Pap test
View on WikipediaThis article needs additional citations for verification. (July 2023) |
| Pap test | |
|---|---|
 High-grade squamous intraepithelial lesion | |
| Specialty | Gynaecology, cytopathology |
| ICD-9-CM | 795.00 |
| MeSH | D014626 |
| MedlinePlus | 003911 |
The Papanicolaou test (abbreviated as Pap test, also known as Pap smear (AE),[1] cervical smear (BE), cervical screening (BE),[2] or smear test (BE)) is a method of cervical screening used to detect potentially precancerous and cancerous processes in the cervix (opening of the uterus or womb) or, more rarely, anus (in both men and women).[3] Abnormal findings are often followed up by more sensitive diagnostic procedures and, if warranted, interventions that aim to prevent progression to cervical cancer. The test was independently invented in the 1920s by the Greek physician Georgios Papanikolaou and named after him. A simplified version of the test was introduced by the Canadian obstetrician Anna Marion Hilliard in 1957.
A Pap smear is performed by opening the vagina with a speculum and collecting cells at the outer opening of the cervix at the transformation zone (where the outer squamous cervical cells meet the inner glandular endocervical cells), using an Ayre spatula or a cytobrush. The collected cells are examined under a microscope to look for abnormalities. The test aims to detect potentially precancerous changes (called cervical intraepithelial neoplasia (CIN) or cervical dysplasia; the squamous intraepithelial lesion system (SIL) is also used to describe abnormalities) caused by human papillomavirus, a sexually transmitted DNA virus. The test remains an effective, widely used method for early detection of precancer and cervical cancer. While the test may also detect infections and abnormalities in the endocervix and endometrium, it is not designed to do so.
Guidelines on when to begin Pap smear screening are varied, but usually begin in adulthood. Guidelines on frequency vary from every three to five years.[4][5][6] If results are abnormal, and depending on the nature of the abnormality, the test may need to be repeated in six to twelve months.[7] If the abnormality requires closer scrutiny, the patient may be referred for detailed inspection of the cervix by colposcopy, which magnifies the view of the cervix, vagina and vulva surfaces. The person may also be referred for HPV DNA testing, which can serve as an adjunct to Pap testing. In some countries, viral DNA is checked for first, before checking for abnormal cells.[8] Additional biomarkers that may be applied as ancillary tests with the Pap test are evolving.[9]
Medical uses
[edit]| Summary of reasons for testing | ||
|---|---|---|
| patient's characteristic | indication | rationale |
| under age 21, regardless of sexual history | no test | more harms than benefits[10] |
| age 20–25 until age 50–60 | test every 3–5 years if results normal | broad recommendation[11] |
| over age 65; history of normal tests | no further testing | recommendation of USPSTF, ACOG, ACS and ASCP;[5][12][13] |
| had total hysterectomy for non-cancer disease – cervix removed | no further testing | harms of screening after hysterectomy outweigh the benefits[10] |
| had partial hysterectomy – cervix remains | continue testing as normal | |
| has received HPV vaccine | continue testing as normal | vaccine does not cover all cancer-causing types of HPV[11] |
| history of endometrial cancer, with history of hysterectomy | discontinue routine testing[14] | test no longer effective and likely to give false positive[14] |
Screening guidelines vary from country to country. In general, screening starts about the age of 20 or 25 and continues until about the age of 50 or 60.[12] Screening is typically recommended every three to five years, as long as results are normal.[11]
American Congress of Obstetricians and Gynecologists (ACOG) and others recommend starting screening at age 21.[5][15] Many other countries wait until age 25 or later to start screening. For instance, some parts of Great Britain start screening at age 25. ACOG's general recommendation is that people with female reproductive organs age 30–65 have an annual well-woman examination, that they not get annual Pap tests, and that they do get Pap tests at three to five year intervals.[16]
HPV is passed through skin to skin contact; sex does not have to occur, although it is a common way for it to spread.[17] It takes an average of a year, but can take up to four years, for a person's immune system to clear the initial infection. Screening during this period may show this immune reaction and repair as mild abnormalities, which are usually not associated with cervical cancer, but could cause the patient stress and result in further tests and possible treatment. Cervical cancer usually takes time to develop, so delaying the start of screening a few years poses little risk of missing a potentially precancerous lesion. For instance, screening people under age 25 does not decrease cancer rates under age 30.[18]
HPV can be transmitted in sex between females, so those who have only had sex with other females should be screened, although they are at somewhat lower risk for cervical cancer.[19]
Guidelines on frequency of screening vary—typically every three to five years for those who have not had previous abnormal smears.[11] Some older recommendations suggested screening as frequently as every one to two years, however there is little evidence to support such frequent screening; annual screening has little benefit but leads to greatly increased cost and many unnecessary procedures and treatments.[5] It has been acknowledged since before 1980 that most people can be screened less often.[20] In some guidelines, frequency depends on age; for instance in Great Britain, screening is recommended every three years for women under 50, and every five years for those over.[21]
Screening should stop at about age 65 unless there is a history of abnormal test result or disease. There is probably no benefit in screening people aged 60 or over whose previous tests have been negative.[13] If a woman's last three Pap results were normal, she can discontinue testing at age 65, according to the USPSTF, ACOG, ACS, and ASCP;[5] England's NHS says 64. There is no need to continue screening after a complete hysterectomy for benign disease.
Pap smear screening is still recommended for those who have been vaccinated against HPV[11] since the vaccines do not cover all HPV types that can cause cervical cancer. Also, the vaccine does not protect against HPV exposure before vaccination.[22]
Those with a history of endometrial cancer should discontinue routine Pap tests after hysterectomy.[14][23] Further tests are unlikely to detect recurrence of cancer but do bring the risk of giving false positive results, which would lead to unnecessary further testing.[14]
More frequent Pap smears may be needed to follow up after an abnormal Pap smear, after treatment for abnormal Pap or biopsy results, or after treatment of cancer (cervical, anal, etc.).[24]
Effectiveness
[edit]The Pap test, when combined with a regular program of screening and appropriate follow-up, can reduce cervical cancer deaths by up to 80%.[11]
Failure of prevention of cancer by the Pap test can occur for many reasons, including not getting regular screening, lack of appropriate follow-up of abnormal results, and sampling and interpretation errors.[25] In the US, over half of all invasive cancers occur in females who have never had a Pap smear; an additional 10 to 20% of cancers occur in those who have not had a Pap smear in the preceding five years.[citation needed] About one-quarter of US cervical cancers were in people who had an abnormal Pap smear but did not get appropriate follow-up (patient did not return for care, or clinician did not perform recommended tests or treatment).[citation needed]
Adenocarcinoma of the cervix has not been shown to be prevented by Pap smears.[25] In the UK, which has a Pap smear screening program, adenocarcinoma accounts for about 15% of all cervical cancers.[26]
Estimates of the effectiveness of the United Kingdom's call and recall system vary widely, but it may prevent about 700 deaths per year in the UK.[27]
Multiple studies have performed sensitivity and specificity analyses on Pap smears. Sensitivity analysis captures the ability of Pap smears to correctly identify women with cervical cancer. Various studies have revealed the sensitivity of Pap smears to be between 47.19 - 55.5%.[28][29][30] Specificity analysis captures the ability of Pap smears to correctly identify women without cervical cancer. Various studies have revealed the specificity of Pap smears to be between 64.79 - 96.8%.[28][29][30] While Pap smears may not be entirely accurate, they remain one of the most effective cervical cancer prevention tools. Pap smears may be supplemented with HPV DNA testing.[citation needed]
Results
[edit]In screening a general or low-risk population, most Pap results are normal.
In the United States, about 2–3 million abnormal Pap smear results are found each year.[31] Most abnormal results are mildly abnormal (ASC-US (typically 2–5% of Pap results) or low-grade squamous intraepithelial lesion (LSIL) (about 2% of results)), indicating HPV infection.[citation needed] Although most low-grade cervical dysplasias spontaneously regress without ever leading to cervical cancer, dysplasia can serve as an indication that increased vigilance is needed.
In a typical scenario, about 0.5% of Pap results are high-grade SIL (HSIL), and less than 0.5% of results indicate cancer; 0.2 to 0.8% of results indicate Atypical Glandular Cells of Undetermined Significance (AGC-NOS).[citation needed]
As liquid-based preparations (LBPs) become a common medium for testing, atypical result rates have increased. The median rate for all preparations with low-grade squamous intraepithelial lesions using LBPs was 2.9% in 2006, compared with a 2003 median rate of 2.1%. Rates for high-grade squamous intraepithelial lesions (median, 0.5%) and atypical squamous cells have changed little.[32]
Abnormal results are reported according to the Bethesda system.[33] They include:[31]
- Atypical squamous cells (ASC)
- Atypical squamous cells of undetermined significance (ASC-US)
- Atypical squamous cells – cannot exclude HSIL (ASC-H)
- Squamous intraepithelial lesion (SIL)
- Low-grade squamous intraepithelial lesion (LGSIL or LSIL)
- High-grade squamous intraepithelial lesion (HGSIL or HSIL)
- Squamous cell carcinoma
- Glandular epithelial cell abnormalities
- Atypical glandular cells not otherwise specified (AGC or AGC-NOS)
Endocervical and endometrial abnormalities can also be detected, as can a number of infectious processes, including yeast, herpes simplex virus and trichomoniasis. However it is not very sensitive at detecting these infections, so absence of detection on a Pap does not mean absence of the infection.[34]
-
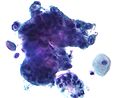
Micrograph of a normal pap smear
-
Micrograph of a Pap test showing a low-grade intraepithelial lesion (LSIL) and benign endocervical mucosa. Pap stain.
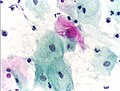
-
Micrograph of a Pap test showing trichomoniasis. Trichomonas organism seen in the upper right. Pap stain.
-
Endocervical adenocarcinoma on a pap test.
-
Candida organisms on a pap test.
-
Viral cytopathic effect consistent with herpes simplex virus on a pap test.
-
Normal squamous epithelial cells in premenopausal women
-
Atrophic squamous cells in postmenopausal women
-
Normal endocervical cells should be present into the slide, as a proof of a good quality sampling
-
The cytoplasms of squamous epithelial cells melted out; many Döderlein bacilli can be seen.
-
Infestation by Trichomonas vaginalis
-
An obviously atypical cell can be seen
Pregnancy
[edit]Pap tests can usually be performed during pregnancy up to at least 24 weeks of gestational age.[35] Pap tests during pregnancy have not been associated with increased risk of miscarriage.[35] An inflammatory component is commonly seen on Pap smears from pregnant women[36] and does not appear to be a risk for subsequent preterm birth.[37]
After childbirth, it is recommended to wait 12 weeks before taking a Pap test because inflammation of the cervix caused by the birth interferes with test interpretation.[38]
In transgender individuals
[edit]Transgender men are also typically at risk for HPV due to retention of the uterine cervix in the majority of individuals in this subgroup.[39][40] As such, professional guidelines recommend that transgender men be screened routinely for cervical cancer using methods such as Pap smear, identical to the recommendations for cisgender women.[41]
However, transgender men have lower rates of cervical cancer screening than cisgender women.[42] Many transgender men report barriers to receiving gender-affirming healthcare,[43][44] including lack of insurance coverage and stigma/discrimination[45][46] during clinical encounters, and may encounter provider misconceptions regarding risk in this population for cervical cancer. Pap smears may be presented to patients as non-gendered screening procedures for cancer rather than one specific for examination of the female reproductive organs. Pap smears may trigger gender dysphoria in patients and gender-neutral language can be used when explaining the pathogenesis of cancer due to infection, emphasizing the pervasiveness of HPV infection regardless of gender.[47]
Transgender women who have not had vaginoplasties are not at risk of developing cervical cancer because they do not have cervices. Transgender women who have had vaginoplasties and have a neo-cervix or neo-vagina have a small chance of developing cancer, according to the Canadian Cancer Society.[48] Surgeons typically use penile skin to create the new vagina and cervix, which can contract HPV and lead to penile cancer, although it is considerably rarer than cervical cancer.[49] Because the risk of this kind of cancer is so low, cervical cancer screening is not routinely offered for those with a neo-cervix.[50][40][51][49]
Procedure
[edit]According to the CDC, intercourse, douching, and the use of vaginal medicines or spermicidal foam should be avoided for 2 days before the test.[52] A number of studies have shown that using a small amount of water-based gel lubricant does not interfere with, obscure, or distort the Pap smear. Further, cytology is not affected, nor are some STD testing.[53] The CDC states that Pap smears can be performed during menstruation.[52] However, the NHS recommends against cervical screening during, or in the 2 days before and after, menstruation.[54] Pap smears can be performed during menstruation, especially if the physician is using a liquid-based test; however if bleeding is extremely heavy, endometrial cells can obscure cervical cells, and if this occurs the test may need to be repeated in 6 months.[55]
Pap smears begin with the insertion of a speculum into the vagina, which spreads the vagina open and allows access to the cervix. The health care provider then collects a sample of cells from the outer opening or external os of the cervix by scraping it with either a spatula or brush.[56]
Obtaining a Pap smear should not cause much pain,[57] but may be uncomfortable.[58] Conditions such as vaginismus, vulvodynia, or cervical stenosis can cause insertion of the speculum to be painful.[59][60]
In a conventional Pap smear, the cells are placed on a glass slide and taken to the laboratory to be checked for abnormalities.[61]
A plastic-fronded broom is sometimes used in place of the spatula or brush. The broom is not as good a collection device, since it is much less effective at collecting endocervical material than the spatula and brush.[62] The broom is used more frequently with the advent of liquid-based cytology, although either type of collection device may be used with either type of cytology.
The sample is stained using the Papanicolaou technique, in which tinctorial dyes and acids are selectively retained by cells. Unstained cells cannot be seen adequately with a light microscope. Papanicolaou chose stains that highlighted cytoplasmic keratinization, which actually has almost nothing to do with the nuclear features used to make diagnoses now.[citation needed]
A single smear has an area of 25 x 50 mm and contains a few hundred thousand cells on average. Screening with light microscopy is first done on low (10x) power and then switched to higher (40x) power upon viewing suspicious findings. Cells are analyzed under high power for morphologic changes indicative of malignancy (including enlarged and irregularly shaped nucleus, an increase in nucleus to cytoplasm ratio, and more coarse and irregular chromatin). Approximately 1,000 fields of view are required on 10x power for screening of a single sample, which takes on average 5 to 10 minutes.[63]
In some cases, a computer system may prescreen the slides, indicating those that do not need examination by a person or highlighting areas for special attention. The sample is then usually screened by a specially trained and qualified cytotechnologist using a light microscope. The terminology for who screens the sample varies according to the country; in the UK, the personnel are known as cytoscreeners, biomedical scientists (BMS), advanced practitioners and pathologists. The latter two take responsibility for reporting the abnormal sample, which may require further investigation.[citation needed]
Automated analysis
[edit]Since the early 2000s, there have been successful attempts to develop automated, computer image analysis systems for screening.[64] Although, on the available evidence, automated cervical screening could not be recommended for implementation into a national screening program, an NHS Health technology appraisal in 2005 concluded that the "general case for automated image analysis ha(d) probably been made".[65] Automation may improve sensitivity and reduce unsatisfactory specimens.[66] Two systems have been approved by the FDA and function in high-volume reference laboratories, with human oversight.[citation needed]
Types of screening
[edit]- Conventional Pap—In a conventional Pap smear, samples are smeared directly onto a microscope slide after collection.
- Liquid-based cytology—The sample of (epithelial) cells is taken from the transitional zone, the squamocolumnar junction of the cervix, between the ectocervix and the endocervix. The cells taken are suspended in a bottle of preservative for transport to the laboratory, where they are analyzed using Pap stains.

Type 1: Completely ectocervical.
Type 2: Endocervical component but fully visible.
Type 3: Endocervical component, not fully visible.


Pap tests commonly examine epithelial abnormalities, such as metaplasia, dysplasia, or borderline changes, all of which may be indicative of CIN. Nuclei will stain dark blue, squamous cells will stain green and keratinised cells will stain pink/ orange. Koilocytes may be observed where there is some dyskaryosis (of epithelium). The nucleus in koilocytes is typically irregular, indicating possible cause for concern; requiring further confirmatory screens and tests.
In addition, human papillomavirus (HPV) test may be performed either as indicated for abnormal Pap results, or in some cases, dual testing is done, where both a Pap smear and an HPV test are done at the same time (also called Pap co-testing).[68]
Practical aspects
[edit]The endocervix may be partially sampled with the device used to obtain the ectocervical sample, but due to the anatomy of this area, consistent and reliable sampling cannot be guaranteed. Since abnormal endocervical cells may be sampled, those examining them are taught to recognize them.[citation needed]
The endometrium is not directly sampled with the device used to sample the ectocervix. Cells may exfoliate onto the cervix and be collected from there, so as with endocervical cells, abnormal cells can be recognised if present but the Pap test should not be used as a screening tool for endometrial malignancy.[citation needed]
In the United States, a Pap test itself costs $20 to $30, but the costs for Pap test visits can cost over $1,000, largely because additional tests are added that may or may not be necessary.[69]
History
[edit]The test was invented by and named after the Greek doctor Georgios Papanikolaou, who started his research in 1923.[70] Aurel Babeș independently made similar discoveries in 1927.[71][72] However, Babeș' method was radically different from Papanikolaou's.[70][73]
The Pap test was finally recognized only after a leading article in the American Journal of Obstetrics and Gynecology in 1941 by Papanikolaou and Herbert F. Traut, an American gynecologist.[70][74] A monograph titled Diagnosis of Uterine Cancer by the Vaginal Smear that they published contained drawings of the various cells seen in patients with no disease, inflammatory conditions, and preclinical and clinical carcinoma.[70] The monograph was illustrated by Hashime Murayama, who later became a staff illustrator with the National Geographic Society.[70] Both Papanikolaou and his wife, Andromachi Papanikolaou, dedicated the rest of their lives to teaching the technique to other physicians and laboratory personnel.[70]
Experimental techniques
[edit]In the developed world, cervical biopsy guided by colposcopy is considered the "gold standard" for diagnosing cervical abnormalities after an abnormal Pap smear. Other techniques such as triple smear are also done after an abnormal Pap smear.[75] The procedure requires a trained colposcopist and can be expensive to perform. However, Pap smears are very sensitive and some negative biopsy results may represent undersampling of the lesion in the biopsy, so negative biopsy with positive cytology requires careful follow-up.[76]
Experimental visualization techniques use broad-band light (e.g., direct visualization, speculoscopy, cervicography, visual inspection with acetic acid or with Lugol's, and colposcopy) and electronic detection methods (e.g., Polarprobe and in vivo spectroscopy). These techniques are less expensive and can be performed with significantly less training. They do not perform as well as Pap smear screening and colposcopy. At this point, these techniques have not been validated by large-scale trials and are not in general use.[citation needed]
Implementation by country
[edit]Australia
[edit]Australia has used the Pap test as part of its cervical screening program since its implementation in 1991 which required women past the age of 18 be tested every two years.[77] In December 2017 Australia discontinued its use of the Pap test and replaced it with a new HPV test that is only required to be conducted once every five years from the age of 25.[78] Medicare covers the costs of testing; however, if a patient's doctor does not allow bulk billing, they may have to pay for the appointment and then claim the Medicare rebate.[79]
Taiwan
[edit]Free Pap tests were offered from 1974–1984 before being replaced by a system in which all women over the age of 30 could have the cost of their Pap test reimbursed by the National Health Insurance in 1995.[80] This policy was still ongoing in 2018 and encouraged women to screen at least every three years.[81]
Despite this, the number of people receiving Pap tests remain lower than countries like Australia. Some believe this is due to a lack of awareness regarding the test and its availability. It has also been found that women who have chronic diseases or other reproductive diseases are less likely to receive the test.[82][83][84]
England
[edit]As of 2020[update] the NHS maintains a cervical screening program in which women between the age of 25–49 are invited for a smear test every three years, and women past 50 every five years. Much like Australia, England uses a HPV test before examining cells that test positive using the Pap test.[85] The test is free as part of the national cervical screening program.[86]
Coccoid bacteria
[edit]The finding of coccoid bacteria on a Pap test is of no consequence with otherwise normal test findings and no infectious symptoms. However, if there is enough inflammation to obscure the detection of precancerous and cancerous processes, it may indicate treatment with a broad-spectrum antibiotic for streptococci and anaerobic bacteria (such as metronidazole and amoxicillin) before repeating the smear. Alternatively, the test will be repeated at an earlier time than it would otherwise.[87] If there are symptoms of vaginal discharge, bad odor or irritation, the presence of coccoid bacteria also may indicate treatment with antibiotics as per above.[87]
References
[edit]- Notes
- ^ "Pap Smear: MedlinePlus Lab Test Information". medlineplus.gov. Retrieved 2018-11-07.
- ^ "Cervical Screening". NHS. 2017-10-20. Retrieved 2018-09-04.
- ^ Lindsey, Kimberley; DeCristofaro, Claire; James, Janet (August 2009). "Anal Pap smears: Should we be doing them?". Journal of the American Academy of Nurse Practitioners. 21 (8): 437–443. doi:10.1111/j.1745-7599.2009.00433.x. PMID 19689440.
- ^ Moyer, VA; U.S. Preventive Services Task, Force (Jun 19, 2012). "Screening for cervical cancer: U.S. Preventive Services Task Force recommendation statement". Annals of Internal Medicine. 156 (12): 880–91, W312. doi:10.7326/0003-4819-156-12-201206190-00424. PMID 22711081. S2CID 36965456.
- ^ a b c d e Saslow, Debbie; Solomon, Diane; Lawson, Herschel W.; Killackey, Maureen; Kulasingam, Shalini L.; Cain, Joanna M.; Garcia, Francisco A. R.; Moriarty, Ann T.; Waxman, Alan G.; Wilbur, David C.; Wentzensen, Nicolas; Downs, Levi S.; Spitzer, Mark; Moscicki, Anna-Barbara; Franco, Eduardo L.; Stoler, Mark H.; Schiffman, Mark; Castle, Philip E.; Myers, Evan R. (July 2012). "American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology Screening Guidelines for the Prevention and Early Detection of Cervical Cancer". Journal of Lower Genital Tract Disease. 16 (3): 175–204. doi:10.1097/LGT.0b013e31824ca9d5. PMC 3915715. PMID 22418039.
- ^ American Cancer Society. (2010). Detailed Guide: Cervical Cancer. Can cervical cancer be prevented? Archived 2016-12-10 at the Wayback Machine Retrieved August 8, 2011.
- ^ The American College of Obstetricians and Gynecologists (2009). "ACOG Education Pamphlet AP085 – The Pap Test". Washington, DC. Archived from the original on June 15, 2010. Retrieved June 5, 2010.
- ^ "Cervical cancer screening in England to use 'more accurate' viral DNA test". Cancer Research UK - Cancer News. 2016-07-05. Retrieved 2023-08-10.
- ^ Shidham, VinodB; Mehrotra, Ravi; Varsegi, George; D′Amore, Krista L; Hunt, Bryan; Narayan, Raj (2011-01-01). "p16 INK4a immunocytochemistry on cell blocks as an adjunct to cervical cytology: Potential reflex testing on specially prepared cell blocks from residual liquid-based cytology specimens". CytoJournal. 8 (1): 1. doi:10.4103/1742-6413.76379. PMC 45765. PMID 21369522.
- ^ a b American Academy of Family Physicians. "Five Things Physicians and Patients Should Question" (PDF). Choosing Wisely: An Initiative of the ABIM Foundation. Archived from the original (PDF) on November 11, 2017. Retrieved August 14, 2012.
- ^ a b c d e f Arbyn M, Anttila A, Jordan J, Ronco G, Schenck U, Segnan N, Wiener H, Herbert A, von Karsa L (2010). "European Guidelines for Quality Assurance in Cervical Cancer Screening. Second Edition—Summary Document". Annals of Oncology. 21 (3): 448–458. doi:10.1093/annonc/mdp471. PMC 2826099. PMID 20176693.
- ^ a b Strander, B (2009). "At what age should cervical screening stop?". Br Med J. 338: 1022–23. doi:10.1136/bmj.b809. PMID 19395422. S2CID 37206485.
- ^ a b Sasieni P, Adams J, Cuzick J (2003). "Benefit of cervical screening at different ages: evidence from the UK audit of screening histories". Br J Cancer. 89 (1): 88–93. doi:10.1038/sj.bjc.6600974. PMC 2394236. PMID 12838306.
- ^ a b c d Society of Gynecologic Oncology (February 2014). "Five Things Physicians and Patients Should Question". Choosing Wisely: An Initiative of the ABIM Foundation. Retrieved 19 February 2013., which cites
- Salani R, Backes FJ, Fung MF, Holschneider CH, Parker LP, Bristow RE, Goff BA (2011). "Posttreatment surveillance and diagnosis of recurrence in women with gynecologic malignancies: Society of Gynecologic Oncologists recommendations". American Journal of Obstetrics and Gynecology. 204 (6): 466–78. doi:10.1016/j.ajog.2011.03.008. PMID 21752752.
- Salani R, Nagel CI, Drennen E, Bristow RE (2011). "Recurrence patterns and surveillance for patients with early stage endometrial cancer". Gynecologic Oncology. 123 (2): 205–7. doi:10.1016/j.ygyno.2011.07.014. PMID 21820709.
- Bristow RE, Purinton SC, Santillan A, Diaz-Montes TP, Gardner GJ, Giuntoli RL (2006). "Cost-effectiveness of routine vaginal cytology for endometrial cancer surveillance". Gynecologic Oncology. 103 (2): 709–13. doi:10.1016/j.ygyno.2006.05.013. PMID 16797686.
- ^ ACOG Committee on Gynecological Practice (2009). "ACOG Committee on Gynecologic Practice; Routine Pelvic Examination and Cervical Cytology Screening, Opinion #413". Obstetrics and Gynecology. 113 (5): 1190–1193. doi:10.1097/AOG.0b013e3181a6d022. PMID 19384150.
- ^ American Congress of Obstetricians and Gynecologists. "Five Things Physicians and Patients Should Question". Choosing Wisely: An Initiative of the ABIM Foundation. Retrieved August 1, 2013., which cites
- Boulware LE, Marinopoulos S, Phillips KA, Hwang CW, Maynor K, Merenstein D, Wilson RF, Barnes GJ, Bass EB, Powe NR, Daumit GL (2007). "Systematic review: The value of the periodic health evaluation". Annals of Internal Medicine. 146 (4): 289–300. doi:10.7326/0003-4819-146-4-200702200-00008. PMID 17310053. S2CID 1683342.
- Saslow D, Solomon D, Lawson HW, Killackey M, Kulasingam SL, Cain J, Garcia FA, Moriarty AT, Waxman AG, Wilbur DC, Wentzensen N, Downs LS, Spitzer M, Moscicki AB, Franco EL, Stoler MH, Schiffman M, Castle PE, Myers ER (2012). "American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer". CA Cancer J Clin. 62 (3): 147–72. doi:10.3322/caac.21139. PMC 3801360. PMID 22422631.
- Committee on Gynecologic Practice (2012). "Committee Opinion No. 534". Obstetrics & Gynecology. 120 (2, Part 1): 421–424. doi:10.1097/AOG.0b013e3182680517. PMID 22825111.
- Committee on Practice Bulletins—Gynecology (2012). "ACOG Practice Bulletin Number 131: Screening for cervical cancer". Obstetrics and Gynecology. 120 (5): 1222–1238. doi:10.1097/AOG.0b013e318277c92a. PMID 23090560.
- ^ "Can Cervical Cancer Be Prevented?". www.cancer.org. Retrieved 2018-11-07.
- ^ Sasieni, P; Castanon, A; Cuzick, J; Snow, J (2009). "Effectiveness of Cervical Screening with Age: Population based Case-Control Study of Prospectively Recorded Data". BMJ. 339: 2968–2974. doi:10.1136/bmj.b2968. PMC 2718082. PMID 19638651.
- ^ Marrazzo JM, Koutsky LA, Kiviat NB, Kuypers JM, Stine K (2001). "Papanicolaou test screening and prevalence of genital human papillomavirus among women who have sex with women". American Journal of Public Health. 91 (6): 947–952. doi:10.2105/AJPH.91.6.947. PMC 1446473. PMID 11392939.
- ^ Smith, RA; et al. (2002). "American Cancer Society Guideline for the Early Detection of Cervical Neoplasia and Cancer". CA: A Cancer Journal for Clinicians. 52 (1): 8–22. doi:10.3322/canjclin.52.6.342. PMID 12469763. S2CID 19919545.
ACS and others have recommended, since before 1980, that conventional cytology can be safely performed up to every three years for most women.
- ^ "Cervical screening: programme overview". Gov.UK. Public Health England. Retrieved 21 June 2023.
- ^ "HPV Vaccination Recommendations". Centers for Disease Prevention and Control. 22 May 2023. Retrieved 21 June 2023.
- ^ Salani, R (2017). "An update on post-treatment surveillance and diagnosis of recurrence in women with gynecologic malignancies: Society of Gynecologic Oncology (SGO) recommendations" (PDF). Gynecologic Oncology. 146 (1): 3–10. doi:10.1016/j.ygyno.2017.03.022. PMID 28372871. Archived (PDF) from the original on 2019-07-02.
- ^ "HPV and Pap Test Results: Next Steps after an Abnormal Test - NCI". www.cancer.gov. 2022-10-13. Retrieved 2024-02-25.
- ^ a b DeMay, M. (2007). Practical principles of cytopathology. Revised edition. Chicago, IL: American Society for Clinical Pathology Press. ISBN 978-0-89189-549-7.
- ^ "Cancer Research UK website". Archived from the original on 2009-01-16. Retrieved 2009-01-03.
- ^ Raffle AE, Alden B, Quinn M, Babb PJ, Brett MT (2003). "Outcomes of screening to prevent cancer: analysis of cumulative incidence of cervical abnormality and modelling of cases and deaths prevented". BMJ. 326 (7395): 901. doi:10.1136/bmj.326.7395.901. PMC 153831. PMID 12714468.
- ^ a b "Specificity, sensitivity and cost". Nature Reviews Cancer. 7 (12): 893. December 2007. doi:10.1038/nrc2287. ISSN 1474-1768. S2CID 43571578.
- ^ a b Najib, Fatemeh sadat; Hashemi, Masooumeh; Shiravani, Zahra; Poordast, Tahereh; Sharifi, Sanam; Askary, Elham (September 2020). "Diagnostic Accuracy of Cervical Pap Smear and Colposcopy in Detecting Premalignant and Malignant Lesions of Cervix". Indian Journal of Surgical Oncology. 11 (3): 453–458. doi:10.1007/s13193-020-01118-2. ISSN 0975-7651. PMC 7501362. PMID 33013127.
- ^ a b Nkwabong, Elie; Laure Bessi Badjan, Ingrid; Sando, Zacharie (January 2019). "Pap smear accuracy for the diagnosis of cervical precancerous lesions". Tropical Doctor. 49 (1): 34–39. doi:10.1177/0049475518798532. ISSN 0049-4755. PMID 30222058. S2CID 52280945.
- ^ a b "Pap Smear". Retrieved 2008-12-27.
- ^ Eversole GM, Moriarty AT, Schwartz MR, Clayton AC, Souers R, Fatheree LA, Chmara BA, Tench WD, Henry MR, Wilbur DC (2010). "Practices of participants in the college of american pathologists interlaboratory comparison program in cervicovaginal cytology, 2006". Archives of Pathology & Laboratory Medicine. 134 (3): 331–5. doi:10.5858/134.3.331. PMID 20196659.
- ^ Nayar, Ritu; Solomon, Diane (2004-01-01). "Second edition of 'The Bethesda System for reporting cervical cytology' - Atlas, website, and Bethesda interobserver reproducibility project". CytoJournal. 1 (1): 4. doi:10.1186/1742-6413-1-4. PMC 526759. PMID 15504231.
- ^ González Pedraza Avilés, A.; Ortiz Zaragoza, C.; Topete Barrera, L.; Mota Vázquez, R.; Ponce Rosas, R. (2001-03-15). "[Is the Papanicolaou smear useful as aid for diagnosing some sexually transmitted infections?]". Atencion Primaria. 27 (4): 222–226. doi:10.1016/S0212-6567(01)78800-2. ISSN 0212-6567. PMC 7684047. PMID 11262330.
- ^ a b PapScreen Victoria > Pregnant women Archived 2014-02-01 at the Wayback Machine from Cancer Council Victoria 2014
- ^ [1] Michael CW (1999). "The Papanicolaou Smear and the Obstetric Patient: A Simple Test with Great Benefits". Diagnostic Cytopathology. 21 (1): 1–3. doi:10.1002/(SICI)1097-0339(199907)21:1<1::AID-DC1>3.0.CO;2-0. hdl:2027.42/35304. PMID 10405797. S2CID 1367319.
- ^ Lanouette JM, Puder KS, Berry SM, Bryant DR, Dombrowski MP (1997). "Is inflammation on Papanicolaou smear a risk factor for preterm delivery?". Fetal Diagnosis and Therapy. 12 (4): 244–247. doi:10.1159/000264477. PMID 9354886.
- ^ "Pregnant women". papscreen.org. Cancer Council Victoria. Archived from the original on 2015-01-08. Retrieved 2015-01-16.
- ^ Grant, Jaime M. (2010). National Transgender Discrimination Survey report on health and health care: findings of a study by the National Center for Transgender Equality and the National Gay and Lesbian Task Force. National Center for Transgender Equality. OCLC 707939280.
- ^ a b "Should trans men have cervical screening tests?". NHS. 2018-06-27. Retrieved 2023-08-10.
- ^ "Committee Opinion No. 512: Health Care for Transgender Individuals". Obstetrics & Gynecology. 118 (6): 1454–1458. December 2011. doi:10.1097/AOG.0b013e31823ed1c1. ISSN 0029-7844. PMID 22105293.
- ^ Peitzmeier, Sarah M.; Khullar, Karishma; Reisner, Sari L.; Potter, Jennifer (December 2014). "Pap Test Use Is Lower Among Female-to-Male Patients Than Non-Transgender Women". American Journal of Preventive Medicine. 47 (6): 808–812. doi:10.1016/j.amepre.2014.07.031. PMID 25455121.
- ^ Grant, Jaime M. (2011). Injustice at every turn: a report of the National Transgender Discrimination Survey. National Center for Transgender Equality. OCLC 777348181.
- ^ Snelgrove, John W; Jasudavisius, Amanda M; Rowe, Bradley W; Head, Evan M; Bauer, Greta R (December 2012). ""Completely out-at-sea" with "two-gender medicine": A qualitative analysis of physician-side barriers to providing healthcare for transgender patients". BMC Health Services Research. 12 (1): 110. doi:10.1186/1472-6963-12-110. ISSN 1472-6963. PMC 3464167. PMID 22559234.
- ^ Poteat, Tonia; German, Danielle; Kerrigan, Deanna (May 2013). "Managing uncertainty: A grounded theory of stigma in transgender health care encounters". Social Science & Medicine. 84: 22–29. doi:10.1016/j.socscimed.2013.02.019. PMID 23517700. S2CID 5339596.
- ^ Reisner, Sari L.; Gamarel, Kristi E.; Dunham, Emilia; Hopwood, Ruben; Hwahng, Sel (September 2013). "Female-to-Male Transmasculine Adult Health: A Mixed-Methods Community-Based Needs Assessment". Journal of the American Psychiatric Nurses Association. 19 (5): 293–303. doi:10.1177/1078390313500693. ISSN 1078-3903. PMID 23963876. S2CID 3285779.
- ^ Potter, Jennifer; Peitzmeier, Sarah M.; Bernstein, Ida; Reisner, Sari L.; Alizaga, Natalie M.; Agénor, Madina; Pardee, Dana J. (2015-07-10). "Cervical Cancer Screening for Patients on the Female-to-Male Spectrum: a Narrative Review and Guide for Clinicians". Journal of General Internal Medicine. 30 (12): 1857–1864. doi:10.1007/s11606-015-3462-8. ISSN 0884-8734. PMC 4636588. PMID 26160483.
- ^ "As a trans woman, do I need to get screened for cervical cancer". Canadian Cancer Society. Archived from the original on 2022-09-12. Retrieved 2022-09-12.
- ^ a b "I'm trans or non-binary, does this affect my cancer screening?". Cancer Research UK. 2019-10-10. Retrieved 2023-08-10.
- ^ Anonymous (2020-09-04). "Cervical screening for trans men and/or non-binary people". Jo's Cervical Cancer Trust. Retrieved 2023-08-10.
- ^ "Tweet misleads on cervical cancer guidance for trans women". AP News. 2023-03-17. Retrieved 2023-08-10.
- ^ a b "Screening for Cervical Cancer". CDC. 26 October 2023. Retrieved 15 August 2024.
- ^ Wright, Jessica L. (2010). "The Effect of Using Water-based Gel Lubricant During a Speculum Exam On Pap Smear Results". School of Physician Assistant Studies. Archived from the original on 24 May 2013. Retrieved 4 February 2012.
- ^ "How to book cervical screening". NHS. 2023-07-14. Retrieved 2023-08-13.
- ^ Chan, Paul D. (2006). Gynecology and obstetrics: new treatment guidelines. Internet Archive. Laguna Hills, CA : Current Clinical Strategies Pub. ISBN 978-1-929622-63-4.
- ^ "Cervical Cancer Screening - NCI". www.cancer.gov. 2022-10-13. Retrieved 2023-08-09.
- ^ "Excerpts from Changing Bodies, Changing Lives". Our Bodies Ourselves. Archived from the original on 2013-12-18. Retrieved 2013-07-02.
- ^ "Cervical screening: support for people who feel anxious about attending". GOV.UK. Retrieved 2023-08-09.
- ^ "All About Speculums: Finding What Works for You". www.ohsu.edu. Retrieved 15 August 2024.
- ^ Biggs, Kirsty V.; Soo Hoo, San; Kodampur, Mallikarjun (April 2022). "Mechanical dilatation of the stenosed cervix under local anesthesia: A prospective case series". Journal of Obstetrics and Gynaecology Research. 48 (4): 956–965. doi:10.1111/jog.15179. ISSN 1341-8076. PMC 9303640. PMID 35132727.
- ^ "Conventional Pap Test". Wisconsin State Laboratory of Hygiene. Retrieved 2023-08-09.
- ^ Martin-Hirsch P, Lilford R, Jarvis G, Kitchener HC (1999). "Efficacy of cervical-smear collection devices: a systematic review and meta-analysis". Lancet. 354 (9192): 1763–1770. doi:10.1016/S0140-6736(99)02353-3. PMID 10577637. S2CID 22733963.
- ^ Bengtsson, Ewert; Malm, Patrik (2014). "Screening for Cervical Cancer Using Automated Analysis of PAP-Smears". Computational and Mathematical Methods in Medicine. 2014 842037. doi:10.1155/2014/842037. ISSN 1748-670X. PMC 3977449. PMID 24772188.
- ^ Biscotti CV, Dawson AE, Dziura B, Galup L, Darragh T, Rahemtulla A, Wills-Frank L (2005). "Assisted primary screening using the automated ThinPrep Imaging System". Am. J. Clin. Pathol. 123 (2): 281–7. doi:10.1309/AGB1MJ9H5N43MEGX. PMID 15842055.
- ^ Willis BH, Barton P, Pearmain P, Bryan S, Hyde C (2005). "Cervical screening programmes: can automation help? Evidence from systematic reviews, an economic analysis and a simulation modelling exercise applied to the UK" (PDF). Health Technol. Assess. 9 (13): 1–207, iii. doi:10.3310/hta9130. PMID 15774236. Archived from the original (PDF) on 2008-09-10. Retrieved 2008-08-02.
- ^ Davey E, d'Assuncao J, Irwig L, Macaskill P, Chan SF, Richards A, Farnsworth A (2007). "Accuracy of reading liquid based cytology slides using the ThinPrep Imager compared with conventional cytology: prospective study". BMJ. 335 (7609): 31. doi:10.1136/bmj.39219.645475.55. PMC 1910624. PMID 17604301.
- ^ International Federation for Cervical Pathology and Colposcopy (IFCPC) classification. References:
-"Transformation zone (TZ) and cervical excision types". Royal College of Pathologists of Australasia.
- Jordan, J.; Arbyn, M.; Martin-Hirsch, P.; Schenck, U.; Baldauf, J-J.; Da Silva, D.; Anttila, A.; Nieminen, P.; Prendiville, W. (2008). "European guidelines for quality assurance in cervical cancer screening: recommendations for clinical management of abnormal cervical cytology, part 1". Cytopathology. 19 (6): 342–354. doi:10.1111/j.1365-2303.2008.00623.x. ISSN 0956-5507. PMID 19040546. S2CID 16462929. - ^ Zhang, Salina; McNamara, Megan; Batur, Pelin (June 2018). "Cervical Cancer Screening: What's New? Updates for the Busy Clinician". The American Journal of Medicine. 131 (6): 702.e1–702.e5. doi:10.1016/j.amjmed.2018.01.020. PMID 29408216. S2CID 46780821.
- ^ Bettigole C (2013). "The Thousand-Dollar Pap Smear". New England Journal of Medicine. 369 (16): 1486–1487. doi:10.1056/NEJMp1307295. PMID 24131176.
- ^ a b c d e f Zheng, Wenxin; Fadare, Oluwole; Quick, Charles Matthew; Shen, Danhua; Guo, Donghui (2019-07-01). "History of Pap Test". Gynecologic and Obstetric Pathology, Volume 2. Springer. ISBN 978-981-13-3019-3.
- ^ M.J. O'Dowd, E.E. Philipp, The History of Obstetrics & Gynaecology, London, Parthenon Publishing Group, 1994, p. 547.
- ^ Pambuccian, Stefan E. (2023). "Was the Cytologic Method for Cervical Cancer Diagnosis discovered by Serendipity or by Design (hypothesis-based Research)". Sudhoffs Archiv. 107 (1): 55–83. doi:10.25162/sar-2023-0004. ISSN 2366-2352.
- ^ Diamantis A, Magiorkinis E, Androutsos G (Jul 2010). "What's in a name? Evidence that Papanicolaou, not Babeș, deserves credit for the Pap test". Diagnostic Cytopathology. 38 (7): 473–6. doi:10.1002/dc.21226. PMID 19813255. S2CID 37757448.
- ^ Papanicolaou, George N.; Traut, Herbert F. (1941). "The Diagnostic Value of Vaginal Smears in Carcinoma of the Uterus**This study has been aided by the Commonwealth Fund. Presented before the New York Obstetrical Society, March 11, 1941". American Journal of Obstetrics and Gynecology. 42 (2): 193–206. doi:10.1016/s0002-9378(16)40621-6. ISSN 0002-9378.
- ^ Krunger, TF; Botha, MH (2007). Clinical gynaecology (3 ed.). South Africa: Juta. p. 23. ISBN 978-0-7021-7305-9. Retrieved 7 December 2016.
- ^ Bewtra, Chhanda; Pathan, Muhammad; Hashish, Hisham (2003-10-01). "Abnormal Pap smears with negative follow-up biopsies: Improving cytohistologic correlations". Diagnostic Cytopathology. 29 (4): 200–202. doi:10.1002/dc.10329. ISSN 1097-0339. PMID 14506671. S2CID 40202036.
- ^ "Cervical cancer screening". www.cancer.org.au. Retrieved 2020-08-13.
- ^ "Cervical Screening". Australian Government Department of Health. 15 August 2019. Retrieved 2020-08-13.
- ^ Cancer Institute of NSW. "Do I need to pay for my cervical screen". Cancer Institute NSW.
- ^ Chen, Y-Y; You, S-L; Chen, C-A; Shih, L-Y; Koong, S-L; Chao, K-Y; Hsiao, M-L; Hsieh, C-Y; Chen, C-J (2009-07-07). "Effectiveness of national cervical cancer screening programme in Taiwan: 12-year experiences". British Journal of Cancer. 101 (1): 174–177. doi:10.1038/sj.bjc.6605139. ISSN 0007-0920. PMC 2713714. PMID 19536091.
- ^ Chen, M.-J.; Wu, C.-Y.; Chen, R.; Wang, Y.-W. (2018-10-01). "HPV Vaccination and Cervical Cancer Screening in Taiwan". Journal of Global Oncology. 4 (Supplement 2): 235s. doi:10.1200/jgo.18.94300. ISSN 2378-9506.
- ^ Fang-Hsin Leea; Chung-Yi Lic; Hsiu-Hung Wanga; Yung-Mei Yang (2013). "The utilization of Pap tests among different female medical personnel: A nationwide study in Taiwan". Preventive Medicine. 56 (6): 406–409. doi:10.1016/j.ypmed.2013.03.001. PMID 23524115.
- ^ "Knowledge of Cervical Cancer Screening among Women". iprojectmaster.com. Retrieved 2020-02-10.
- ^ Peterson NB, Murff HJ, Cui Y, Hargreaves M, Fowke JH (Jul–Aug 2008). "Papanicolaou testing among women in the southern United States". Journal of Women's Health. 17 (6): 939–946. doi:10.1089/jwh.2007.0576. PMC 2942751. PMID 18582173.
- ^ "Cervical screening (smear testing) | Health Information | Bupa UK". www.bupa.co.uk. Retrieved 2020-08-14.
- ^ Admin (2013-08-30). "About cervical screening". Jo's Cervical Cancer Trust. Retrieved 2020-08-14.
- ^ a b OB-GYN 101: Introductory Obstetrics & Gynecology > Coccoid Bacteria Archived 2014-02-22 at the Wayback Machine by Michael Hughey Hughey at Texas Tech University Health Sciences Center. Retrieved Feb 2014.
External links
[edit]- The Pap Test: Questions and Answers — from the U.S.'s National Cancer Institute
- MedlinePlus: Cervical Cancer Prevention/Screening — from MedlinePlus
- NHS Cervical Screening Programme — from the UK's National Health Service
- Cervical cancer screening information – from Cancer Research UK
- Pap Smear – from Lab Tests Online
- Pap Smear — from eMedicineHealth
- PapScreen – Australian information about Pap tests (or Pap smears)
- Canadian Guidelines for Cervical Cancer Screening – Society of Obstetricians and Gynaecologists of Canada
Pap test
View on GrokipediaOverview
Definition and mechanism
The Pap test, also known as the Papanicolaou test or Pap smear, is an exfoliative cytopathology procedure that collects and examines cells from the cervix to identify precancerous or cancerous changes, infections, inflammatory conditions, and hormonal influences.[17][18] Developed by Georgios Papanicolaou and first detailed in a 1943 monograph, it relies on the principle that abnormal cells shed or can be mechanically dislodged from the cervical epithelium for microscopic analysis.[18] Cell collection targets the transformation zone—the junction between squamous ectocervical epithelium and columnar endocervical epithelium—where most cervical dysplasia arises, often due to persistent human papillomavirus infection. A cytobrush rotates within the endocervical canal to sample glandular cells and mucus, while a plastic spatula scrapes the ectocervix to gather squamous cells; for conventional smears, samples are immediately smeared onto labeled glass slides and fixed in 95% ethanol or aerosol fixative to preserve morphology and prevent autolysis.[18][19] Liquid-based cytology variants suspend cells in preservative fluid, reducing obscuring factors like blood or mucus through automated thin-layer preparation.[19] Stained slides undergo Papanicolaou staining, a polychromatic technique using hematoxylin to basophilically stain nuclei dark blue-purple for chromatin detail assessment, followed by acidophilic counterstains like Orange G (OG-6) for keratinized cytoplasm (orange) and eosin-azure (EA) for non-keratinized elements (pink, blue, green), enabling differentiation of mature squamous cells, endocervical cells, and metaplastic intermediates.[20][21] Under light microscopy at magnifications from 10x to 40x, cytotechnologists screen for cytologic atypia—such as nuclear enlargement, hyperchromasia, irregular nuclear membranes, increased nuclear-to-cytoplasmic ratio, or cytoplasmic vacuolization—while pathologists confirm diagnoses using standardized systems like the Bethesda System, which classifies findings from negative for intraepithelial lesion or malignancy to high-grade squamous intraepithelial lesion.[18][22] This visual detection of dysplastic changes exploits causal cellular responses to oncogenic insults, prioritizing empirical morphology over molecular assays in primary screening.[18]Primary purpose and target population
The Pap test, formally known as the Papanicolaou test, functions primarily as a cytological screening tool to identify atypical squamous or glandular cells in cervical samples, which may indicate precancerous dysplasias such as cervical intraepithelial neoplasia or early invasive carcinoma, thereby facilitating timely diagnostic follow-up and treatment to avert progression to cervical cancer.[23] [24] This screening detects cellular abnormalities caused predominantly by persistent high-risk human papillomavirus (HPV) infection, though it is not diagnostic for HPV itself nor intended for active disease surveillance or symptomatic evaluation.[25] [26] The target population comprises asymptomatic women with an intact cervix who are at average risk for cervical cancer, typically those aged 21 to 65 years, excluding individuals with prior total hysterectomy for non-cancerous conditions or those adequately screened previously without high-grade lesions.[27] [28] Screening initiation occurs at age 21 regardless of sexual history, with cytology alone recommended every 3 years for ages 21–29 to balance detection benefits against overdiagnosis risks in this low-prevalence group.[29] [30] For ages 30–65, while primary high-risk HPV testing every 5 years is increasingly preferred due to superior sensitivity for detecting persistent infections, Pap cytology remains a viable standalone option every 3 years or in co-testing with HPV every 5 years, per guidelines from the U.S. Preventive Services Task Force (USPSTF, reaffirmed December 2024 draft) and American College of Obstetricians and Gynecologists (ACOG).[29] [30] Discontinuation after age 65 is advised for those with three consecutive negative cytology results or two negative co-tests within the prior 10 years, absent high-risk history.[27] The American Cancer Society advocates starting at age 25 with primary HPV testing to 65, reflecting evolving evidence favoring HPV-led strategies in vaccinated cohorts, though Pap cytology retains utility where HPV assays are unavailable.[31] Screening is not routinely recommended before age 21 or after hysterectomy in low-risk cases, as benefits do not outweigh procedural harms.[27] [28]Procedure
Sample collection techniques
The standard Pap test sample collection is performed with the patient lying on her back on an examination table in the lithotomy position, with feet placed in stirrups. A speculum is inserted into the vagina to visualize the cervix, followed by gentle scraping of cells using a spatula and/or brush for laboratory analysis.[32] This step ensures unobstructed sampling of the transformation zone, where precancerous changes most commonly arise.[33] Providers typically collect endocervical cells first using a cytobrush or endocervical brush, which is gently inserted into the external os until the bristles are no longer visible, then rotated 360 degrees clockwise for 5-10 seconds to dislodge cells without causing trauma.[34] Ectocervical sampling follows with an Ayre's or wooden spatula, inserted parallel to the cervix and rotated firmly 360 degrees around the exocervix to scrape squamous epithelial cells, particularly from the squamocolumnar junction.[19] Alternative devices include a single broom-like sampler for non-pregnant individuals with an ectropion, where the device is inserted into the endocervical os and rotated five full turns to capture cells from both endocervical and ectocervical regions simultaneously.[35] In liquid-based cytology (LBC) methods, such as ThinPrep or SurePath, collected cells are rinsed directly into a preservative solution vial rather than smeared on a slide, reducing obscuring factors like blood or mucus; the brush and spatula are swirled or broken off into the vial and discarded.[35] Conventional smear techniques, though less common today, involve spreading the endocervical brush sample along one slide long axis and the spatula sample perpendicularly on the same or a second slide, immediately fixed with 95% ethanol spray or solution to preserve morphology.[36] To optimize sample quality and test accuracy, the Pap test should ideally be scheduled during mid-cycle (approximately days 10-20 of the menstrual cycle or in the first half following the end of menstruation), when estrogen dominance enhances squamous cell maturation and visibility for easier evaluation. It should be avoided during menstruation, as blood can interfere with cell assessment, increase unsatisfactory specimen rates, and reduce accuracy. In the second half of the cycle, progesterone dominance may obscure cytologic details due to changes such as increased leukocytes or altered mucus properties. Other factors that can contaminate or degrade samples include recent douching (within 24-48 hours), vaginal medications, or lubricants; vaginal cuff sampling may be used post-hysterectomy if indicated.[37][38] Self-collection techniques, primarily validated for HPV testing rather than cytology, involve tampon-like devices or swabs but yield lower cellularity for Pap interpretation and are not standard for routine screening.[39] Adequate sampling requires the presence of endocervical or squamous metaplastic cells in the final preparation to confirm transformation zone representation.[33] 3D animations exist to demonstrate the Pap smear procedure for educational purposes, though exact versions featuring an Indian female doctor and patient are uncommon in mainstream medical animations; some Indian health channels provide narrated or animated explanations in Hindi or with local context.Laboratory processing and types
Laboratory processing of Pap test samples involves preparing cervical cells for microscopic evaluation to detect abnormalities indicative of precancerous or cancerous changes. Following collection, specimens are transported to a cytology laboratory where they are accessioned, stained, and mounted for examination by cytotechnologists or pathologists. The Papanicolaou (Pap) stain, a polychromatic method using hematoxylin for nuclear staining, Orange G 6 for keratinized cytoplasm, and modified eosin azure for non-keratinized elements, is standard for highlighting cellular morphology, nuclear details, and cytoplasmic features.[40][20] Two main types of Pap test preparations exist: conventional smears and liquid-based cytology (LBC). In conventional processing, cells are directly smeared onto a glass slide using a spatula or brush, immediately fixed with 95% ethanol or a cytologic spray fixative to preserve morphology and prevent air-drying artifacts, then allowed to dry before transport. Upon lab receipt, slides undergo hydration if needed, followed by the Pap staining sequence: immersion in Harris hematoxylin for nuclear counterstaining, differentiation in acid-alcohol, bluing in alkaline solution, staining with Orange G and eosin azure mixtures, dehydration through graded alcohols, clearing in xylene, and coverslipping. This method, while cost-effective, can result in overlapping cells, obscuring elements like blood or mucus, leading to higher rates of unsatisfactory specimens in some studies, reported at up to 5-10% compared to LBC.[20][41][42] Liquid-based cytology, introduced in the 1990s and now predominant in many settings, collects cells into a preservative liquid medium (e.g., methanol-based for ThinPrep or ethanol-based for SurePath), which disperses and preserves cells while allowing removal of debris. Processing entails vortexing or agitating the vial, then automated deposition of a thin, uniform cell layer onto a slide via filtration (ThinPrep) or density gradient sedimentation (SurePath), followed by the same Pap staining protocol. LBC reduces unsatisfactory rates by minimizing artifacts—studies show 1-2% inadequacy versus 3-5% for conventional—and enables ancillary testing like HPV DNA detection from residual liquid, but randomized trials indicate no significant improvement in sensitivity or specificity for high-grade lesions over conventional methods.[41][43][42] Quality assurance in processing includes evaluating specimen adequacy, requiring at least 10-12 well-preserved squamous cells or presence of endocervical or transformation zone cells to confirm representative sampling. Automated screening aids like the ThinPrep Imaging System review pre-selected fields to enhance efficiency, though manual review remains essential for final interpretation. Variations such as ultrafast Pap staining adapt the procedure for rapid intraoperative assessment but are not routine for screening.[19][44]Reporting and interpretation standards
The Bethesda System for Reporting Cervical Cytology, first developed in 1988 and revised in 2014, provides the standardized framework for interpreting and reporting results from cervicovaginal cytologic specimens, including Pap tests.[44] This system ensures uniform terminology across laboratories to facilitate consistent communication between pathologists, clinicians, and patients.[45] Reports must include specimen type (e.g., conventional smear or liquid-based preparation), adequacy assessment, general categorization, automated review if applicable, cellular interpretation, ancillary testing results, and optional educational notes.[46] Specimen adequacy is evaluated first and is mandatory for reporting. A specimen is deemed satisfactory for evaluation if it contains at least 5,000 nucleated squamous cells in liquid-based preparations (or 8,000–12,000 in conventional smears), with the presence of endocervical or squamous metaplastic cells indicating adequate sampling of the transformation zone; lower cell counts may still qualify if abnormal cells are identified.[44] Unsatisfactory specimens, defined by fewer than 2,000 evaluable cells or obscuration of more than 75% of cells by blood, inflammation, or other factors, require repeat collection and must specify the reason for inadequacy.[45] Interpretation proceeds only on satisfactory specimens, with quality indicators such as transformation zone component noted to assess sampling reliability.[46] General categorization divides reports into "Negative for Intraepithelial Lesion or Malignancy" (NILM) or "Epithelial Cell Abnormality," with the latter subdivided by squamous or glandular origin.[44] NILM encompasses non-neoplastic findings, such as reactive cellular changes from inflammation or infection (e.g., Trichomonas vaginalis, Candida, herpes simplex virus), atrophy, squamous metaplasia, or endometrial cells in women over 40 (which warrant clinical correlation for malignancy risk).[45] Epithelial abnormalities are interpreted based on cytomorphologic criteria, including nuclear size, hyperchromasia, irregular nuclear membranes, and cytoplasmic changes. Squamous epithelial abnormalities are classified as:- Atypical squamous cells of undetermined significance (ASC-US): Mild atypia insufficient for a definitive lesion.
- Atypical squamous cells, cannot exclude high-grade squamous intraepithelial lesion (ASC-H): Features suggestive but not diagnostic of high-grade changes.
- Low-grade squamous intraepithelial lesion (LSIL): Koilocytosis, mild dysplasia corresponding to HPV infection or CIN1.
- High-grade squamous intraepithelial lesion (HSIL): Moderate to severe dysplasia (CIN2/3), with or without invasion suspicion.
- Squamous cell carcinoma: Malignant features like tumor diathesis.[44][45]
- Atypical glandular cells (AGC): Endocervical, endometrial, or not otherwise specified, favoring neoplasia or not.
- Adenocarcinoma in situ (AIS).
- Adenocarcinoma: Endocervical, endometrial, extrauterine, or not otherwise specified.
- Other specified malignancies.[46] Interpretation incorporates adjunctive testing, such as high-risk HPV genotyping (e.g., types 16/18), which refines risk stratification but does not alter core cytologic categories.[44] Computer-assisted screening devices, if used, must be documented, though final pathologist review is required.[45] These standards emphasize descriptive precision to guide clinical management while acknowledging inherent subjectivity in borderline cases like ASC-US.[46]
Clinical Effectiveness
Evidence from randomized trials and observational data
Observational studies, including population-based analyses and case-control designs, provide the strongest evidence for the effectiveness of Pap test screening in reducing cervical cancer incidence and mortality, as large randomized controlled trials directly comparing cytology to no screening have not been feasible in high-income settings due to established practice and ethical constraints.[6] In countries implementing organized cytology programs, such as Finland, Sweden, and Iceland, cervical cancer mortality declined by 50%, 34%, and 80%, respectively, over periods spanning decades following program initiation, with reductions attributed to early detection and treatment of precancerous lesions.[6] Case-control studies further corroborate these findings, showing that unscreened women face 3- to 10-fold higher risks of invasive cervical cancer compared to regularly screened women, with protective effects increasing with screening frequency and recency.[6] A systematic review and meta-analysis of observational data confirmed that cervical screening is associated with reductions in invasive cervical cancer incidence, with odds ratios indicating 40% to 90% lower risk among screened versus unscreened populations, though effect estimates vary by screening coverage and interval.[47] Cohort studies, such as one analyzing U.S. surveillance data, estimated that Pap smear screening averted between 105,000 and 492,000 cervical cancer cases from 1975 to 2009 by preventing progression from dysplasia, after adjusting for pre-screening era baselines.[48] In Sweden, an analysis of screening-attributable mortality calculated a 53% reduction (95% confidence interval: 23–72%) linked to Pap program participation.[49] Randomized trials exist primarily for comparative purposes, such as HPV versus cytology arms, where cytology serves as an active control demonstrating baseline efficacy; for instance, in trials like the Swedish Malmo study, cytology screening yielded cumulative reductions in cervical intraepithelial neoplasia grade 3 or higher, though HPV arms showed superior long-term outcomes.[50] Attendance at screening episodes in observational audits, such as Finland's program, correlates with 41%–92% mortality reductions among participants, underscoring dose-response relationships but highlighting gaps for non-attenders, who account for disproportionate cancer deaths.[51] These data collectively support causal attribution to screening via temporal associations, biological plausibility of interrupting HPV-driven carcinogenesis, and consistency across diverse populations, despite potential confounders like concurrent improvements in healthcare access.[6]Quantitative metrics: sensitivity, specificity, and predictive values
The sensitivity of conventional Pap tests for detecting cervical intraepithelial neoplasia grade 2 or higher (CIN2+), a common endpoint for high-grade lesions, is typically reported in the range of 50% to 70% across studies, reflecting variability due to factors such as laboratory protocols, reader experience, and verification bias in evaluations against colposcopy or biopsy.[52] [53] A 2024 review of screening strategies found conventional cytology sensitivity at 59% for CIN2+, lower than alternatives like HPV testing, while liquid-based cytology shows marginally higher rates, around 60-65% in comparative analyses.[53] [54] Sensitivity for CIN3+ or invasive cancer is somewhat higher, with pooled estimates from individual patient data reaching 76% at an atypical squamous cells of undetermined significance (ASC-US) or worse threshold, though this drops to 45-50% for stricter high-grade squamous intraepithelial lesion (HSIL) cutoffs in primary screening contexts.[55] [54] Specificity for CIN2+ is consistently higher, often exceeding 90%, as the test effectively rules out disease in most negative cases but suffers from trade-offs with sensitivity due to heterogeneous study designs and thresholds.[52] In the same 2024 review, conventional Pap specificity reached 94%, outperforming HPV-based methods in avoiding false positives, while a Latin American evaluation reported 97.5% specificity for cytology versus 91.7% for HPV testing.[53] [54] Earlier meta-analyses confirm this pattern, with specificities from 84% to 97%, negatively correlated with sensitivity estimates due to diagnostic verification issues where only abnormal results prompt histology.[52] Liquid-based methods yield similar specificity, around 92-95%, but real-world performance can decline with high-volume screening due to subtle cytologic changes being missed.[56] Positive predictive value (PPV) for CIN2+ following an abnormal Pap result varies with the cytologic grade and underlying prevalence, typically 20-40% for low-grade abnormalities like LSIL (reflecting regression in many cases) and rising to 60-80% for HSIL referrals.[57] [58] In low-prevalence screening populations (e.g., <1% CIN2+ rate), PPV for ASC-US is often below 10%, necessitating triage, whereas negative predictive value (NPV) exceeds 99% for negative cytology, providing reassurance against high-grade disease for 3-5 years.[59] [60] These values underscore the test's role in interval cancer prevention despite modest PPV, with combined cytology-HPV strategies enhancing overall accuracy in recent evaluations.[61]| Metric | Typical Range for CIN2+ Detection | Key Influencing Factors | Example Pooled Estimate |
|---|---|---|---|
| Sensitivity | 50-70% | Cutoff threshold (ASC-US vs. HSIL), cytology type (conventional vs. liquid-based) | 59% (conventional)[53] |
| Specificity | 90-97% | Population prevalence, verification bias | 94% (conventional)[53] |
| PPV | 20-40% (abnormal result) | Cytologic grade, HPV co-testing | 37% (LSIL/HSIL referrals)[58] |
| NPV | >99% (negative result) | Low disease prevalence in screened cohorts | 99.8% (with triage)[61] |
Impact on cervical cancer incidence and mortality
The introduction of widespread Pap test screening in the mid-20th century correlated with marked declines in cervical cancer incidence and mortality across multiple populations. In the United States, cervical cancer incidence and mortality rates have decreased by over 50% in the four decades leading up to 2020, primarily due to the uptake of Pap smear screening programs that enable early detection and treatment of precancerous lesions. Similarly, population-based studies in Europe and elsewhere attribute reductions of 41% to 92% in cervical cancer mortality to participation in screening, with organized invitation systems yielding 17% to 79% lower mortality compared to non-invited groups.[51] Quantitative evidence from cohort and observational data underscores the screening's causal role in averting advanced disease. Regular Pap screening has been associated with at least an 80% reduction in both cervical cancer incidence and mortality, as evidenced by long-term follow-up in screened versus unscreened women.[6] For instance, screened individuals exhibited a 38% lower risk of cervical cancer death overall, with reductions of 59%, 35%, and 38% for localized, regional, and unknown-stage cancers, respectively, independent of stage at diagnosis.[62] Ecological analyses further estimate that Pap smears prevented 105,000 to 492,000 cervical cancer deaths in the U.S. from 1975 to 2010 by shifting detection toward early-stage disease, evidenced by a halving of early-stage incidence from 9.8 to 4.9 cases per 100,000 women between 1976 and 2009, alongside declines in late-stage cases.[48] These impacts are most pronounced in settings with high screening coverage, such as organized programs in Nordic countries, where a 53% reduction in mortality (95% CI: 23-72%) was directly attributable to screening after adjusting for pre-screening trends.[63] The International Agency for Research on Cancer concludes that cervical screening reduces mortality by 80% or more among adherent women, primarily through prevention of progression from dysplasia to invasive carcinoma via colposcopy-guided interventions.[64] However, residual mortality persists in under-screened subgroups, highlighting coverage as a key limiter, while recent U.S. data show sustained precancer declines (79-80% from 2008-2022 in young women), reinforcing ongoing benefits despite HPV vaccination's emergence.[65]Limitations and Associated Harms
Sources of inaccuracy: false positives and negatives
False-negative results in Pap tests occur when precancerous or cancerous lesions are present but not detected, primarily due to sampling errors where inadequate collection misses abnormal cells from the transformation zone, endocervical canal, or submucosal lesions, accounting for up to 81% of false negatives in some analyses.[66] Obscuring factors such as blood, mucus, infection, or inflammatory exudate can mask abnormal cells on the slide, while douching or vaginal medications may dilute or wash away atypical cells prior to sampling.[28][66] Screening errors involve overlooking subtle abnormalities during initial review by cytotechnologists, and interpretive errors stem from subjective misclassification of atypical cells as benign, with interobserver reproducibility limited (kappa=0.46 for low-grade lesions) and rescreening reclassifying over 50% of normal slides as abnormal in certain studies.[66] These issues contribute to Pap test sensitivity ranging from 30% to 87% for detecting high-grade lesions, with false-negative rates of 13% to 70% reported across reviews.[66] False-positive results arise when benign cellular alterations are misinterpreted as dysplastic or malignant, often leading to unnecessary colposcopy or biopsy. Reactive inflammatory changes from non-neoplastic causes, such as cervicitis or irritation, frequently mimic high-grade squamous intraepithelial lesions through nuclear enlargement, hyperchromasia, and irregular contours.[67][68] Reparative processes following epithelial injury, including those from infections like candida or trichomonas, produce streaming sheets of cells with atypical features that challenge distinction from true dysplasia.[68] Atrophic changes in postmenopausal women cause squamous cells to exhibit enlarged, hyperchromatic nuclei resembling atypia, while immature metaplasia or follicular cervicitis can further confound interpretation.[67] Benign conditions overall represent a common source of false-positive atypia reports, as evidenced by high rates of negative follow-up biopsies after atypical squamous cells of undetermined significance (ASC-US) designations.[69][70] Pap tests detect abnormal cervical cells that may result from HPV infections, and related HPV DNA tests identify high-risk HPV types, but neither can reveal the source of the infection from a specific sexual partner, the timing of acquisition, or details of sexual history such as multiple partners or fidelity. HPV infections can persist latently for years before causing detectable cellular changes or be reactivated after long periods of dormancy, making it impossible to determine when or from whom the virus was contracted based on screening results. Abnormal findings therefore do not indicate recent infection or behavior such as infidelity.[71][72]Overdiagnosis of precancerous lesions
Overdiagnosis in cervical screening refers to the detection of precancerous lesions, such as cervical intraepithelial neoplasia (CIN), that would spontaneously regress or remain subclinical without progressing to invasive cancer, thereby subjecting asymptomatic individuals to unnecessary diagnostic follow-up and potential treatment.[73] This phenomenon arises because Pap tests identify cellular abnormalities across a spectrum of CIN grades, many of which exhibit high rates of natural regression, particularly in younger women.[74] The natural history of CIN1 (low-grade squamous intraepithelial lesion, LSIL) is characterized by spontaneous regression in approximately 80% of cases, often within 2 years, with progression to higher-grade lesions or cancer occurring rarely.[75] For CIN2 (a borderline high-grade lesion), regression rates range from 40% to 60% over 24 months, especially among HPV-negative or young patients, while persistence or progression is less predictable but lower than for CIN3.[76] 00542-2/fulltext) CIN3 lesions show lower regression, with complete resolution to normal tissue in about 14% of observed cases and partial regression to CIN1 or CIN2 in another 9-17%, though most persist or progress without intervention.[77] In population screening, Pap cytology contributes to overdiagnosis by flagging these regressive lesions for colposcopy and biopsy, amplifying detection of non-progressive abnormalities; randomized trials and observational data indicate that screening in adolescents and women under 21 yields minimal cancer prevention benefits but increases overtreatment of transient precancers by up to 50-fold compared to older cohorts.[74] [78] Longitudinal studies confirm that many CIN2+ diagnoses represent overdiagnosis, as autopsy and untreated cohort data reveal that only a fraction of such lesions would evolve into invasive disease within lifetimes, with regression favored by immune clearance of transient HPV infections underlying most low-grade changes.[79] This underscores the need for risk-stratified management, such as observation for CIN1 and select CIN2 cases, to mitigate harms from interventions like excision, which carry risks of cervical incompetence and preterm birth.[80]Overtreatment risks and downstream complications
Overtreatment in cervical screening arises from the excision or ablation of precervical lesions, particularly cervical intraepithelial neoplasia (CIN) grades 1 and 2, many of which regress spontaneously without progressing to invasive cancer. Studies indicate regression rates of approximately 60% for CIN1 lesions and 40-70% for CIN2, with higher rates (up to 60%) observed in women under 30 years old during short-term follow-up. [77] [81] [82] For instance, in a cohort of untreated CIN2 cases, 44.1% regressed within 12 months, and cumulative regression reached 55.4% by 24 months. [82] [83] Immediate intervention thus exposes women to procedural risks without averting disease in a substantial proportion of cases, particularly when triggered by abnormal Pap results that prompt colposcopy and biopsy. [78] Common overtreatment procedures include loop electrosurgical excision procedure (LEEP) and cold knife cone biopsy, performed for confirmed CIN2 or higher to remove potentially precancerous tissue. These interventions, while effective for high-grade lesions, carry downstream complications such as bleeding, infection, and cervical stenosis, which can impair menstrual flow or fertility. [6] [84] More critically, excisional treatments increase the risk of adverse obstetric outcomes in subsequent pregnancies, including preterm birth and low birth weight. A meta-analysis linked LEEP conization to elevated preterm delivery risk, with odds ratios heightened among women without prior preterm history. [85] [86] The association with preterm birth stems from removal of cervical tissue, which may compromise structural integrity and lead to cervical incompetence. Reviews of conization effects report consistent increases in preterm delivery rates, influenced by factors like specimen volume and procedure type, with second-trimester cervical shortening as a predictor. [87] [88] Although one recent study found comparable preterm risks between surveillance and immediate LEEP for CIN2, broader evidence supports a small but significant elevation in adverse outcomes post-procedure, affecting a minority of women but underscoring the harm of unnecessary treatment. [89] [90] These complications highlight the trade-off in screening programs, where detection of transient abnormalities prompts interventions that, for regressing lesions, yield net harm rather than benefit. [91]Complementary and Alternative Screening Methods
Integration with HPV DNA testing
The integration of human papillomavirus (HPV) DNA testing with the Pap test addresses limitations in cytology sensitivity by detecting high-risk HPV types responsible for nearly all cervical cancers. In co-testing, both liquid-based cytology and polymerase chain reaction or hybrid capture assays for high-risk HPV (hrHPV) are performed concurrently on the same sample, allowing for risk stratification: negative results on both confer low risk, while hrHPV positivity prompts further evaluation regardless of cytology, and abnormal cytology with negative HPV may warrant observation. This approach, applicable to women aged 30–65 years, extends screening intervals to 5 years for dual negatives, as endorsed by the United States Preventive Services Task Force (USPSTF) in its 2018 guidelines and reaffirmed in the December 2024 draft recommendation.[27][29] Randomized controlled trials, including pooled analyses from European studies, demonstrate that co-testing yields higher sensitivity for detecting cervical intraepithelial neoplasia grade 3 or greater (CIN3+)—approximately 95–100% versus 60–80% for cytology alone—while providing comparable long-term protection against invasive cervical cancer to primary HPV testing. A meta-analysis of three such trials found co-testing reduced CIN3+ detection at subsequent rounds compared to cytology, though with marginally lower specificity (around 90% versus 95% for HPV alone), leading to more colposcopy referrals. Observational data from U.S. implementation corroborate these findings, showing co-testing detects persistent hrHPV infections causal to precancerous lesions earlier than cytology, which primarily identifies downstream morphologic changes that may regress spontaneously in up to 50% of cases.[92][93] HPV testing also serves as triage for equivocal Pap results, such as atypical squamous cells of undetermined significance (ASC-US), where hrHPV negativity identifies over 90% of low-risk cases for routine rescreening, avoiding unnecessary procedures and improving efficiency over repeat cytology. The American College of Obstetricians and Gynecologists (ACOG) and American Cancer Society (ACS) guidelines from 2021 and 2023, respectively, position co-testing as an acceptable alternative to primary hrHPV screening every 5 years, particularly where standalone HPV assays are unavailable, though they emphasize primary HPV's superior negative predictive value (over 99% for CIN3+ at 5–6 years). Integration has contributed to declining U.S. cervical cancer incidence, with hrHPV-positive cases triaged to colposcopy reducing missed precancers by 30–50% relative to cytology-only protocols in cohort studies.[30][94][95] Despite enhanced detection, co-testing's dual thresholds can amplify over-referral for transient HPV infections in women under 30, where prevalence exceeds 20% but cancer risk remains low; thus, guidelines restrict it to ages 30 and older to minimize harms. Cost-effectiveness analyses indicate co-testing is comparable to primary HPV in high-resource settings, with quality-adjusted life years gained similar to cytology alone but at higher upfront costs due to assay expenses, estimated at $50–100 per test in U.S. implementations. Ongoing refinements, informed by HPV vaccination reducing hrHPV prevalence by 80–90% in vaccinated cohorts, prioritize de-intensification for low-risk dual negatives to balance benefits against procedural risks like anxiety and biopsy complications.[6][96]Comparison of Pap test to primary HPV screening
Primary HPV screening detects high-risk human papillomavirus (hrHPV) DNA or RNA as the initial test for cervical cancer precursors, with reflex cytology or genotyping for positive results, whereas the Pap test relies on cytological examination of cervical cells to identify atypical or malignant changes. Randomized controlled trials, such as the New Technologies for Cervical Cancer (NTCC) trial in Italy, have demonstrated that HPV testing exhibits substantially higher sensitivity for detecting cervical intraepithelial neoplasia grade 2 or worse (CIN2+), with a 39.2% relative increase over Pap cytology (HPV sensitivity approximately 96% vs. Pap 55-68%), though specificity is modestly lower (HPV 90-94% vs. Pap 93-97%).[97][98][99] Meta-analyses of screening strategies confirm primary HPV testing's superior performance in identifying CIN2+, with pooled sensitivity exceeding 90% compared to 40-60% for cytology, enabling detection of persistent infections that precede cytological abnormalities by years; however, this advantage incurs higher rates of unnecessary colposcopies due to transient HPV infections that regress without progression, affecting specificity and increasing overdiagnosis risks.[54][100] In long-term follow-up from trials like NTCC and ARTISTIC, HPV-based screening reduced CIN3+ incidence by 50-60% over 6-10 years relative to cytology, with a 40% lower invasive cancer rate in meta-analyses, attributed to earlier intervention on high-risk cases, though cytology's higher specificity minimizes immediate procedural harms.[27][97]| Metric | Pap Test (Cytology) | Primary HPV Testing |
|---|---|---|
| Sensitivity for CIN2+ | 40-68% | 90-96% |
| Specificity for CIN2+ | 93-97% | 90-94% |
| CIN3+ Detection (relative) | Baseline | 50-60% greater |
| Colposcopy Referrals | Lower | 2-3x higher |
Emerging self-sampling and automated technologies
Self-sampling technologies enable individuals to collect vaginal or cervicovaginal specimens at home or in clinical settings without requiring a pelvic examination by a clinician, primarily for human papillomavirus (HPV) detection as a complement or alternative to traditional Pap cytology. A 2024 U.S. Food and Drug Administration (FDA) approval permits self-collection of vaginal samples for certain HPV tests in health care settings, demonstrating detection rates comparable to clinician-collected samples, with pooled sensitivity for high-risk HPV of approximately 92-95% in meta-analyses of randomized trials.[102] [103] These kits, often using brushes like the Evalyn Brush or Cervex-Brush, have shown higher screening uptake—up to 20-30% increases in participation rates among hard-to-reach populations—due to reduced barriers such as embarrassment, access, and discomfort associated with speculum-based Pap collection.[104] [105] However, direct self-sampling for cytological analysis (akin to Pap smears) yields lower adequacy and sensitivity for detecting squamous intraepithelial lesions, with clinician-collected samples outperforming self-collected by 10-15% in detecting high-grade lesions in comparative studies, limiting its standalone use for cytology-based screening.[104] Automated technologies for Pap test interpretation leverage artificial intelligence (AI) and digital imaging to assist cytotechnologists in slide review, addressing inter-observer variability and workload burdens in manual microscopy. The Hologic Genius Digital Diagnostics System, FDA-cleared in 2023 as the first fully digital cytology platform, employs AI algorithms to scan liquid-based cytology slides, prioritize regions of interest, and reduce full slide review time by up to 33% while maintaining or improving abnormality detection rates.[106] Deep learning models, validated in 2024 studies on digitized whole-slide images, achieve AUC values exceeding 0.95 for classifying cervical cytology as negative, low-grade, or high-grade intraepithelial lesions, outperforming traditional manual screening in sensitivity for precancerous changes by identifying subtle morphological features like nuclear irregularities.[107] [108] Collaborations such as BD and Techcyte's AI-based digital cytology system digitize slides for remote review, enhancing scalability in resource-limited settings, though prospective trials report false-negative reductions of 5-10% but require human oversight to mitigate AI errors in rare atypical cases.[109] Low-cost AI-microscopy platforms, developed in 2025 prototypes, integrate computer vision for automated cell classification in conventional smears, achieving over 90% concordance with expert pathologists in pilot evaluations from low-resource contexts.[110] These advancements prioritize empirical validation against gold-standard histopathology, yet ongoing concerns include algorithmic bias from training datasets skewed toward high-prevalence populations and the need for regulatory standardization to ensure generalizability across diverse demographics.[107]Historical Development
Invention by George Papanicolaou
George Nicholas Papanicolaou, a Greek-born physician and researcher, developed the foundational technique of the Pap test through studies in exfoliative cytology beginning in the early 20th century. Arriving in the United States in 1913, he joined Cornell University Medical College's Department of Anatomy in 1914, where he initially investigated reproductive cycles in guinea pigs using vaginal smears to identify estrous phases via cellular changes observed under a microscope.[111] By 1916, this approach allowed precise timing of guinea pig reproductive cycles, laying groundwork for applying similar cytological methods to humans.[112] In 1920, Papanicolaou extended his research to human vaginal smears, starting with his wife Mary as the initial subject to map menstrual cycle variations through epithelial cell morphology.[111] Examining smears from patients at Cornell Clinic and later in collaboration with Woman's Hospital from 1925, he noted atypical cellular features in cases of known uterine pathology, including enlarged, irregular nuclei indicative of malignancy during routine cycle studies.[111] This led to the core innovation: non-invasive sampling of exfoliated cervical and vaginal cells via speculum insertion, followed by microscopic analysis to detect early cancerous or precancerous changes, distinct from invasive biopsy methods.[113] Papanicolaou first publicly presented the cancer-detection potential of vaginal smears in 1928 at the Third Race Betterment Conference, describing it as "New Cancer Diagnosis" based on observations of abnormal cells in smears from affected patients; however, the medical community largely overlooked these findings at the time.[111] He refined the technique, including a specialized staining protocol—later termed the Papanicolaou stain—using hematoxylin, orange G, and EA (a polychrome mixture) to enhance nuclear and cytoplasmic detail for accurate classification of cell types.[112] Mary Papanicolaou contributed extensively as lab technician and daily smear provider for calibration over two decades, enabling consistent methodological validation.[112] Renewed efforts in 1939, spurred by Cornell's Dr. Joseph Hinsey and collaboration with gynecologist Herbert F. Traut, involved systematic testing on thousands of cases at New York Hospital, confirming the smear's diagnostic value for asymptomatic uterine carcinoma.[111] In 1941, they published "The Diagnostic Value of Vaginal Smears in Carcinoma of the Uterus" in the American Journal of Obstetrics and Gynecology, analyzing smears from over 300 patients and demonstrating detection rates for early lesions.[113] This culminated in their 1943 monograph, Diagnosis of Uterine Cancer by the Vaginal Smear, documenting over 3,000 cases and establishing the Pap test as a reproducible, low-cost screening tool reliant on cytological criteria rather than symptomatic presentation.[113][111]Path to clinical adoption and standardization
Following the initial publication of the Papanicolaou method in 1928 and its validation through clinical studies by 1941, adoption faced initial resistance due to skepticism among physicians regarding cytology's reliability for early cancer detection.[3] A pivotal advancement occurred in 1943 with the publication of Diagnosis of Uterine Cancer by the Vaginal Smear by George Papanicolaou and Herbert Traut, which provided detailed protocols and case evidence, prompting demonstrations at institutions like Harlem Hospital in New York City that year. These efforts led to rapid uptake in urban medical centers, as gynecologists observed high sensitivity in identifying precancerous lesions, reducing reliance on more invasive biopsies.[114] By the late 1940s, the Pap test had become a standard component of women's healthcare in the United States, coinciding with post-World War II public health initiatives that emphasized preventive screening amid cervical cancer's status as a leading cause of female mortality.[111] Nationwide programs expanded in the 1950s and 1960s, with clinical trials confirming efficacy in detecting squamous intraepithelial lesions, leading to routine integration into gynecological practice by the early 1960s.[115] This era marked a shift from opportunistic testing to systematic screening, supported by organizations like the American Cancer Society, which advocated for annual exams, though evidence later tempered frequency recommendations.[116] Standardization of reporting lagged behind adoption, initially relying on the Papanicolaou classification system (Classes I–V, denoting normal to malignant cells), which suffered from inter-observer variability and inconsistent terminology across labs. In December 1988, the National Cancer Institute convened a workshop in Bethesda, Maryland, resulting in The Bethesda System (TBS), a uniform framework for cervical cytology interpretation that emphasized descriptive diagnoses like atypical squamous cells of undetermined significance (ASC-US) and low-grade squamous intraepithelial lesion (LSIL).[117] TBS revisions in 1991, 2001, and 2014 refined criteria to incorporate ancillary testing and reduce ambiguity, facilitating global comparability and quality control in screening programs.[44] This system addressed prior inconsistencies, improving diagnostic reproducibility without altering the core smearing technique.Post-adoption refinements amid HPV discoveries
The identification of high-risk human papillomavirus (HPV) types, particularly HPV-16 and HPV-18, as the primary causative agents of cervical cancer by Harald zur Hausen in the mid-1980s prompted a reevaluation of cytological screening practices, revealing that most Pap test abnormalities represented transient HPV infections rather than inevitable progression to malignancy.[116] This causal insight shifted focus from cytology alone to risk stratification, emphasizing persistent high-risk HPV infection as the key precursor to high-grade lesions. Early refinements included the 1988 introduction of the Bethesda System for reporting cervical cytology, which standardized terminology such as atypical squamous cells of undetermined significance (ASC-US), low-grade squamous intraepithelial lesion (LSIL), and high-grade squamous intraepithelial lesion (HSIL), facilitating more consistent interpretation amid emerging virological data.[45] Subsequent revisions in 1991, 2001, and 2014 incorporated nuances like HPV-related changes to refine diagnostic categories and reduce interpretive variability.[45] Technological advancements in sample preparation paralleled these discoveries, with the U.S. Food and Drug Administration (FDA) approving liquid-based cytology (LBC) methods, such as ThinPrep in 1996, as alternatives to conventional smears.[118] LBC minimized obscuring artifacts like blood and mucus—common in up to 20% of conventional slides—improving cellular preservation and readability while enabling residual sample use for molecular HPV assays.[119] This preparation technique increased sensitivity for detecting high-grade lesions by 5-10% compared to smears and laid the groundwork for integrated testing, addressing Pap test limitations such as 20-40% false-negative rates attributed to sampling errors and transient HPV-driven changes.[120] HPV DNA testing emerged as a critical adjunct, with the Hybrid Capture 2 (HC2) assay receiving FDA approval in 1999 for triaging ASC-US results, identifying high-risk HPV to prioritize colposcopy and averting unnecessary procedures in low-risk cases where cytology alone over-referred up to 70% of women with benign transient infections.[116] The American Society for Colposcopy and Cervical Pathology (ASCCP) formalized this in 2001 consensus guidelines, recommending high-risk HPV testing for ASC-US management, which reduced colposcopy referrals by 40-60% while maintaining detection of precancerous lesions.[121] By 2003, FDA clearance extended HC2 use to co-testing with cytology for women aged 30 and older, enabling every-5-year screening intervals for double-negative results, as HPV negativity provided >99% negative predictive value for high-grade disease over 5-10 years.[116] These protocols, validated in trials like ALTS (ASCUS/LSIL Triage Study), demonstrated superior efficiency over cytology alone, though implementation highlighted challenges like higher upfront costs and the need for triage algorithms to manage HPV-positive, cytology-negative findings.[121]Guidelines and Global Implementation
Major organizational recommendations (e.g., USPSTF, WHO, ACS)
The United States Preventive Services Task Force (USPSTF) recommends cervical cytology (Pap test) every 3 years for women aged 21-29; for ages 30-65, primary high-risk human papillomavirus (hrHPV) testing every 5 years is preferred, with cytology alone every 3 years or co-testing (cytology plus hrHPV) every 5 years as acceptable alternatives.[29] Screening should cease after age 65 for those with adequate prior negative results and no high-risk history.[29] The American Cancer Society (ACS) advises against routine screening before age 25 due to low cervical cancer incidence in younger women; for ages 25-65 at average risk, primary hrHPV testing every 5 years is recommended as the preferred method, with cytology alone every 3 years acceptable only if hrHPV is unavailable.[122] Co-testing is not routinely endorsed. Discontinue after 65 with three consecutive negative hrHPV tests or two negative co-tests in the prior decade, absent high risk.[95] The World Health Organization (WHO) prioritizes hrHPV DNA testing as the primary screening modality globally, particularly in resource-limited settings, with cytology reserved for triage or where HPV testing is inaccessible; screening initiation at age 30 with HPV every 5 years (or 10 years if triage-negative) is suggested, extending to 49-65 based on context and HIV status.[123] WHO endorses "screen-and-treat" or "screen-triage-treat" approaches to pre-cancer lesions, emphasizing equity and feasibility over cytology-alone strategies in low-income regions.[124] The American College of Obstetricians and Gynecologists (ACOG) aligns closely with USPSTF, recommending cytology every 3 years from ages 21-29 (with optional hrHPV co-testing at 25-29 if preferred); for 30-65, options include primary hrHPV every 5 years, co-testing every 5 years, or cytology alone every 3 years.[30] Cessation follows USPSTF criteria post-65.[28]| Organization | Screening Start Age | Cytology (Pap) Frequency (Ages 21-29) | Preferred Method (Ages 30-65) | Cessation Criteria |
|---|---|---|---|---|
| USPSTF | 21 | Every 3 years | hrHPV every 5 years (cytology or co-test alternatives) | Age 65+ with adequate negatives, no high risk[29] |
| ACS | 25 | Not routine (use only if HPV unavailable) | hrHPV every 5 years (cytology alternative if needed) | Age 65+ with recent negatives, no high risk[122] |
| WHO | 30 (contextual) | Triage or limited use | hrHPV every 5-10 years | Resource-dependent, post-65 if low risk[123] |
| ACOG | 21 | Every 3 years (HPV optional 25+) | hrHPV, co-test, or cytology every 3-5 years | Aligns with USPSTF post-65[30] |