Lactococcus
View on Wikipedia
| Lactococcus | |
|---|---|
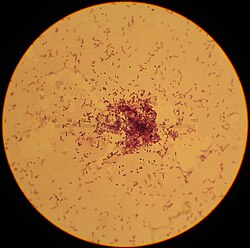
| |
| Lactococcus lactis | |
| Scientific classification | |
| Domain: | Bacteria |
| Kingdom: | Bacillati |
| Phylum: | Bacillota |
| Class: | Bacilli |
| Order: | Lactobacillales |
| Family: | Streptococcaceae |
| Genus: | Lactococcus Schleifer et al. 1986 |
| Species | |
|
L. allomyrinae | |
Lactococcus, from Latin lac, meaning "milk", and Ancient Greek κόκκος (kókkos), meaning "berry", is a genus of lactic acid bacteria that were formerly included in the genus Streptococcus Group N1.[2] They are known as homofermenters meaning that they produce a single product, lactic acid in this case, as the major or only product of glucose fermentation. Their homofermentative character can be altered by adjusting environmental conditions such as pH, glucose concentration, and nutrient limitation. They are gram-positive, catalase-negative, non-motile cocci that are found singly, in pairs, or in chains. The genus contains strains known to grow at or below 7˚C.[3]
Twelve species of Lactococcus are currently recognized.[4] They are:
- Lactococcus allomyrinae
- Lactococcus carnosus
- Lactococcus chungangensis
- Lactococcus cremoris
- L. cremoris subsp. cremoris
- L. cremoris subsp. tructae
- Lactococcus formosensis
- L. formosensis subsp. bovis
- L. formosensis subsp. formosensis
- Lactococcus fujiensis
- Lactococcus garvieae
- L. garvieae subsp. garvieae
- L. garvieae subsp. bovis
- Lactococcus hircilactis
- Lactococcus hodotermopsidis
- Lactococcus insecticola
- Lactococcus kimchii
- Lactococcus lactis
- L. lactis subsp. hordniae
- L. lactis subsp. lactis
- Lactococcus laudensis
- Lactococcus nasutitermitis
- Lactococcus paracarnosus
- Lactococcus petauri
- Lactococcus piscium
- Lactococcus plantarum
- Lactococcus protaetiae
- Lactococcus raffinolactis
- Lactococcus reticulitermitis
- Lactococcus taiwanensis
- Lactococcus termiticola
These organisms are commonly used in the dairy industry in the manufacture of fermented dairy products such as cheeses. They can be used in single-strain starter cultures, or in mixed-strain cultures with other lactic acid bacteria such as Lactobacillus and Streptococcus. Special interest is placed on the study of L. lactis subsp. lactis and L. lactis subsp. cremoris, as they are the strains used as starter cultures in industrial dairy fermentations.[5] Their main purpose in dairy production is the rapid acidification of milk; this causes a drop in the pH of the fermented product, which prevents the growth of spoilage bacteria. The bacteria also play a role in the flavor of the final product.[6] Lactococci are currently being used in the biotechnology industry. They are easily grown at industrial scale on whey-based media. As food-grade bacteria, they are used in the production of foreign proteins that are applied to the food industry.
Diseases
[edit]Lactococcosis refers to a group of disorders caused by the bacterium L. garvieae. Most "Lacto" species dwell on the bodies of humans and animals, and while they do not cause serious problems in higher animals, they do cause chronic illnesses in lower animals, particularly fish.[7][8]
References
[edit]- ^ a b c Parte, A.C. "Lactococcus". LPSN.
- ^ Schleifer KH, Kraus J, Dvorak C, Kilpper-Balz R, Collins MD, Fischer W (1985). "Transfer of Streptococcus lactis and related streptococci to the genus Lactococcus gen. nov". Syst. Appl. Microbiol. 6 (2): 183–195. Bibcode:1985SyApM...6..183S. doi:10.1016/s0723-2020(85)80052-7. ISSN 0723-2020.
- ^ James M. Modern Food Microbiology, 4th ed. Van Nostrand Reinhold, 1992
- ^ LPSN entry for Lactococcus
- ^ de Vos, W.M. Simons, GFM. Genetics and Biotechnology of Lactic Acid Bacteria. eds. Gasson, M.J. de Vos, W.M. Gene Cloning and Expression Systems in Lactococci. Blackie Academic & Professional, 1994. pg. 52 – 53.
- ^ Kok, Jan. Genetics and Molecular Biology of Streptococci, Lactococci, and Enterococci.. eds Dunny, Gary M. Cleary, P. Patrick. McKay, Larry L. Special Purpose Vectors for Lactococci. American Society for Microbiology, 1991. pg. 97
- ^ Rao, S, Pham, TH, Poudyal, S, et al. First report on genetic characterization, cell-surface properties and pathogenicity of Lactococcus garvieae, emerging pathogen isolated from cage-cultured cobia (Rachycentron canadum). Transbound Emerg Dis. 2022; 69: 1197– 1211. https://doi.org/10.1111/tbed.14083
- ^ Rao, S, Chen, M-Y, Sudpraseart, C, et al. Genotyping and phenotyping of Lactococcus garvieae isolates from fish by pulse-field gel electrophoresis (PFGE) and electron microscopy indicate geographical and capsular variations. J Fish Dis. 2022; 45: 771– 781. https://doi.org/10.1111/jfd.13601
