood supply to any one of the parathyroid glands is endangered through surgery, the parathyroid gland(s) involved may be re-implanted in surrounding muscle tissue.
The recurrent laryngeal nerves provide motor control for all external muscles of the larynx except for the cricothyroid muscle, which also runs along the posterior thyroid. Accidental laceration of either of the two or both recurrent laryngeal nerves may cause paralysis of the vocal cords and their associated muscles, changing the voice quality. A 2019 systematic review concluded that the available evidence shows no difference between visually identifying the nerve or utilizing intraoperative neuroimaging during surgery, when trying to prevent injury to recurrent laryngeal nerve during thyroid surgery.[33]
Radioiodine
[edit]Radioiodine therapy with iodine-131 can be used to shrink the thyroid gland (for instance, in the case of large goiters that cause symptoms but do not harbor cancer—after evaluation and biopsy of suspicious nodules has been done), or to destroy hyperactive thyroid cells (for example, in cases of thyroid cancer). The iodine uptake can be high in countries with iodine deficiency, but low in iodine sufficient countries. To enhance iodine-131 uptake by the thyroid and allow for more successful treatment, TSH is raised prior to therapy in order to stimulate the existing thyroid cells. This is done either by withdrawal of thyroid hormone medication or injections of recombinant human TSH (Thyrogen),[31] released in the United States in 1999. Thyrogen injections can reportedly boost uptake up to 50-60%. Radioiodine treatment can also cause hypothyroidism (which is sometimes the end goal of treatment) and, although rare, a pain syndrome (due to radiation thyroiditis).[34]
Epidemiology
[edit]In the United States, autoimmune inflammation is the most common form of thyroid disease while worldwide hypothyroidism and goiter due to dietary iodine deficiency is the most common.[35][4] According to the American Thyroid Association in 2015, approximately 20 million people in the United States alone are affected by thyroid disease.[11][36] Hypothyroidism affects 3-10% percent of adults, with a higher incidence in women and the elderly.[37][38][39] An estimated one-third of the world's population currently lives in areas of low dietary iodine levels. In regions of severe iodine deficiency, the prevalence of goiter is as high as 80%.[40] In areas where iodine-deficiency is not found, the most common type of hypothyroidism is an autoimmune subtype called Hashimoto's thyroiditis, with a prevalence of 1-2%.[40] As for hyperthyroidism, Graves' disease, another autoimmune condition, is the most common type with a prevalence of 0.5% in males and 3% in females.[41] Although thyroid nodules are common, thyroid cancer is rare. Thyroid cancer accounts for less than 1% of all cancer in the UK, though it is the most common endocrine tumor and makes up greater than 90% of all cancers of the endocrine glands.[40]
See also
[edit]References
[edit]- ^ Hall JE, Guyton AC. Guyton and Hall textbook of medical physiology. OCLC 434319356.
- ^ a b c d e f g Bauer DC (2013). Hammer G, McPhee SJ (eds.). Pathophysiology of Disease: An Introduction to Clinical Medicine (Seventh ed.). New York, NY.: McGraw-Hill – via AccessMedicine.
- ^ Hollowell, Joseph G.; Staehling, Norman W.; Flanders, W. Dana; Hannon, W. Harry; Gunter, Elaine W.; Spencer, Carole A.; Braverman, Lewis E. (2002). "Serum TSH, T4, and Thyroid Antibodies in the United States Population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III)". The Journal of Clinical Endocrinology & Metabolism. 87 (2): 489–499. doi:10.1210/jcem.87.2.8182. PMID 11836274. S2CID 10850710.
- ^ a b c d e f Papadakis MA, McPhee SJ, Rabow MW, eds. (2018-09-07). "Endocrine Disorders". Current medical diagnosis & treatment 2019. ISBN 978-1-260-11743-1. OCLC 1050994785.
- ^ "Hypothyroidism (Underactive Thyroid)". www.niddk.nih.gov. Retrieved 2016-12-16.
- ^ "Hyperthyroidism". www.niddk.nih.gov. Archived from the original on 2016-12-20. Retrieved 2016-12-16.
- ^ a b c Kasper DL, Fauci AS, Hauser SL, Longo DL, Larry Jameson J, Loscalzo J (2018-02-06). "Thyroid Nodular Disease and Thyroid Cancer". Harrison's principles of internal medicine (Twentieth ed.). New York. ISBN 978-1-259-64404-7. OCLC 990065894.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ "Final Recommendation Statement: Thyroid Dysfunction: Screening - US Preventive Services Task Force". www.uspreventiveservicestaskforce.org. Retrieved 2018-11-30.
- ^ a b c d e f g h i Hammer GD, McPhee SJ (2018-11-26). "Thyroid Disease". Pathophysiology of disease: an introduction to clinical medicine (Eighth ed.). New York. ISBN 978-1-260-02650-4. OCLC 1056106178.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Turkish Endocrinology and Metabolism Association. (2013). Tiroid hastalıkları tanı ve tedavi kılavuzu [Diagnosis and treatment of thyroid diseases guide] (5th Edition). Ankara, Turkey: Miki Matbaacılık.
- ^ a b Atasayar S, Guler Demir S (June 2019). "Determination of the Problems Experienced by Patients Post-Thyroidectomy". Clinical Nursing Research. 28 (5): 615–635. doi:10.1177/1054773817729074. PMID 28882054. S2CID 8593999.
- ^ Müller PE, Jakoby R, Heinert G, Spelsberg F (November 2001). "Surgery for recurrent goitre: its complications and their risk factors". The European Journal of Surgery = Acta Chirurgica. 167 (11): 816–21. doi:10.1080/11024150152717634. PMID 11848234.
- ^ a b c d Garber JR, Cobin RH, Gharib H, Hennessey JV, Klein I, Mechanick JI, et al. (2012-12-17). "Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association". Endocrine Practice. 18 (6): 988–1028. doi:10.4158/ep12280.gl. PMID 23246686.
- ^ a b c Ross DS, Burch HB, Cooper DS, Greenlee MC, Laurberg P, Maia AL, et al. (October 2016). "2016 American Thyroid Association Guidelines for Diagnosis and Management of Hyperthyroidism and Other Causes of Thyrotoxicosis". Thyroid (Submitted manuscript). 26 (10): 1343–1421. doi:10.1089/thy.2016.0229. PMID 27521067.
- ^ "Hypothyroidism - American Thyroid Association". www.thyroid.org. Retrieved 2017-04-25.
- ^ "Underactive thyroid (hypothyroidism) - Symptoms - NHS Choices". NHS Choices. Retrieved 17 December 2016.
- ^ "Hyperthyroidism - American Thyroid Association". www.thyroid.org. Retrieved 2017-04-25.
- ^ a b "Hyperthyroidism - American Thyroid Association". www.thyroid.org. Retrieved 2016-12-13.
- ^ a b "Hypothyroidism - American Thyroid Association". www.thyroid.org. Retrieved 2016-12-13.
- ^ Vitti P, Latrofa F (2013-01-01). "Iatrogenic Hypothyroidism and Its Sequelae". In Miccoli P, Terris DJ, Minuto MN, Seybt MW (eds.). Thyroid Surgery. John Wiley & Sons, Ltd. pp. 291–303. doi:10.1002/9781118444832.ch32. ISBN 978-1-118-44483-2.
- ^ Batcher EL, Tang XC, Singh BN, Singh SN, Reda DJ, Hershman JM (October 2007). "Thyroid function abnormalities during amiodarone therapy for persistent atrial fibrillation". The American Journal of Medicine. 120 (10): 880–885. doi:10.1016/j.amjmed.2007.04.022. PMID 17904459.
- ^ a b Haugen BR (December 2009). "Drugs that suppress TSH or cause central hypothyroidism". Best Practice & Research. Clinical Endocrinology & Metabolism. 23 (6): 793–800. doi:10.1016/j.beem.2009.08.003. PMC 2784889. PMID 19942154.
- ^ a b c d e Gardner DG, Shoback DM, Greenspan FS (eds.). "The Endocrinology of Pregnancy". Greenspan's Basic & Clinical Endocrinology. OCLC 995848612.
- ^ a b Jameson JL, Fauci AS, Kasper DL, Hauser SL, Longo DL, Loscalzo J, Harrison TR (2018). "Medical Disorders During Pregnancy". Harrison's principles of internal medicine. The McGraw-Hill Companies. OCLC 1043046717.
- ^ Longo DL, Jameson JL, Kaspe D (2015). Harrison's Principal of Internal Medicine (19th ed.). New York: NY: McGraw-Hill. pp. Ch 405 – via Access Medicine.
- ^ a b Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. (January 2016). "2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer". Thyroid. 26 (1): 1–133. doi:10.1089/thy.2015.0020. PMC 4739132. PMID 26462967.
- ^ Wells SA, Asa SL, Dralle H, Elisei R, Evans DB, Gagel RF, et al. (June 2015). "Revised American Thyroid Association guidelines for the management of medullary thyroid carcinoma". Thyroid. 25 (6): 567–610. doi:10.1089/thy.2014.0335. PMC 4490627. PMID 25810047.
- ^ Giannini AJ, Malone DA, Loiselle RH, Price WA (January 1987). "Blunting of TSH response to TRH in chronic cocaine and phencyclidine abusers". The Journal of Clinical Psychiatry. 48 (1): 25–26. PMID 3100509.
- ^ "Thyroid Nodules - American Thyroid Association". www.thyroid.org. Retrieved 2016-12-13.
- ^ "Diagnostic approach to and treatment of thyroid nodules". www.uptodate.com. Retrieved 2016-12-13.
- ^ a b "Radioactive Iodine - American Thyroid Association". www.thyroid.org. Retrieved 2016-12-14.
- ^ Dean DS, Gharib H (2000). "Fine-Needle Aspiration Biopsy of the Thyroid Gland". In Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, et al. (eds.). Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc. PMID 25905400. Retrieved 29 Jun 2020.
- ^ Cirocchi R, Arezzo A, D'Andrea V, Abraha I, Popivanov GI, Avenia N, et al. (Cochrane Metabolic and Endocrine Disorders Group) (January 2019). "Intraoperative neuromonitoring versus visual nerve identification for prevention of recurrent laryngeal nerve injury in adults undergoing thyroid surgery". The Cochrane Database of Systematic Reviews. 1 (1) CD012483. doi:10.1002/14651858.CD012483.pub2. PMC 6353246. PMID 30659577.
- ^ Shah KK, Tarasova VD, Davidian M, Anderson RJ (2014-06-01). "Non-Neoplastic Thyroid Disorders-Clinical and Case Reports". Endo Meetings. Meeting A bstracts: SAT–0523–SAT–0523.
- ^ Taylor PN, Albrecht D, Scholz A, Gutierrez-Buey G, Lazarus JH, Dayan CM, Okosieme OE (May 2018). "Global epidemiology of hyperthyroidism and hypothyroidism" (PDF). Nature Reviews. Endocrinology. 14 (5): 301–316. doi:10.1038/nrendo.2018.18. PMID 29569622. S2CID 205482747.
- ^ "Prevalence and Impact of Thyroid Disease". American Thyroid Association.
- ^ Gharib H, Tuttle RM, Baskin HJ, Fish LH, Singer PA, McDermott MT (2004). "Subclinical thyroid dysfunction: a joint statement on management from the American Association of Clinical Endocrinologists, the American Thyroid Association, and the Endocrine Society". Endocrine Practice. 10 (6): 497–501. doi:10.4158/ep.10.6.497. PMID 16033723.
- ^ Fatourechi V (2009). "Subclinical hypothyroidism: an update for primary care physicians". Mayo Clinic Proceedings. 84 (1): 65–71. doi:10.4065/84.1.65. PMC 2664572. PMID 19121255.[dead YouTube link]
- ^ Villar HC, Saconato H, Valente O, Atallah AN (July 2007). "Thyroid hormone replacement for subclinical hypothyroidism". The Cochrane Database of Systematic Reviews. 2007 (3) CD003419. doi:10.1002/14651858.CD003419.pub2. PMC 6610974. PMID 17636722.
- ^ a b c Vanderpump MP (2011-09-01). "The epidemiology of thyroid disease". British Medical Bulletin. 99 (1): 39–51. doi:10.1093/bmb/ldr030. PMID 21893493.
- ^ Burch HB, Cooper DS (December 2015). "Management of Graves Disease: A Review". JAMA. 314 (23): 2544–2554. doi:10.1001/jama.2015.16535. PMID 26670972.
External links
[edit]Thyroid disease
View on GrokipediaAnatomy and Physiology
Thyroid gland structure
The thyroid gland is a butterfly-shaped endocrine organ located in the anterior neck, positioned anterior to the trachea and overlying the C5 to T1 vertebral levels within the middle compartment of the neck, bounded anteriorly by the strap muscles and posteriorly by the deep cervical fascia.[8] It consists of two symmetrical lobes, a right and a left, connected by a narrow isthmus that crosses the anterior surface of the trachea at the level of the second and third tracheal rings.[8] The gland is anchored to the trachea via the Berry ligament and may include a pyramidal lobe or tubercle of Zuckerkandl extending superiorly from the isthmus in some individuals.[8] The thyroid's parenchyma is derived from endoderm and comprises follicular units surrounded by a connective tissue capsule that extends septa into the gland to form lobules.[8] Histologically, it features spherical follicles lined by a simple epithelium of cuboidal follicular cells that vary in height—squamous when inactive and columnar when active—resting on a basement membrane.[9] These follicles contain a central colloid-filled lumen composed of thyroglobulin, an iodinated glycoprotein stored for hormone synthesis.[9] Interspersed among the follicular cells are parafollicular (C) cells, which are large, pale-staining, polyhedral cells located between the follicular epithelium and the basement membrane, not extending to the follicular lumen; these cells originate from the ultimobranchial body and produce calcitonin.[8][9] Blood supply to the thyroid gland is provided primarily by the superior thyroid artery, the first branch of the external carotid artery, which supplies the upper poles and isthmus via branches such as the cricothyroid artery, and the inferior thyroid artery, the third branch of the thyrocervical trunk, which supplies the lower poles and parathyroids.[10] These arteries anastomose bilaterally to ensure robust dual perfusion.[10] An accessory thyroid ima artery arises in approximately 10% of cases from variable origins, such as the brachiocephalic trunk, to supplement the supply.[8] Venous drainage occurs via the superior and middle thyroid veins into the internal jugular vein and the inferior thyroid veins into the brachiocephalic veins.[10] Lymphatic drainage from the thyroid follows patterns critical for understanding metastatic spread in thyroid cancer, with initial drainage to prelaryngeal, pretracheal, and paratracheal nodes, and further to lower deep cervical nodes; the isthmus and inferior lobes particularly drain to paratracheal nodes, while superior poles favor prelaryngeal routes.[8] The gland is closely related to surrounding structures, including the recurrent laryngeal nerves posterolaterally, the parathyroid glands posteriorly, and the carotid sheath laterally.[8]Hormone production and regulation
The thyroid gland synthesizes thyroid hormones primarily in the form of thyroxine (T4) and triiodothyronine (T3) within the follicular lumen. Iodide, essential for hormone production, is actively transported into thyroid follicular cells via the sodium-iodide symporter (NIS), a plasma membrane glycoprotein that uses the sodium gradient established by the Na+/K+-ATPase to mediate a 2 Na+:1 I- stoichiometry, concentrating iodide up to 20-40 times higher than in plasma.[11] NIS expression and activity are primarily regulated by thyroid-stimulating hormone (TSH), ensuring adequate iodide availability for hormone synthesis.[11] Thyrocytes produce thyroglobulin (Tg), a large glycoprotein precursor synthesized on ribosomes and secreted into the follicular lumen, where it serves as the scaffold for hormone formation. Iodide is oxidized by thyroid peroxidase (TPO) in the presence of hydrogen peroxide to form reactive iodine, which undergoes organification by iodinating specific tyrosine residues on Tg to produce monoiodotyrosine (MIT) and diiodotyrosine (DIT).[12] The coupling reaction, also catalyzed by TPO, links two DIT residues to form T4 or one MIT and one DIT to form T3, with these hormones remaining bound to Tg for storage in the colloid.[12] Upon stimulation by TSH, colloid is endocytosed, and lysosomal enzymes cleave Tg to release T4 (about 80% of secreted hormone) and T3 (about 20%), which are then transported into the bloodstream via the monocarboxylate transporter 8 (MCT8).[12] Most circulating T4 serves as a prohormone, converted peripherally to the more active T3 by deiodinases. Type 1 deiodinase (D1), expressed mainly in the liver, kidney, and thyroid, performs outer-ring deiodination to convert T4 to T3 and contributes to about 20-30% of serum T3 levels while also recycling iodide.[13] Type 2 deiodinase (D2), found in the central nervous system, pituitary, brown adipose tissue, and skeletal muscle, locally generates T3 from T4 to maintain intracellular hormone levels, accounting for 70-80% of brain T3.[13] In contrast, type 3 deiodinase (D3), predominant in fetal tissues, placenta, and certain adult brain regions, inactivates T4 by inner-ring deiodination to reverse T3 (rT3) and T3 to 3,3'-T2, protecting tissues from excess hormone exposure.[13] In circulation, over 99% of thyroid hormones are bound to transport proteins, with only the free fraction being biologically active. Thyroxine-binding globulin (TBG), the primary carrier, binds approximately 70-75% of T4 and T3 with high affinity (association constant ~10^10 M^{-1} for T4), while transthyretin (TTR) binds 15-20% of T4 and less than 1% of T3 (association constant ~2 \times 10^8 M^{-1} for T4), and albumin binds the remainder (10-15% of T4, 25-30% of T3) with lower affinity (association constant ~1.5 \times 10^6 M^{-1} for T4).[14] These proteins modulate hormone delivery to tissues and protect against rapid clearance.[14] Hormone production and secretion are tightly regulated by the hypothalamic-pituitary-thyroid (HPT) axis. The hypothalamus secretes thyrotropin-releasing hormone (TRH), which stimulates the anterior pituitary to release TSH; TSH binds to receptors on thyrocytes, activating adenylate cyclase and cAMP pathways to upregulate NIS, TPO, Tg synthesis, iodide uptake, and hormone release.[12] Circulating T3 and T4 exert negative feedback by inhibiting TRH and TSH secretion at the hypothalamus and pituitary, respectively, maintaining homeostasis.[12] In healthy adults, the thyroid produces approximately 90-120 \mu g of T4 and 6-8 \mu g of T3 daily, requiring about 150 \mu g of iodine intake to support this synthesis, as each T4 molecule incorporates four iodine atoms and T3 three.[15] The gland concentrates 70-80% of total body iodine (15-20 mg) to meet these demands.[15]Clinical Presentation
Hypothyroidism symptoms
Hypothyroidism manifests through a range of symptoms resulting from reduced thyroid hormone levels, which slow metabolic processes across multiple body systems. These symptoms can develop gradually and vary in severity, often mimicking other conditions, but collectively point to underactive thyroid function.[6][16] Common symptoms include profound fatigue, where individuals experience persistent tiredness that interferes with daily activities despite adequate rest.[6] Weight gain occurs due to slowed metabolism and fluid retention, even without changes in diet or exercise.[16] Patients often report cold intolerance, feeling unusually sensitive to low temperatures.[6] Gastrointestinal effects manifest as constipation from decreased gut motility.[16] Skin and hair changes include dry skin and hair loss, with brittle, thinning hair and rough, scaly skin.[6] Cardiovascular signs feature bradycardia, a slowed heart rate that may lead to reduced cardiac output.[16] Myxedema, characterized by non-pitting edema, presents as puffy, doughy swelling in the face, hands, and legs due to mucopolysaccharide accumulation in the dermis.[17] Neurological examination may reveal a prolonged relaxation phase in deep tendon reflexes, reflecting delayed muscle response.[16] Cognitive effects encompass depression, memory impairment, and slowed thinking, contributing to difficulties in concentration and mental processing.[6] Reproductive issues include menstrual irregularities such as heavy or prolonged periods in women, along with infertility in both sexes due to hormonal disruptions.[16] In severe, untreated cases, myxedema coma can occur, a life-threatening emergency marked by hypothermia, hypotension, and hyponatremia, often accompanied by altered mental status and organ failure.[17][16] In pediatric cases, particularly congenital hypothyroidism, symptoms lead to cretinism, featuring severe developmental delays, intellectual disability, stunted physical growth, and motor deficits if untreated in early infancy.[18]Hyperthyroidism symptoms
Hyperthyroidism, characterized by excessive production of thyroid hormones, leads to a hypermetabolic state that manifests in a wide array of symptoms affecting multiple organ systems. Common early signs include unintentional weight loss despite an increased appetite, as the accelerated metabolism burns calories at a higher rate. Heat intolerance and excessive sweating (diaphoresis) are also frequent, resulting from heightened thermogenesis and vasodilation.[7][19][20] Cardiovascular symptoms are prominent, with tachycardia (rapid heartbeat) and palpitations often reported due to the stimulatory effects of thyroid hormones on the heart. Irregular heart rhythms, such as atrial fibrillation, can occur, particularly in older adults, increasing the risk of stroke. Nervous system involvement produces tremors, typically fine and affecting the hands or fingers, alongside anxiety, irritability, and insomnia, reflecting the influence on the central nervous system.[7][20][19] Gastrointestinal disturbances include more frequent bowel movements or diarrhea, stemming from enhanced gut motility. Musculoskeletal effects encompass proximal muscle weakness, which may impair daily activities, and an elevated risk of osteoporosis due to accelerated bone turnover and reduced bone mineral density. An enlarged thyroid gland, or goiter, is often palpable as swelling at the base of the neck.[7][19][20] In cases associated with autoimmune processes like Graves' disease, the most common cause of hyperthyroidism, additional manifestations may appear. Ocular symptoms include exophthalmos (bulging eyes), lid lag, and a characteristic stare from eyelid retraction, arising from inflammation of orbital tissues. Rare skin changes, such as pretibial myxedema—a thickened, discolored area on the shins—can also occur due to mucin deposition. These extrathyroidal features highlight the systemic autoimmune nature of the condition.[7][21][22]Structural abnormality symptoms
Structural abnormalities in the thyroid gland, such as goiters and nodules, can lead to symptoms arising from mechanical compression or distortion of surrounding neck structures, rather than alterations in hormone production. These manifestations often result from the physical enlargement or irregular growth of the gland, which may press on the esophagus, trachea, or adjacent nerves. Common presentations include neck swelling, which is frequently the initial noticeable sign, creating a visible or palpable mass at the base of the neck.[23] Goiters, characterized by diffuse or nodular enlargement of the thyroid, can cause compressive symptoms when sufficiently large. Dysphagia, or difficulty swallowing, occurs due to pressure on the esophagus, while dysphonia, a change in voice quality, may arise from compression of the trachea or recurrent laryngeal nerve. Additional effects include a sensation of choking or tightness in the neck, particularly during swallowing or neck movement. In severe cases, tracheal compression can lead to stridor or wheezing, though this is less common in non-substernal goiters.[24][25] Thyroiditis, an inflammatory condition, often presents with thyroid enlargement that can be either painless or painful. In subacute thyroiditis, the gland becomes tender and swollen, accompanied by localized pain that may radiate to the jaw or ears. Painless thyroiditis, such as silent or postpartum variants, typically involves a nontender enlargement without significant discomfort from the swelling itself. These changes contribute to a feeling of fullness in the neck but are distinguished from compressive issues by their inflammatory origin.[26][27] Hoarseness is a key symptom when structural abnormalities involve the recurrent laryngeal nerve, which runs close to the thyroid gland. Enlargement from goiters or masses can compress or stretch this nerve, leading to unilateral vocal cord paralysis and altered voice production. This hoarseness is often persistent and may worsen with gland growth, prompting evaluation for nerve entrapment.[25] Thyroid nodules, as discrete lumps within the gland, primarily cause symptoms if they grow large enough to exert compressive effects. Large nodules (>3-4 cm) may produce dysphagia, neck pressure, or a globus sensation (lump in the throat), similar to goiter-related issues. While most nodules are asymptomatic, functional ones rarely contribute to endocrine symptoms, but the focus here remains on mechanical impacts like airway narrowing in oversized cases.[28][29] Massive goiters, often weighing over 200 grams, heighten the risk of severe compressive symptoms, including significant respiratory distress from tracheal deviation or narrowing. Patients may experience orthopnea (difficulty breathing when lying flat) or exertional dyspnea, alongside prominent cosmetic concerns from the visible neck deformity. These large enlargements can also lead to venous congestion, causing facial plethora in extreme retrosternal extensions.[30][31] Multinodular goiters feature multiple irregular lumps that create an uneven, bosselated thyroid contour, often palpable as distinct nodules under the skin. These can produce localized pressure symptoms if dominant nodules enlarge, and in some instances, autonomous nodules within the multinodular structure may drive uneven growth patterns. The irregular lumps contribute to cosmetic dissatisfaction and intermittent compressive discomfort, particularly with neck flexion. Symptoms from such goiters occasionally overlap with those of thyroid cancer, such as unexplained hoarseness, necessitating further investigation.[32][28]Types of Thyroid Disorders
Hypothyroidism
Hypothyroidism is a condition characterized by insufficient production of thyroid hormones by the thyroid gland, leading to a slowdown in metabolism and various systemic effects. It is one of the most common endocrine disorders, affecting approximately 4.6% of the U.S. population aged 12 years and older, with higher prevalence in women and older adults. The disorder can manifest in overt or subclinical forms and arises from disruptions in thyroid hormone synthesis, release, or regulation. While symptoms such as fatigue, weight gain, and cold intolerance may occur, they are addressed elsewhere in this entry. Hypothyroidism is classified into primary, secondary, and tertiary types based on the level of dysfunction in the hypothalamic-pituitary-thyroid axis. Primary hypothyroidism, the most common form, results from intrinsic thyroid gland failure, accounting for over 99% of cases in iodine-sufficient regions. Secondary hypothyroidism stems from pituitary gland disorders that reduce thyroid-stimulating hormone (TSH) production, while tertiary hypothyroidism involves hypothalamic dysfunction leading to insufficient thyrotropin-releasing hormone. Common causes of primary hypothyroidism include autoimmune destruction via Hashimoto's thyroiditis, which is the leading etiology in developed countries; surgical removal of the thyroid; and treatment with radioactive iodine for hyperthyroidism, which induces permanent hypothyroidism in 80-90% of patients within months to years. In regions with iodine deficiency, such as parts of Central Sub-Saharan Africa and South Asia, inadequate iodine intake remains the predominant cause, contributing to endemic goiter and hypothyroidism with prevalence rates exceeding 5-10% in affected populations. Subclinical hypothyroidism represents a milder variant, defined by elevated serum TSH levels with normal free thyroxine (T4) concentrations, often serving as a precursor to overt disease. It affects about 4-10% of the general population, particularly women over 60, and may progress to clinical hypothyroidism in 2-5% of cases annually without intervention. Untreated hypothyroidism carries significant complications, including increased risk of cardiovascular disease due to dyslipidemia, hypertension, and atherosclerosis; infertility through disrupted ovulation and menstrual irregularities; and, in severe cases, myxedema coma, a life-threatening emergency marked by hypothermia, bradycardia, and multi-organ failure with mortality rates up to 40%. Congenital hypothyroidism, present at birth due to thyroid agenesis, dyshormonogenesis, or maternal factors, affects 1 in 2,000-4,000 newborns globally. Universal newborn screening, implemented since the 1970s using blood TSH measurements, enables early detection and levothyroxine replacement therapy, resulting in near-normal neurocognitive and physical development outcomes in over 90% of treated infants, preventing intellectual disability and growth stunting.Hyperthyroidism
Hyperthyroidism is a condition characterized by the overproduction of thyroid hormones by the thyroid gland, leading to a state of thyrotoxicosis that accelerates metabolism and affects multiple organ systems. It most commonly arises from autoimmune processes, nodular thyroid disease, or inflammation, with Graves' disease accounting for 60-80% of cases in iodine-sufficient regions. This disorder is more prevalent in women and can occur at any age, though it peaks between 20 and 40 years.[33][7] The primary subtypes include Graves' disease, in which circulating autoantibodies stimulate the thyroid-stimulating hormone (TSH) receptor to promote excessive hormone synthesis; toxic multinodular goiter, involving multiple autonomously functioning nodules that produce thyroid hormones independently of TSH regulation, often in older adults or iodine-deficient areas; and toxic adenoma, a solitary hyperfunctioning nodule driven by somatic mutations leading to unchecked hormone secretion. Subclinical hyperthyroidism represents a milder form, defined by suppressed TSH levels with normal free thyroxine (T4) and triiodothyronine (T3) concentrations, which may progress to overt disease or persist asymptomatically, particularly in the elderly. Iatrogenic hyperthyroidism can result from excessive exogenous levothyroxine administration, mimicking endogenous overproduction and suppressing TSH.[19][33][34] Triggers for hyperthyroidism onset or exacerbation include psychosocial stress, which may precipitate Graves' disease through immune dysregulation; smoking, a known risk factor that increases disease severity and relapse rates, particularly in men; and the postpartum period, during which immune rebound can initiate autoimmune thyroid overactivity or thyroiditis leading to transient hyperthyroidism. Complications encompass thyroid storm, a rare but life-threatening exacerbation with systemic decompensation including fever, tachycardia, and altered mental status; accelerated bone loss contributing to osteoporosis, especially in postmenopausal women due to enhanced bone resorption; and cardiovascular risks such as atrial fibrillation and high-output heart failure from chronic tachycardia and increased myocardial oxygen demand. In Graves' disease, thyroid eye disease manifests as orbital inflammation and tissue expansion, causing proptosis, periorbital edema, and potential vision impairment in up to one-third of patients, often independently of thyroid hormone levels.[33][35][34]Goiter and thyroid nodules
A goiter refers to an abnormal enlargement of the thyroid gland, which can be classified as diffuse or nodular based on its structure. Diffuse goiter involves uniform swelling of the entire gland, often resulting from compensatory hyperplasia due to chronic stimulation, such as in iodine deficiency or autoimmune conditions. In contrast, nodular goiter features discrete lumps or masses within the gland, which may evolve from a diffuse form over time. Endemic goiter, a subtype of diffuse or nodular goiter, arises primarily from iodine deficiency in regions with low dietary iodine intake, leading to impaired thyroid hormone synthesis and subsequent glandular hypertrophy to maintain hormone production.[24][23][24] Thyroid nodules are discrete lesions within the thyroid parenchyma, categorized as solitary or multinodular depending on their number and distribution. Solitary nodules occur as isolated masses, while multinodular goiters involve multiple nodules, often developing asymmetrically and potentially causing compressive symptoms like dysphagia or dyspnea if large. Nodules are further distinguished by function: hot or hyperfunctioning nodules actively produce excess thyroid hormone independently of thyroid-stimulating hormone (TSH) regulation, appearing as areas of increased uptake on scintigraphy; cold or nonfunctioning nodules do not produce hormone and show reduced or absent uptake. Most nodules are benign, with common etiologies including cysts (fluid-filled sacs), hemorrhage (intrathyroidal bleeding into a nodule), and inflammation (such as in thyroiditis).[36][37][38] Autonomously functioning nodules, particularly hot ones, carry a risk of progressing to overt hyperthyroidism, known as toxic nodules, due to unchecked hormone secretion that suppresses TSH and leads to thyrotoxicosis. This autonomy often stems from somatic mutations in TSH receptor genes, allowing nodules to grow and function independently, with approximately 5-10% of longstanding nodules developing this feature over time. Evaluation of nodules for malignancy potential relies on clinical and imaging characteristics, such as size greater than 4 cm, which raises concern regardless of cytology in some guidelines, and microcalcifications, indicative of possible psammoma bodies or dystrophic changes. Other worrisome features include irregular margins and hypoechogenicity on ultrasound, prompting further assessment to differentiate benign from suspicious lesions.[39][40][41] Historically, goiter prevalence was dramatically reduced through public health initiatives like salt iodization programs, which began in the early 20th century. In the United States, mandatory iodization of table salt introduced in 1924 led to a sharp decline in endemic goiter rates, from over 40% in some regions to less than 5% within decades, serving as a model for global efforts that have prevented millions of cases worldwide.[42][43]Thyroid cancer
Thyroid cancer refers to a group of malignancies originating in the thyroid gland, with differentiated thyroid cancers comprising the majority of cases and generally carrying a favorable prognosis. Papillary thyroid carcinoma is the most common subtype, accounting for about 80% to 85% of all thyroid cancers, and it typically grows slowly with a high cure rate following treatment. Follicular thyroid carcinoma, representing 10% to 15% of cases, arises from follicular cells and shares similar indolent behavior but may more readily spread via the bloodstream to distant sites like the lungs or bones. Medullary thyroid carcinoma, which originates from parafollicular C cells, makes up 2% to 5% of thyroid cancers and can be sporadic or hereditary. Anaplastic thyroid carcinoma, although rare (less than 2% of cases), is extremely aggressive, often presenting in older adults and leading to rapid progression and poor outcomes.[44][45] Key risk factors for thyroid cancer include exposure to ionizing radiation, especially during childhood, which is strongly linked to the development of papillary and follicular subtypes through mechanisms like DNA damage in thyroid cells. For medullary thyroid carcinoma, genetic predisposition plays a prominent role, with germline mutations in the RET proto-oncogene causing up to 25% of cases, often in association with multiple endocrine neoplasia type 2 (MEN2) syndromes or familial medullary thyroid cancer. Other factors, such as female sex and certain genetic syndromes, modestly elevate risk across subtypes, but radiation and RET mutations remain the most established.[46][47][45] Staging for thyroid cancer employs the American Joint Committee on Cancer (AJCC) TNM system, which evaluates primary tumor size and extent (T), regional lymph node involvement (N), and distant metastasis (M), with age-specific adjustments for differentiated cancers and separate schemas for medullary and anaplastic types. Lymph node metastasis, particularly in the central neck, occurs in up to 50% of papillary cases at diagnosis and upregulates the N stage, influencing surgical extent and recurrence risk. Prognosis varies markedly by histology: differentiated thyroid cancers (papillary and follicular) have five-year relative survival rates exceeding 98% for localized disease and over 90% overall, reflecting effective therapies and indolent biology. Medullary thyroid cancer survival is around 80% to 90% at five years for localized tumors, while anaplastic carcinoma has a median survival of months, with five-year rates below 10%.[48][45][49] The incidence of thyroid cancer has risen steadily since the 1990s, tripling in some populations, primarily due to overdiagnosis of subclinical papillary microcarcinomas detected incidentally via ultrasound and other imaging during evaluations for unrelated conditions. Notable therapeutic advances include FDA approvals in 2020 for targeted tyrosine kinase inhibitors such as selpercatinib and pralsetinib, which specifically inhibit RET alterations in advanced or refractory medullary and differentiated thyroid cancers, improving progression-free survival in these challenging cases. The 2025 American Thyroid Association Management Guidelines for Adult Patients with Differentiated Thyroid Cancer provide updated recommendations based on recent advances in molecular causes and treatment options.[50][51][45][52]Drug-induced thyroid dysfunction
Drug-induced thyroid dysfunction encompasses a range of hypo- and hyperthyroid states triggered by medications or toxins that interfere with thyroid hormone synthesis, release, or metabolism. These disruptions often stem from the drug's direct effects on thyroid follicular cells, iodine handling, or autoimmune modulation, affecting up to 20% of patients on certain long-term therapies. Unlike primary thyroid disorders, iatrogenic cases are typically reversible upon drug discontinuation, though monitoring is essential to prevent complications such as arrhythmias or myxedema.[53] Amiodarone, an antiarrhythmic agent rich in iodine, frequently induces thyroid abnormalities due to its high iodine load (37% by weight) and inhibition of deiodinase enzymes, leading to altered T4-to-T3 conversion. Amiodarone-induced hypothyroidism (AIH) occurs in 5-20% of treated patients, particularly in iodine-sufficient regions, presenting with elevated TSH and low free T4 levels; it is more common in women and those with preexisting autoimmune thyroiditis. Amiodarone-induced thyrotoxicosis (AIT), seen in 2-10% of cases, is classified into type 1 (iodine-induced hyperthyroidism in nodular glands) and type 2 (destructive thyroiditis from direct toxicity), with type 2 predominating in iodine-replete areas. Diagnosis involves color-flow Doppler ultrasound to differentiate types, and treatment may require drug cessation or thyroidectomy in severe AIT.[54][55][56] Lithium, used in bipolar disorder management, impairs thyroid hormone release and iodination, resulting in hypothyroidism in 10-20% of long-term users, often subclinical with goiter formation. This effect is mediated by lithium's interference with inositol signaling and TSH receptor expression, increasing the risk in women over 50 and those with positive antithyroid antibodies. Hyperthyroidism is rarer (less than 5%), but goiter prevalence can reach 40-50% after years of therapy. Baseline TSH screening and monitoring every 6-12 months are recommended, with levothyroxine supplementation for overt cases.[57][58][59] Interferon-alpha (IFN-α), employed in hepatitis C and malignancy treatment, triggers thyroid autoimmunity by upregulating MHC class I expression and cytokine production, leading to destructive thyroiditis or Graves' disease in 5-15% of patients. Up to 40% develop thyroid antibodies during therapy, with hypothyroidism being the most common outcome (10%), though hyperthyroidism occurs in 3-5%. Risk factors include preexisting autoimmunity and female sex; prospective studies show peak incidence within 6 months of initiation. Discontinuation of IFN-α often resolves dysfunction, but persistent autoimmunity may require long-term levothyroxine.[60][61][62] Immune checkpoint inhibitors (ICIs), such as PD-1 inhibitors (e.g., nivolumab, pembrolizumab), have surged in use for cancer immunotherapy since 2015, causing thyroid immune-related adverse events (irAEs) in 10-20% of recipients through T-cell mediated thyroiditis. Hypothyroidism develops in 10-15%, often following a transient hyperthyroid phase from follicular destruction, while isolated hyperthyroidism affects 5-10%; combination ICI therapy elevates risks to 20-30%. These events typically onset within 3-6 months and correlate with better oncologic outcomes, possibly indicating robust immune activation. Management involves baseline TSH/thyroid antibody testing and monitoring every 4-6 weeks during therapy.[63][64][65] Iodine-containing agents, including radiographic contrast media and antiseptics like povidone-iodine, can precipitate thyroid dysfunction via excess iodide load. The Wolff-Chaikoff effect, a protective inhibition of thyroid hormone synthesis, causes transient hypothyroidism in 1-5% of exposed individuals, particularly those with underlying thyroid disease or neonates; it usually resolves within 48 hours via "escape" mechanisms. In contrast, the Jod-Basedow phenomenon induces hyperthyroidism in iodine-deficient patients with nodular goiters, occurring in up to 10% after high-dose exposure, due to unchecked hormone synthesis post-adaptation failure. Pre-exposure screening is advised for at-risk groups, such as those with multinodular goiter.[66][67][68] Antithyroid drugs like methimazole (MMI), used to treat hyperthyroidism, paradoxically cause agranulocytosis—a severe neutropenia—in 0.2-0.5% of patients, typically within the first 3 months of therapy. This idiosyncratic reaction involves immune-mediated bone marrow suppression, presenting with fever, sore throat, or infection; absolute neutrophil count below 500/μL confirms diagnosis. Immediate MMI discontinuation and granulocyte colony-stimulating factor (G-CSF) support recovery, which occurs in most cases within 1-2 weeks, though rechallenge is contraindicated. Patient education on symptom vigilance is a key monitoring strategy.[69][70][71] Tyrosine kinase inhibitors (TKIs), such as sunitinib and lenvatinib in cancer therapy, disrupt thyroid function primarily through vascular endothelial growth factor (VEGF) inhibition, causing hypothyroidism in 20-50% of users by impairing thyroid perfusion and hormone synthesis. Hyperthyroidism is less common (5-10%), often subclinical. Onset averages 2-6 months into treatment, with higher risks for multi-targeted TKIs; thyroid function worsens with prolonged use. Dose-dependent effects necessitate TSH monitoring at baseline, monthly during initiation, and every 3 months thereafter, with levothyroxine replacement as needed.[72][73][74] Recovery from drug-induced thyroid dysfunction varies by agent: amiodarone and lithium effects often reverse within 3-6 months post-discontinuation, though AIT type 2 may persist longer; ICI-related hypothyroidism is frequently permanent in 50-70% of cases due to glandular destruction. Guidelines from the American Thyroid Association recommend baseline thyroid function tests for high-risk drugs, followed by TSH assessments every 3-6 months or upon symptoms like fatigue or palpitations. In cancer patients on TKIs or ICIs, endocrine consultation is advised for grade 2+ toxicities to balance oncologic benefits against thyroid risks.[59][75][76]Pathophysiology
Autoimmune processes
Autoimmune processes in thyroid disease primarily involve dysregulation of the immune system, leading to the production of autoantibodies and infiltration of inflammatory cells into the thyroid gland, which disrupts normal thyroid function. These mechanisms are central to conditions such as Hashimoto's thyroiditis and Graves' disease, where T-cell mediated cytotoxicity and antibody responses target thyroid-specific antigens like thyroglobulin (Tg), thyroid peroxidase (TPO), and the thyrotropin receptor (TSHR).[77] The interplay of genetic predisposition and environmental triggers initiates a breakdown in immune tolerance, resulting in chronic inflammation and glandular damage.[78] Hashimoto's thyroiditis, the most common cause of autoimmune hypothyroidism, is characterized by the presence of anti-TPO antibodies (detected in over 90% of affected individuals) and anti-Tg antibodies (detected in 50–60% of affected individuals), which contribute to thyroid follicular cell destruction through antibody-dependent cellular cytotoxicity and complement activation.[79] Lymphocytic infiltration, predominantly involving CD4+ and CD8+ T cells, forms germinal centers within the thyroid stroma, leading to progressive fibrosis and atrophy of thyroid tissue.[80] This cell- and antibody-mediated immune attack destroys thyroid follicular cells, impairing hormone synthesis and release.[78] Graves' disease, in contrast, features autoantibodies targeting the TSH receptor, including stimulating TSH receptor antibodies (TSAb) that mimic TSH action, binding to the receptor's conformational site and inducing cyclic AMP production to drive excessive thyroid hormone synthesis and glandular hyperplasia.[81] Blocking TSH receptor antibodies (TBAb) inhibit TSH binding and can lead to hypothyroidism in some cases, while neutral antibodies bind without functional effect, though their role remains under investigation.[82] These antibodies arise from B-cell activation and T-cell help, perpetuating hyperthyroidism through sustained receptor stimulation.[83] Atrophic thyroiditis represents a variant of Hashimoto's thyroiditis, distinguished by marked thyroid gland atrophy and minimal enlargement, often progressing from the goitrous form due to extensive fibrosis following chronic lymphocytic infiltration.[84] Similar to Hashimoto's, it involves anti-TPO and anti-Tg antibodies, with T-cell mediated injury via cytokines like IL-17 from Th17 cells contributing to follicular cell pyroptosis and glandular shrinkage.[84] In some cases, IgG4-positive plasma cells are prominent, linking it to IgG4-related disease and accelerated hypothyroidism.[84] Genetic factors play a crucial role in susceptibility to autoimmune thyroid diseases, with associations to human leukocyte antigen (HLA) alleles such as HLA-DR3 and HLA-DR4, which influence antigen presentation and T-cell recognition of thyroid autoantigens.[85] Polymorphisms in the CTLA-4 gene, particularly the +49A/G variant, impair T-cell regulation by reducing inhibitory signaling, thereby promoting autoreactive lymphocyte proliferation and increasing risk for both Hashimoto's and Graves' disease.[86] These genetic elements, accounting for up to 50% of heritability, interact with environmental factors to initiate autoimmunity.[87] Postpartum thyroiditis is an autoimmune condition occurring within the first year after delivery, often in women with preexisting thyroid autoantibodies, featuring an initial destructive thyrotoxic phase due to rapid release of preformed hormones from inflamed follicles, followed by a hypothyroid phase from depleted glandular reserves.[88] The thyrotoxicosis typically emerges 1-3 months postpartum and lasts 1-3 months, driven by lymphocytic infiltration similar to subacute thyroiditis, while hypothyroidism develops 3-6 months later, persisting for 4-6 months in many cases.[89] This biphasic pattern reflects transient immune rebound post-pregnancy immunosuppression.[90] Autoimmune thyroid diseases frequently overlap with other autoimmune disorders, such as type 1 diabetes, where shared genetic loci like HLA-DR and CTLA-4 contribute to polyautoimmunity, with up to 30% of type 1 diabetes patients developing thyroid autoimmunity.[91] This clustering forms autoimmune polyendocrinopathy syndromes, highlighting common pathways in immune dysregulation.[92]Nutritional and environmental factors
Nutritional deficiencies play a significant role in thyroid pathology, particularly through inadequate intake of essential micronutrients required for hormone synthesis and metabolism. Iodine deficiency remains a primary cause of endemic goiter, where thyroid enlargement affects populations in iodine-poor regions, often leading to hypothyroidism due to insufficient thyroid hormone production despite compensatory glandular hypertrophy.[93] In severe cases, this deficiency impairs neurodevelopment and increases susceptibility to other thyroid disorders. Conversely, excess iodine intake, such as from supplements or iodized products in previously deficient areas, can precipitate hyperthyroidism, known as Jod-Basedow phenomenon, by overwhelming the thyroid's regulatory mechanisms and inducing autonomous hormone release.[94][66] Selenium deficiency further disrupts thyroid function by inhibiting the activity of iodothyronine deiodinases, enzymes critical for converting thyroxine (T4) to the active triiodothyronine (T3). This impairment reduces peripheral thyroid hormone availability, potentially exacerbating hypothyroidism in selenium-poor soils common in certain agricultural areas.[95][96] Environmental contaminants like perchlorate and thiocyanates, found in water supplies and tobacco smoke respectively, competitively block iodine uptake by the sodium-iodide symporter in the thyroid, mimicking iodine deficiency and elevating the risk of hypothyroidism even in iodine-replete settings.[97][98] Radiation exposure, particularly from nuclear accidents, heightens thyroid cancer risk in children due to the thyroid's sensitivity to ionizing radiation during development. For instance, post-Chernobyl studies show elevated thyroid cancer incidence among those exposed under age 5, with risks persisting decades later.[99][100] Dietary goitrogens, such as glucosinolates in cruciferous vegetables (e.g., broccoli, cabbage) and cyanogenic compounds in cassava, interfere with iodine organification and thyroid hormone synthesis, contributing to goiter formation when consumed in large amounts without adequate iodine.[101] Cooking mitigates much of this effect, but chronic high intake in staple-based diets can sustain thyroid enlargement.[102] Geographic and climatic factors influence thyroid disease prevalence through variations in soil nutrient content and environmental exposures. Inland and high-altitude regions often exhibit higher rates of iodine deficiency disorders due to lower iodine bioavailability in water and crops, while coastal areas benefit from marine iodine sources but may face excess from iodized salt programs.[102] Tropical climates exacerbate issues via low-iodine staples like cassava and millet, leading to endemic goiter in affected populations.[103][104]Thyroid dysfunction in pregnancy
During pregnancy, estrogen levels rise significantly, leading to an increase in thyroxine-binding globulin (TBG) concentrations, which elevates total thyroxine (T4) levels while free T4 remains stable.[105] This physiological adaptation necessitates a 25-50% increase in maternal T4 production to maintain euthyroid free hormone levels and support fetal development.[106] The thyroid gland's response involves heightened responsiveness to thyroid-stimulating hormone (TSH), ensuring adequate hormone synthesis amid increased renal clearance and distribution volume.[107] Hypothyroidism during pregnancy, whether overt or subclinical, poses substantial risks to maternal and fetal health. Untreated cases are associated with adverse outcomes including preeclampsia, miscarriage, preterm birth, placental abruption, and postpartum hemorrhage.[108] Fetal implications extend to neurodevelopmental deficits, such as reduced IQ and impaired cognitive function, due to insufficient thyroid hormone availability during critical brain development phases.[107] These risks underscore the importance of early detection and management to mitigate long-term consequences.[109] Hyperthyroidism in pregnancy affects approximately 0.2-1.0% of cases, predominantly due to Graves' disease, which may flare in the first trimester before improving in the third due to immune modulation and high-output heart failure risks.[110] Maternal complications include thyroid storm, heart failure, and exacerbated symptoms like tachycardia.[111] A distinct entity, transient gestational thyrotoxicosis, arises from high human chorionic gonadotropin (hCG) levels stimulating the TSH receptor, typically resolving by 14-20 weeks without antithyroid antibodies.[112] Differentiation from Graves' relies on clinical history and antibody testing to guide appropriate intervention.[109] Postpartum thyroiditis, an autoimmune-mediated inflammation, occurs in 5-10% of women in the United States, often presenting with a hyperthyroid phase followed by hypothyroidism.[113] Incidence is higher in those with preexisting thyroid peroxidase antibodies or type 1 diabetes, reflecting underlying autoimmune predisposition.[114] Most cases resolve within 12-18 months, though up to 20-30% may progress to permanent hypothyroidism.[88] Screening for thyroid dysfunction is recommended via serum TSH measurement in the first trimester for high-risk women, including those with a history of thyroid disease, autoimmune disorders, or infertility.[105] The American College of Obstetricians and Gynecologists (ACOG) advises against universal screening but supports targeted testing, with TSH reference ranges adjusted lower in pregnancy (e.g., 0.1-2.5 mIU/L in the first trimester).[115] The American Thyroid Association (ATA) guidelines similarly emphasize trimester-specific thresholds to detect subclinical hypothyroidism early.[116] Fetal thyroid development begins around 12 weeks of gestation, when the gland starts producing thyroid hormones, though full maturation occurs near term and relies initially on maternal transfer of T4 across the placenta.[117] Maternal thyroid-stimulating hormone receptor antibodies (TRAb) can cross the placenta, potentially inducing fetal hyperthyroidism in Graves' disease cases, with monitoring via ultrasound for goiter or tachycardia.[118] This transplacental antibody transfer highlights the interplay between maternal autoimmune status and fetal thyroid function.[119]Diagnosis
Blood tests
Blood tests are essential for evaluating thyroid function and diagnosing thyroid diseases by measuring hormone levels and related markers in the serum. The thyroid-stimulating hormone (TSH) assay serves as the primary screening test due to its high sensitivity in detecting both primary and secondary hypothyroidism or hyperthyroidism. In primary thyroid disorders, TSH levels inversely reflect thyroid hormone production, while elevated TSH with low free thyroxine (FT4) indicates secondary hypothyroidism from pituitary dysfunction.[120] Free T4 and free triiodothyronine (FT3) measurements provide direct assessments of active thyroid hormones available to tissues, distinguishing them from total T4 and total T3, which include hormone bound to carrier proteins like thyroxine-binding globulin and may be influenced by factors such as pregnancy or estrogen use.[121] Free hormone assays are preferred for accuracy in most clinical scenarios, as total levels can fluctuate with binding protein alterations without reflecting true thyroid function.[122] Abnormal TSH prompts FT4 testing to confirm overt disease, with FT3 added if hyperthyroidism is suspected, as it correlates with symptoms like tachycardia.[121] Antithyroid antibodies help identify autoimmune etiologies of thyroid dysfunction. Thyroid peroxidase antibodies (anti-TPO) are the most common and strongly associated with Hashimoto's thyroiditis, present in over 90% of cases, while thyroglobulin antibodies (anti-Tg) occur in about 60% and aid in confirming autoimmune hypothyroidism.[123] TSH receptor antibodies (TRAb), particularly thyroid-stimulating immunoglobulins, are specific for Graves' disease, detectable in 80-90% of patients and correlating with disease severity.[33] These tests are recommended when autoimmune thyroiditis is suspected based on clinical features or elevated TSH.[77] Serum calcitonin measurement is used for screening medullary thyroid carcinoma (MTC), particularly in patients with family history of multiple endocrine neoplasia type 2 or thyroid nodules suspicious for MTC. Basal calcitonin levels >100 pg/mL are highly suggestive of MTC or C-cell hyperplasia with high specificity.[124] Thyroglobulin serves as a tumor marker for monitoring differentiated thyroid cancer recurrence after thyroidectomy and radioactive iodine ablation. Undetectable thyroglobulin levels post-treatment predict excellent response, while detectable levels (e.g., ≥0.2 ng/mL post-ablation) or rising levels suggest persistent or recurrent disease, though antithyroglobulin antibodies can interfere and require concurrent monitoring.[125] Reference ranges for thyroid function tests vary by age and physiological state to avoid misdiagnosis. In older adults, TSH upper limits may extend to 4.5-6.0 mIU/L due to age-related changes, reducing overdiagnosis of subclinical hypothyroidism.[126] During pregnancy, trimester-specific ranges are critical; for instance, first-trimester TSH upper limit is approximately 2.5 mIU/L, reflecting hCG-mediated TSH suppression, with FT4 ranges adjusted for method-specific assays.[116] These adjustments ensure accurate interpretation in contexts like subclinical hypothyroidism, where mild TSH elevations may indicate early disease.Imaging modalities
Imaging modalities play a crucial role in the evaluation of thyroid disease by providing structural and functional information to guide diagnosis and management. These non-invasive techniques help characterize thyroid nodules, assess glandular function, and detect extensions or metastases, often integrated with blood test results such as thyroid-stimulating hormone levels to determine the need for further imaging.[127] Ultrasound is the initial imaging modality of choice for evaluating thyroid nodules due to its accessibility, lack of radiation, and ability to assess multiple features. It characterizes nodules based on composition (solid, cystic, or mixed), echogenicity, margins, shape, calcifications, and vascularity using Doppler to evaluate blood flow patterns, which can indicate benign or suspicious features.[127] The American College of Radiology Thyroid Imaging Reporting and Data System (ACR TI-RADS) standardizes risk stratification by assigning points to ultrasound features—such as composition, echogenicity, shape, margins, and echogenic foci—to categorize nodules into risk levels (TR1 to TR5), recommending fine-needle aspiration for higher-risk nodules greater than specific size thresholds (e.g., TR5 nodules ≥1 cm).[128] Ultrasound also evaluates cervical lymph nodes for suspicious characteristics like loss of fatty hilum, hyperechogenicity, or irregular borders, aiding in the detection of regional spread in thyroid cancer.[127] Nuclear medicine scans provide functional assessment of the thyroid gland. Radioiodine uptake scans, using iodine-123 or iodine-131, measure thyroidal iodine avidity and distinguish hyperfunctioning "hot" nodules (rarely malignant, with 96-99% negative predictive value for cancer) from nonfunctioning "cold" nodules (80-90% benign but requiring further evaluation).[129] In Graves' disease, these scans typically show diffuse increased uptake throughout the gland, confirming autonomous hyperfunction.[129] Scintigraphy with technetium-99m pertechnetate offers a comparable functional evaluation with advantages of lower radiation dose, shorter imaging time (within 30 minutes post-injection), and wider availability, particularly useful for assessing multinodular goiter or nodule autonomy.[129][130] Computed tomography (CT) and magnetic resonance imaging (MRI) are employed for advanced structural evaluation, particularly when ultrasound is limited. CT excels in delineating retrosternal goiter extension into the mediastinum or assessing local invasion in larger masses, providing detailed cross-sectional images for preoperative planning.[127] MRI offers superior soft-tissue contrast without ionizing radiation, making it preferable for evaluating invasion into adjacent structures like the trachea or esophagus in suspected malignancy.[127] Positron emission tomography-computed tomography (PET-CT) using 18F-fluorodeoxyglucose (FDG) has become a standard for detecting metastatic differentiated thyroid cancer since the 2010s, especially in cases with elevated thyroglobulin but negative radioiodine scans, identifying non-iodine-avid lesions with high sensitivity (up to 89%).[127][131] It is recommended by guidelines for staging persistent or recurrent disease and guiding therapy in advanced cases.[132] A key limitation of radioiodine uptake scans, scintigraphy, CT, and PET-CT is radiation exposure, which necessitates careful consideration in vulnerable populations such as pregnant individuals, where these modalities are contraindicated unless essential.[129][127]Biopsy procedures
Biopsy procedures are essential invasive methods for obtaining tissue samples from thyroid lesions, particularly nodules, to confirm or rule out malignancy when imaging and blood tests suggest concern. These procedures primarily involve fine-needle aspiration (FNA), which is the gold standard for cytological evaluation, often performed under ultrasound guidance to target suspicious areas accurately.[133][127] Fine-needle aspiration (FNA) entails inserting a thin needle (typically 25-27 gauge) into the thyroid nodule to aspirate cells for cytological analysis, usually requiring multiple passes for adequate sampling. Indications for FNA include thyroid nodules greater than 1 cm in size with high-suspicion ultrasound features, such as hypoechoic solid composition, irregular margins, microcalcifications, or taller-than-wide shape, as these carry a higher risk of malignancy. For nodules with intermediate suspicion, FNA is recommended if the nodule is at least 1 cm; low-suspicion nodules may warrant FNA if 1.5 cm or larger, while very low-suspicion spongiform nodules typically require evaluation only if exceeding 2 cm.[127] The procedure is outpatient, quick (under 30 minutes), and performed by endocrinologists, radiologists, or cytopathologists trained in the technique.[133] FNA results are classified using the Bethesda System for Reporting Thyroid Cytopathology, a standardized six-category framework that stratifies malignancy risk to guide management. The system, endorsed by major thyroid organizations, categorizes samples as follows:| Category | Description | Implied Risk of Malignancy (%) |
|---|---|---|
| I | Nondiagnostic or unsatisfactory | 1–4 |
| II | Benign | 0–3 |
| III | Atypia of undetermined significance (AUS) or Follicular lesion of undetermined significance (FLUS) | 5–15 |
| IV | Follicular neoplasm or Suspicious for follicular neoplasm | 15–30 |
| V | Suspicious for malignancy | 60–75 |
| VI | Malignant | 97–99 |

