Recent from talks
Nothing was collected or created yet.
Pamidronic acid
View on Wikipedia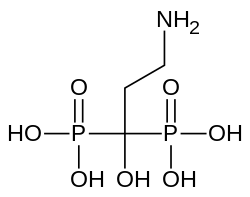 | |
| Clinical data | |
|---|---|
| Trade names | Aredia, Pamimed, among others |
| Other names | Pamidronate disodium pentahydrate, pamidronate disodium |
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a601163 |
| Pregnancy category |
|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | n/a |
| Protein binding | 54% |
| Metabolism | Nil |
| Elimination half-life | 28 ± 7 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.049.897 |
| Chemical and physical data | |
| Formula | C3H11NO7P2 |
| Molar mass | 235.069 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Pamidronic acid or pamidronate disodium or APD (marketed as Aredia among others), is a nitrogen-containing bisphosphonate used to prevent osteoporosis.
It was patented in 1971 and approved for medical use in 1987.[1]
Medical uses
[edit]It is used to prevent bone loss, and treat osteoporosis. It is also used to strengthen bone in Paget's disease, to prevent bone loss due to steroid use, and in certain cancers with high propensity to bone, such as multiple myeloma. Due to its ability to sequester calcium in bone, it is also used to treat high calcium levels. It is also used as an experimental treatment of the bone disorder osteogenesis imperfecta. It has been studied in the treatment of complex regional pain syndrome.[2]
Administration
[edit]Intravenous, usually 90 mg monthly. 30 mg, 60 mg, 90 mg and for hospitals, 120 mg vials are available, mixed with mannitol.
Side effects
[edit]Common side effects include bone pain, low calcium levels, nausea, and dizziness. Osteonecrosis of the jaw is a rare complication which has been associated with the use of bisphosphonates, including pamidronate.[3]
Pamidronate activates human γδ T cells in vitro and in vivo, which may lead to flu-like symptoms upon administration.
Pharmacology
[edit]| Bisphosphonate | Relative potency |
|---|---|
| Etidronate | 1 |
| Tiludronate | 10 |
| Pamidronate | 100 |
| Alendronate | 100-500 |
| Ibandronate | 500-1000 |
| Risedronate | 1000 |
| Zoledronate | 5000 |
References
[edit]- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 523. ISBN 9783527607495.
- ^ Kubalek I, Fain O, Paries J, Kettaneh A, Thomas M (December 2001). "Treatment of reflex sympathetic dystrophy with pamidronate: 29 cases". Rheumatology. 40 (12): 1394–1397. doi:10.1093/rheumatology/40.12.1394. PMID 11752511.
- ^ Zarychanski R, Elphee E, Walton P, Johnston J (January 2006). "Osteonecrosis of the jaw associated with pamidronate therapy". American Journal of Hematology. 81 (1): 73–75. doi:10.1002/ajh.20481. PMID 16369966. S2CID 11830192.
- ^ Tripathi KD (2013-09-30). Essentials of medical pharmacology (Seventh ed.). New Delhi: Jaypee Brothers Medical Publishers Pvt. Limited. ISBN 9789350259375. OCLC 868299888.
Pamidronic acid
View on GrokipediaChemistry
Structure
Pamidronic acid, with the molecular formula C₃H₁₁NO₇P₂, has a molar mass of 235.07 g/mol.[4] Its IUPAC name is (3-amino-1-hydroxy-1-phosphonopropyl)phosphonic acid.[4] As a second-generation nitrogen-containing bisphosphonate, pamidronic acid features a geminal bisphosphonate backbone characterized by a non-hydrolyzable P-C-P linkage, which mimics the structure of pyrophosphate and distinguishes it from first-generation non-nitrogen bisphosphonates like etidronate.[1][5] The core structure consists of a central carbon atom at the 1-position bonded to two phosphonic acid groups, a hydroxyl substituent at the same carbon, and a primary amino group attached to the 3-position of the propane chain.[4][6] In its three-dimensional conformation, pamidronic acid exhibits flexibility in the propane chain, with nuclear magnetic resonance studies indicating a preference for gauche conformations over trans due to intramolecular hydrogen bonding involving the amino and hydroxyl groups, which influences its overall molecular shape.[7] The two phosphonate groups (-PO₃H₂) serve as the primary functional moieties, enabling strong chelation and binding to calcium ions in hydroxyapatite, the main mineral component of bone, thereby conferring its characteristic bone affinity.[8][9]Properties
Pamidronic acid appears as a white to off-white crystalline powder.[10] It is poorly soluble in water (approximately 5 mg/mL with heating and sonication) and organic solvents such as methanol.[11][12] The disodium salt form, known as pamidronate disodium, is employed in pharmaceutical preparations to enhance solubility in aqueous solutions.[13] The compound possesses multiple ionizable groups, with pKa values of approximately 1.1 (first phosphonic acid), 6.0 (second phosphonic acid), and 10.0 (amino group), influencing its behavior in physiological environments.[7] Pamidronic acid has a LogP value of approximately -4.5, reflecting its highly hydrophilic nature.[1] Under normal conditions, it remains stable but is hygroscopic and sensitive to light and elevated temperatures; stability in solution is pH-dependent, with optimal conditions around neutrality.[12][13]Pharmacology
Mechanism of action
Pamidronic acid, a second-generation nitrogen-containing bisphosphonate, primarily inhibits bone resorption by selectively targeting osteoclasts, the cells responsible for bone breakdown. It binds with high affinity to hydroxyapatite crystals in bone, particularly at sites of active remodeling, where it is subsequently released during the acidification process of osteoclast resorption. Once internalized by osteoclasts through endocytosis or fluid-phase pinocytosis, pamidronic acid exerts its effects intracellularly, leading to detachment of osteoclasts from the bone surface by disrupting podosome assembly and the ruffled border formation essential for resorption.[14][1][15] At the molecular level, pamidronic acid inhibits farnesyl pyrophosphate synthase (FPPS), a key enzyme in the mevalonate pathway, which is crucial for the synthesis of isoprenoid lipids such as farnesyl pyrophosphate (FPP) and geranylgeranyl pyrophosphate (GGPP). This inhibition prevents the posttranslational prenylation of small GTP-binding proteins, including Ras, Rho, Rab, and Rac, which are vital for osteoclast survival, cytoskeletal organization, and vesicular trafficking. The disruption of protein prenylation accumulates upstream intermediates like isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP), further contributing to cellular dysfunction in osteoclasts. Unlike first-generation non-nitrogen bisphosphonates, the amino group in pamidronic acid's structure enhances its potency by 10- to 1000-fold, allowing lower doses for equivalent antiresorptive effects.[15][1][14] The cellular consequences include reduced osteoclast recruitment, impaired activity, and induction of apoptosis. By inhibiting prenylation, pamidronic acid disrupts osteoclast migration and adhesion to bone, limiting precursor maturation and fusion into mature osteoclasts. It activates apoptotic pathways, including caspase-3 and caspase-9, leading to programmed cell death specifically in osteoclasts, despite FPPS being present in other cell types; this selectivity arises from the drug's preferential uptake and accumulation in resorbing osteoclasts. As a nitrogen-containing bisphosphonate, pamidronic acid is not primarily metabolized to cytotoxic ATP analogs like ApppI in the same manner as simpler bisphosphonates, but its FPPS inhibition indirectly affects mitochondrial function through prenylation defects.[15][1][16] Pamidronic acid has no direct effects on osteoblasts, the bone-forming cells, but indirectly promotes net bone formation by suppressing osteoclast-mediated resorption, thereby uncoupling bone remodeling and allowing osteoblast activity to predominate. This results in increased bone mineral density without altering osteoblast proliferation or differentiation directly.[15][14]Pharmacokinetics
Pamidronic acid, marketed as pamidronate disodium, is administered exclusively by intravenous infusion owing to its negligible oral bioavailability of less than 1%.[17] After intravenous administration, pamidronate achieves immediate and complete systemic availability, with plasma concentrations rising rapidly during infusion and declining promptly upon cessation.[18] Distribution of pamidronate is characterized by rapid uptake into bone, where up to 50% of the administered dose binds preferentially to sites of active bone turnover, such as hydroxyapatite crystals. It reflects extensive tissue distribution beyond the plasma compartment, while plasma protein binding remains low at approximately 54%.[19][1][18] Pamidronate undergoes no metabolism in the body and exerts its effects as the unchanged parent compound.[18][1] Elimination is predominantly renal, with approximately 50% of the dose excreted unchanged in the urine within 24 hours and a cumulative 46 ± 16% by 120 hours; the mean elimination half-life is 28 ± 7 hours, extended by reversible bone binding that contributes to prolonged retention. Bone accumulation increases with repeated dosing due to this avid skeletal affinity.[18][1] Total plasma clearance averages 107 ± 50 mL/min, with renal clearance at 49 ± 28 mL/min, and both decline in renal impairment as excretion correlates closely with glomerular filtration rate; dose adjustments may be necessary to prevent excessive accumulation in such patients.[18][1]Medical uses
Indications
Pamidronic acid, also known as pamidronate disodium, is approved for the treatment of moderate to severe hypercalcemia associated with malignancy, with or without metastases to bone.[20] This indication targets patients who have not responded adequately to rehydration and who exhibit corrected serum calcium levels greater than 12 mg/dL. Clinical trials have demonstrated its efficacy in reducing serum calcium levels, with normalization achieved in 97% of patients receiving 60 mg over 24 hours and 100% with 90 mg over 24 hours by day 7 following treatment. Response times for pamidronate in treating hypercalcemia can vary widely depending on factors such as the severity of hypercalcemia, underlying cause, and individual patient response.[20][14] In a double-blind, randomized study comparing pamidronate to placebo, complete response rates (defined as normalization of serum calcium) reached approximately 70% by day 10 in the pamidronate group, significantly outperforming saline hydration alone.[21] It is also indicated for moderate to severe Paget's disease of bone in patients with elevated serum alkaline phosphatase levels at least twice the upper limit of normal or those at risk for complications such as fractures or hearing loss.[20] Pamidronic acid normalizes bone turnover markers, with a ≥50% decrease in serum alkaline phosphatase observed in 60% of patients and in urinary hydroxyproline/creatinine in 72% after a 90 mg total dose administered over three days.[20] This therapy is particularly selected for patients unresponsive to first-line treatments like calcitonin or those with symptomatic disease.[14] Additionally, pamidronic acid is approved, in conjunction with standard antineoplastic therapy, for the treatment of osteolytic bone metastases from breast cancer and osteolytic lesions from multiple myeloma.[20] In breast cancer patients, monthly 90 mg infusions reduced the skeletal morbidity rate from 3.7 to 2.5 events per year and the proportion experiencing skeletal-related events (such as pathological fractures or need for radiation) from 65% to 46% compared to placebo.[20] For multiple myeloma, it decreased the incidence of skeletal-related events to 24% at one year versus 41% with placebo.[20] These indications apply to patients with documented bone lesions confirmed by imaging and who are receiving concurrent cancer therapy.[20] In the European Union, approvals align closely, covering tumor-induced hypercalcemia, osteolytic bone metastases from breast cancer, and osteolytic lesions in advanced multiple myeloma.[22]Administration
Pamidronic acid, marketed as pamidronate disodium, is administered exclusively by intravenous infusion after dilution in a compatible fluid. It is supplied as a sterile lyophilized powder in vials containing 15 mg, 30 mg, or 90 mg, which must be reconstituted prior to use. Reconstitution is performed by adding 5 mL or 10 mL of sterile water for injection to the appropriate vial (depending on strength), yielding a concentration of 3 mg/mL or 6 mg/mL, respectively; the reconstituted solution is stable for up to 24 hours under refrigeration at 2°C to 8°C (36°F to 46°F). The solution is then further diluted in 250 to 1000 mL of 0.45% or 0.9% sodium chloride injection or 5% dextrose injection, and it remains stable for up to 24 hours at room temperature when protected from light. Pamidronate must not be mixed with calcium-containing infusion fluids, such as Ringer's solution, and a separate infusion line should be used if other medications are administered concurrently.[20][23] Dosage regimens are tailored to the specific indication and patient factors. For moderate hypercalcemia of malignancy (corrected serum calcium 12–13.5 mg/dL), a single 60–90 mg dose is recommended, while severe hypercalcemia (>13.5 mg/dL) typically requires 90 mg; retreatment may occur after at least 7 days if hypercalcemia recurs. For Paget's disease of bone, the standard course is 30 mg infused once daily for 3 consecutive days, totaling 90 mg, with retreatment possible after 6 months if disease activity persists. In osteolytic bone metastases from breast cancer, 90 mg is given every 3–4 weeks; for multiple myeloma with osteolytic lesions, the same dose is administered every 4 weeks. These doses should not exceed 90 mg per treatment to limit renal risk.[20][24] Infusions are conducted over 2–24 hours to reduce the potential for nephrotoxicity, with durations of at least 2 hours for 90 mg doses in bone metastasis indications and up to 4 hours for Paget's disease; slower rates (e.g., 24 hours) may be used for hypercalcemia if renal function is compromised. Prior to initiating therapy for Paget's disease or osteolytic lesions (in the absence of hypercalcemia), patients should receive oral calcium supplementation (e.g., 500 mg daily) and vitamin D (e.g., 400–800 international units daily) to minimize the risk of hypocalcemia. Serum electrolytes, including calcium, phosphate, magnesium, and potassium, should be monitored closely, particularly in the first 1–2 weeks after treatment initiation, with short-term supplementation provided if deficiencies occur; for hypercalcemia treatment, monitoring blood calcium levels is essential to assess response time and determine the need for retreatment.[23][25][20] Renal function must be assessed via serum creatinine measurement before each dose; treatment should be withheld if creatinine rises by more than 0.5 mg/dL above baseline in patients with normal function or more than 1 mg/dL above baseline in those with abnormal function prior to therapy. No specific dose adjustments are required for mild to moderate renal impairment (creatinine clearance ≥30 mL/min), but pamidronate is not recommended for patients with severe renal impairment (creatinine clearance <30 mL/min), particularly in bone metastasis treatment, due to the risk of further deterioration. Hydration with normal saline is essential before and after infusion for hypercalcemia to promote calciuresis, protect renal function, and influence response time by enhancing the drug's efficacy in normalizing calcium levels.[20][24][23]Adverse effects
Common adverse effects
Pamidronic acid commonly causes flu-like symptoms as part of an acute phase response, particularly following the initial infusion. These include fever (reported in up to 41% of patients), chills, myalgia (up to 26%), and arthralgia (up to 14%), with onset typically within 48 hours and resolution within 2-3 days.[26][20][14] These symptoms occur less frequently with subsequent doses due to the development of tolerance.[14] Gastrointestinal adverse effects are also prevalent, encompassing nausea (up to 54%), vomiting (up to 36%), and abdominal pain (up to 23%), generally mild and transient.[20][26] Musculoskeletal effects often involve bone pain (up to 67%) and joint pain, which may exacerbate at sites of underlying bone lesions and typically resolve within days.[20][26] Hematologic changes include mild anemia (up to 43%) and occasional lymphopenia, usually self-limiting and monitored during the first two weeks of treatment.[20][23][26] The incidence of these effects is dose-related, with higher rates observed at doses of 90 mg, and they often diminish over time with repeated administration.[26][20] Prophylaxis with acetaminophen or ibuprofen prior to infusion can reduce the occurrence and severity of flu-like symptoms.[27] Management generally involves supportive care, such as hydration and analgesics, with most effects resolving spontaneously.[14][26]Serious adverse effects
Pamidronic acid, administered intravenously, is associated with renal toxicity, manifesting as acute kidney injury or deterioration in renal function, particularly in patients with preexisting renal impairment or those receiving higher doses or shorter infusion times. This nephrotoxicity can present as toxic acute tubular necrosis or collapsing focal segmental glomerulosclerosis, with a cumulative risk increasing after repeated doses; monitoring of serum creatinine and creatinine clearance is essential before each infusion, and the drug should be withheld if creatinine rises more than 10% above baseline.[14][20][28] Hypocalcemia is another serious adverse effect, which may become symptomatic several days after infusion and include tetany, seizures, or cardiac arrhythmias due to reduced bone resorption leading to decreased serum calcium levels. Risk is heightened in patients with renal insufficiency or vitamin D deficiency, necessitating pre-treatment supplementation with calcium and vitamin D, along with ongoing electrolyte monitoring.[14][29][30] Osteonecrosis of the jaw (ONJ) occurs rarely, with reported incidences ranging from 0.1% to 1% in general use but increasing to 1.5–7.7% with prolonged intravenous therapy (over 12 months), particularly in cancer patients with bone metastases; it is often triggered by dental procedures or invasive oral surgery during long-term treatment. Dental evaluation and preventive care are recommended prior to initiating therapy in at-risk patients.[14][31][32] Atypical femoral fractures, characterized by subtrochanteric or diaphyseal location with minimal trauma, are very rare but have been documented in a small number of cases associated with pamidronic acid use, similar to other bisphosphonates, and may involve delayed healing.[14][33] Ocular effects, though infrequent, include scleritis, anterior uveitis, or episcleritis, which typically require discontinuation of the drug for resolution and have been linked to pamidronic acid in case reports with positive rechallenge.[34][35] These serious adverse effects are more prevalent in cancer patients receiving intravenous pamidronic acid for conditions like multiple myeloma or bone metastases, underscoring the need for vigilant monitoring and risk assessment.[14][31]Contraindications and precautions
Contraindications
Pamidronate disodium is contraindicated in patients with known hypersensitivity to pamidronate disodium, other bisphosphonates, or mannitol, as reactions including anaphylaxis have been reported.[20]Precautions
Use of pamidronate is not recommended in patients with severe renal impairment, defined as creatinine clearance less than 30 mL/min, particularly for indications such as bone metastases, due to increased risk of further renal deterioration; treatment should be withheld if renal function worsens during therapy.[20] Pamidronate can cause fetal harm. Based on findings from animal studies, it is expected to cause fetal harm when administered to pregnant women. There are no available data on pamidronate use in pregnant women to inform the drug-associated risk. Advise pregnant women and females of reproductive potential of the potential risk to the fetus and use effective contraception during and after treatment.[20] Breastfeeding should be avoided during treatment with pamidronate and afterward due to the potential for serious adverse reactions in nursing infants, as the drug binds to bone long-term and may be released over weeks to years.[20] Safety and effectiveness of pamidronate have not been established in pediatric patients and it is not approved for use in this population.[20] In elderly patients, no specific contraindications exist, but renal function should be monitored closely due to age-related decline in clearance, with dosing initiated at the lower end of the range.[20] Therapy with pamidronate should be avoided in patients with pre-existing hypocalcemia unless serum calcium levels are corrected prior to initiation, as the drug can exacerbate hypocalcemia; calcium and vitamin D supplementation is recommended as needed.[20] Patients should have a dental examination and appropriate preventive dentistry before treatment with pamidronate, and maintain good oral hygiene during therapy to minimize the risk of osteonecrosis of the jaw.[20]Drug interactions
Pamidronate, an intravenous bisphosphonate, exhibits several drug interactions that can impact its renal safety, calcium homeostasis, and overall efficacy. Concomitant use with nephrotoxic agents heightens the risk of renal impairment, as pamidronate itself is associated with nephrotoxicity through mechanisms involving tubular damage and reduced glomerular filtration.[20][14] Aminoglycosides, such as gentamicin, can potentiate pamidronate's hypocalcemic effects and contribute to additive nephrotoxicity by further impairing renal function and prolonging serum calcium suppression.[38][39] Nonsteroidal anti-inflammatory drugs (NSAIDs), including ibuprofen, may exacerbate renal toxicity through prostaglandin inhibition, leading to reduced renal blood flow and potential acute kidney injury when combined with pamidronate.[40] Loop diuretics, like furosemide, do not alter pamidronate's calcium-lowering action but can increase the risk of hypocalcemia due to enhanced urinary calcium excretion.[20][40] In patients with multiple myeloma, thalidomide co-administration elevates the risk of renal dysfunction, necessitating close monitoring of serum creatinine.[20] Pamidronate must not be co-administered intravenously with calcium-containing solutions, such as Ringer's lactate, due to the formation of insoluble calcium-pamidronate precipitates that can obstruct infusion lines or cause embolism.[41][42] Concurrent use of pamidronate with other bisphosphonates, such as zoledronic acid, should be avoided to prevent cumulative toxicity, including heightened risks of hypocalcemia, osteonecrosis of the jaw, and renal deterioration from excessive osteoclast inhibition.[13][42] When pamidronate is used with interacting drugs, renal function should be monitored via serum creatinine levels before each dose, with adjustments to dosing intervals or withholding treatment if creatinine rises significantly (e.g., by 0.5 mg/dL from baseline). Electrolytes, including calcium, should also be assessed to mitigate hypocalcemia risks.[41][14]History
Development
Pamidronic acid, also known as 3-amino-1-hydroxypropylidene-1,1-bisphosphonate (APD), was developed during the late 1960s and early 1970s as part of broader research into bisphosphonates for inhibiting bone resorption. The compound was synthesized by Henkel KGaA in Germany and patented around 1971, then provided to researchers for initial pharmacological evaluation at Leiden University Medical Center in the Netherlands, in collaboration with Henkel, building on earlier work with non-nitrogen-containing bisphosphonates like etidronate and clodronate. The compound was first described in the scientific literature in 1977, marking its emergence as a second-generation bisphosphonate with potential for treating bone disorders characterized by excessive resorption.[1][43] Ciba-Geigy Corporation (now part of Novartis) acquired rights to the compound and advanced its development, emphasizing intravenous formulations for clinical viability. Preclinical studies conducted in the 1970s at Leiden demonstrated its superior potency in animal models, particularly in rats induced with hypercalcemia and conditions mimicking Paget's disease. These experiments showed that pamidronic acid inhibited bone resorption up to 10 times more effectively than clodronate at doses below 40 μmol/kg/day, while exerting minimal effects on bone mineralization or ectopic calcification.[1][43] Initial clinical trials began in the late 1970s, with the first reported use in 1979 for treating Paget's disease of bone, where oral and intravenous administrations normalized biochemical markers of bone turnover in patients. By the early 1980s, multicenter trials focused on hypercalcemia of malignancy, revealing pamidronic acid's greater efficacy compared to etidronate in rapidly restoring normocalcemia and reducing osteolytic lesions associated with bone metastases. A pivotal milestone in its development was the recognition of the nitrogen-containing side chain, which conferred enhanced antiresorptive activity by disrupting mevalonate pathways in osteoclasts, distinguishing it from first-generation bisphosphonates and guiding further optimization efforts.[43]Regulatory approvals
Pamidronic acid, marketed as the disodium salt under the brand name Aredia, received its first marketing authorizations in several European countries in 1987.[1] It was authorized for the treatment of hypercalcemia associated with malignancy and other bone-related conditions, with the European Medicines Agency (EMA) granting centralized authorization in 2004 and ongoing approvals for generic formulations available since the early 2000s.[1] In the United States, the Food and Drug Administration (FDA) approved pamidronate disodium injection (Aredia) on October 31, 1991, initially for the treatment of moderate to severe hypercalcemia of malignancy.[4] Subsequent expansions included FDA approval in 1994 for moderate to severe Paget's disease of bone, in 1995 for osteolytic bone lesions of multiple myeloma, and in 1996 for osteolytic bone metastases of breast cancer.[44][45] Generic versions of pamidronate disodium became available in the US following the expiration of key patents, such as US Patent No. 4,711,880 in 2005, with initial FDA approvals for generics granted as early as 2001 and broader market entry by 2002.[46] In various other countries, generic approvals followed patent expirations in the mid-2000s, enhancing accessibility.[47] No major withdrawals or restrictions have occurred for pamidronic acid, though regulatory agencies issued label updates in the early 2000s to include warnings and monitoring recommendations for osteonecrosis of the jaw (ONJ), based on postmarketing reports.[18] Pamidronate is included on the World Health Organization's Model List of Essential Medicines for palliative care in cancer patients, particularly for managing hypercalcemia and preventing skeletal-related events in bone metastases.[48]Society and culture
Brand names
Pamidronic acid, more commonly known in its therapeutic form as pamidronate disodium, is marketed under the primary brand name Aredia by Novartis, which serves as the original trade name for the disodium pamidronate injection used in clinical settings.[1][49] Other notable brand names include Pamimed (Curacell Biotech), and Pamisol, alongside regional variants such as pms-Pamidronate produced by Pharmascience.[1][50] Since the expiration of Novartis's patents in the early 2000s, generic forms of pamidronate disodium have become widely available from multiple manufacturers, including Teva Parenteral Medicines, Hospira (now part of Pfizer), Sagent Pharmaceuticals, and regional producers like Sun Pharma Global.[1][51] These products are primarily formulated as intravenous injections at a concentration of 3 mg/mL, supplied in vials containing 15 mg, 30 mg, 60 mg, or 90 mg for reconstitution and administration.[1][49] In early scientific literature, the drug was often referred to by the abbreviation APD (aminohydroxypropylidene diphosphonate), while branding consistently emphasizes the disodium salt form to highlight its pharmaceutical stability and solubility.[52]Availability
Pamidronic acid, marketed as pamidronate disodium, requires a medical prescription for use worldwide and is strictly administered intravenously in clinical settings, such as hospitals or outpatient facilities, rather than being available over-the-counter. In India, it is classified under Schedule H of the Drugs and Cosmetics Rules, mandating a physician's prescription and pharmacist oversight for dispensing. This prescription-only status ensures its use is supervised due to the need for dose monitoring and potential side effects associated with bisphosphonate therapy. The drug is widely accessible in developed countries through hospital pharmacies and specialized oncology or endocrinology clinics, where it is supplied in vials of 30 mg or 90 mg for IV infusion. Generic formulations from manufacturers including Pfizer, Viatris, and others have significantly lowered costs compared to the original branded version, Aredia, making it more affordable for routine use in treating conditions like hypercalcemia of malignancy and osteolytic bone lesions. In the United States, the wholesale acquisition cost for a standard 90 mg dose typically ranges from $50 to $100, depending on the supplier and volume purchased, though retail prices can vary with pharmacy markups. Globally, access remains uneven, with robust availability in high-income regions contrasted by challenges in low- and middle-income countries, where limited healthcare infrastructure hinders IV administration and storage requirements. Pamidronic acid's inclusion in certain national essential medicines lists supports its role in palliative care for cancer-related bone complications, but procurement barriers and dependency on imported supplies exacerbate disparities. Cost factors further influence accessibility; generics are substantially cheaper than branded products—often 50-80% less—while insurance coverage is common in systems like the U.S. Medicare program for FDA-approved indications such as Paget's disease and bone metastases, reducing out-of-pocket expenses for eligible patients. As of September 2024, the U.S. shortage was resolved, with products available from Pfizer and Viatris; global access continues to vary, with inclusion in national essential medicines lists in countries like Eswatini and South Africa supporting palliative care.[53] Supply chain issues have occasionally disrupted availability, particularly for injectable bisphosphonates like pamidronic acid. In the 2010s, manufacturing disruptions and quality control problems led to national shortages in the U.S., affecting oncology treatments and prompting reliance on alternative agents. Shortages occurred in the 2010s and early 2020s due to manufacturing disruptions; by 2024, following Areva Pharmaceuticals' discontinuation of marketing, supplies stabilized with availability from Pfizer and Viatris, resolving the shortage.[53] These interruptions highlight vulnerabilities in generic sterile injectables, often tied to concentrated production and regulatory compliance demands.References
- https://www.[drugs.com](/page/Drugs.com)/pro/pamidronate.html
- https://www.[drugs.com](/page/Drugs.com)/pregnancy/pamidronate.html

