Recent from talks
Nothing was collected or created yet.
Balance disorder
View on Wikipedia| Balance disorder | |
|---|---|
 | |
| The image shows the labyrinth of the inner ear, and labels the semicircular canals, which help maintain balance. | |
| Specialty | Neurology, Otolaryngology |
| Symptoms | Unsteadiness, wooziness, dizziness, giddiness, sense of floating, vertigo, nausea |
| Diagnostic method | Hearing and vision tests, ENG, VNG, rotary chair test, computerized dynamic posturography |
| Treatment | Vestibular rehabilitation, medication, surgery, Tai chi |
A balance disorder is a disturbance that causes an individual to feel unsteady, for example when standing or walking. It may be accompanied by feelings of giddiness, or wooziness, or having a sensation of movement, spinning, or floating. Balance is the result of several body systems working together: the visual system (eyes), vestibular system (ears) and proprioception (the body's sense of where it is in space). Degeneration or loss of function in any of these systems can lead to balance deficits.[1]
Signs and symptoms
[edit]Cognitive dysfunction (disorientation) may occur with vestibular disorders. Cognitive deficits are not just spatial in nature, but also include non-spatial functions such as object recognition memory.[citation needed] Vestibular dysfunction has been shown to adversely affect processes of attention and increased demands of attention can worsen the postural sway associated with vestibular disorders. Recent MRI studies also show that humans with bilateral vestibular damage (damage to both inner ears) undergo atrophy of the hippocampus which correlates with their degree of impairment on spatial memory tasks.[2][3]
Causes
[edit]Problems with balance can occur when there is a disruption in any of the vestibular, visual, or proprioceptive systems. Abnormalities in balance function may indicate a wide range of pathologies from causes like inner ear disorders, low blood pressure, brain tumors, and brain injury including stroke.[citation needed]
Related to the ear
[edit]
Causes of dizziness related to the ear are often characterized by vertigo (spinning) and nausea. Nystagmus (flickering of the eye, related to the Vestibulo-ocular reflex [VOR]) is often seen in patients with an acute peripheral cause of dizziness.[citation needed]
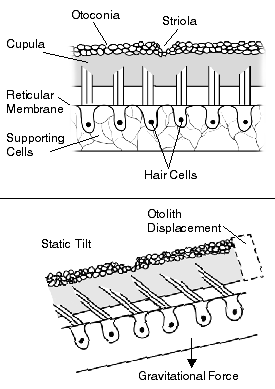
- Benign paroxysmal positional vertigo (BPPV) – The most common cause of vertigo. It is typically described as a brief, intense sensation of spinning that occurs when there are changes in the position of the head with respect to gravity. An individual may experience BPPV when rolling over to the left or right, upon getting out of bed in the morning, or when looking up for an object on a high shelf.[4] The cause of BPPV is the presence of normal but misplaced calcium crystals called otoconia, which are normally found in the utricle and saccule (the otolith organs) and are used to sense movement. If they fall from the utricle and become loose in the semicircular canals, they can distort the sense of movement and cause a mismatch between actual head movement and the information sent to the brain by the inner ear, causing a spinning sensation.[4]
Migraine
[edit]Migraine headaches are a common neurological disease. Although typical migraines are characterized by moderate to severe throbbing headaches, vestibular migraines may be accompanied by symptoms of vestibular disorders such as dizziness, disequilibrium, nausea, and vomiting.[5]
Presyncope
[edit]Presyncope is a feeling of lightheadedness or simply feeling faint. Syncope, by contrast, is actually fainting. A circulatory system deficiency, such as low blood pressure, can contribute to a feeling of dizziness when one suddenly stands up.[6]
Diagnosis
[edit]The difficulty of making the right vestibular diagnosis is reflected in the fact that in some populations, more than one-third of the patients with a vestibular disease consult more than one physician – in some cases up to more than fifteen.[7]
Treatment
[edit]There are various options for treating balance disorders. One option includes treatment for a disease or disorder that may be contributing to the balance problem, such as ear infection, stroke, multiple sclerosis, spinal cord injury, Parkinson's, neuromuscular conditions, acquired brain injury, cerebellar dysfunctions and/or ataxia, or some tumors, such as acoustic neuroma. Individual treatment will vary and will be based upon assessment results including symptoms, medical history, general health, and the results of medical tests. Additionally, tai chi may be a cost-effective method to prevent falls in the elderly.[8]
Vestibular rehabilitation
[edit]Many types of balance disorders will require balance training, prescribed by an occupational therapist or physiotherapist. Physiotherapists often administer standardized outcome measures as part of their assessment in order to gain useful information and data about a patient's current status. Some standardized balance assessments or outcome measures include but are not limited to the Functional Reach Test, Clinical Test for Sensory Integration in Balance (CTSIB),[9] Berg Balance Scale and/or Timed Up and Go[10] The data and information collected can further help the physiotherapist develop an intervention program that is specific to the individual assessed. Intervention programs may include training activities that can be used to improve static and dynamic postural control, body alignment, weight distribution, ambulation, fall prevention and sensory function.[11]
Bilateral vestibular loss
[edit]Dysequilibrium arising from bilateral loss of vestibular function – such as can occur from ototoxic drugs such as gentamicin – can also be treated with balance retraining exercises (vestibular rehabilitation) although the improvement is not likely to be full recovery.[12][13]
Research
[edit]Scientists at the National Institute on Deafness and Other Communication Disorders (NIDCD) are working to understand the various balance disorders and the complex interactions between the labyrinth, other balance-sensing organs, and the brain. NIDCD scientists are studying eye movement to understand the changes that occur in aging, disease, and injury, as well as collecting data about eye movement and posture to improve diagnosis and treatment of balance disorders. They are also studying the effectiveness of certain exercises as a treatment option.[14] Recently, a study published in JAMA Otolaryngology-Head & Neck Surgery found that balance problems are an indicator of mortality potentially due to altered metabolism of vestibular system.[15]
See also
[edit]References
[edit]- ^ Sturnieks DL, St George R, Lord SR (December 2008). "Balance disorders in the elderly". Neurophysiol Clin. 38 (6): 467–78. doi:10.1016/j.neucli.2008.09.001. PMID 19026966. S2CID 10370640.
- ^ Smith PF, Zheng Y, Horii A, Darlington CL (2005). "Does vestibular damage cause cognitive dysfunction in humans?". J Vestib Res. 15 (1): 1–9. doi:10.3233/VES-2005-15101. PMID 15908735.
- ^ Brandt T, Schautzer F, Hamilton DA, Brüning R, Markowitsch HJ, Kalla R, Darlington C, Smith P, Strupp M (November 2005). "Vestibular loss causes hippocampal atrophy and impaired spatial memory in humans". Brain. 128 (Pt 11): 2732–41. doi:10.1093/brain/awh617. PMID 16141283.
- ^ a b Bhattacharyya N, Baugh RF, Orvidas L, Barrs D, Bronston LJ, Cass S, Chalian AA, Desmond AL, Earll JM, Fife TD, Fuller DC, Judge JO, Mann NR, Rosenfeld RM, Schuring LT, Steiner RW, Whitney SL, Haidari J (November 2008). "Clinical practice guideline: benign paroxysmal positional vertigo". Otolaryngol Head Neck Surg. 139 (5 Suppl 4): S47–81. doi:10.1016/j.otohns.2008.08.022. PMID 18973840. S2CID 16175316.
- Lay summary in: "ENT doctors release national guideline on treatment for common cause of dizziness". EurekAlert.org (Press release). November 1, 2008.
- ^ Carvalho, Gabriela F.; Luedtke, Kerstin; Bevilaqua-Grossi, Debora (August 2023). "Balance disorders and migraine". Musculoskeletal Science & Practice. 66 102783. doi:10.1016/j.msksp.2023.102783. ISSN 2468-7812. PMID 37263900.
- ^ "Balance Disorders Symptoms, Causes, Treatment – What are the symptoms of a balance disorder?". MedicineNet. Retrieved 2014-03-02.
- ^ van de Berg R, van Tilburg M, Kingma H (2015). "Bilateral Vestibular Hypofunction: Challenges in Establishing the Diagnosis in Adults". ORL J Otorhinolaryngol Relat Spec. 77 (4): 197–218. doi:10.1159/000433549. PMID 26366566. S2CID 3667457.
- ^ Fife TD, Iverson DJ, Lempert T, Furman JM, Baloh RW, Tusa RJ, Hain TC, Herdman S, Morrow MJ, Gronseth GS (May 2008). "Practice parameter: therapies for benign paroxysmal positional vertigo (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology". Neurology. 70 (22): 2067–74. doi:10.1212/01.wnl.0000313378.77444.ac. PMID 18505980. S2CID 24158281.
- ^ "Clinical Test of Sensory Interaction on Balance (CTSIB) | Interacoustics". www.interacoustics.com. Retrieved 2025-08-29.
- ^ O'Sullivan S, Schmitz T (August 2006). "8". In O'Sullivan S (ed.). Physical Rehabilitation. Vol. 5. F.A. Davis. ISBN 978-0-8036-1247-1.
- ^ O'Sullivan S, Schmitz T (August 2006). "13". In O'Sullivan S (ed.). Physical Rehabilitation. Vol. 5. F. A. Davis Company. ISBN 978-0-8036-1247-1.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Horak FB (2010). "Postural compensation for vestibular loss and implications for rehabilitation". Restor Neurol Neurosci. 28 (1): 57–68. doi:10.3233/RNN-2010-0515. PMC 2965039. PMID 20086283.
- ^ Alrwaily M, Whitney SL (2011). "Vestibular rehabilitation of older adults with dizziness". Otolaryngologic Clinics of North America. 44 (2): 473–496. doi:10.1016/j.otc.2011.01.015. PMID 21474018.
- ^ "Balance Disorders". National Institute on Deafness and Other Communication Disorders.
- ^ Cao C, Cade WT, Li S, McMillan J, Friedenreich C, Yang L (May 2021). "Association of Balance Function With All-Cause and Cause-Specific Mortality Among US Adults". JAMA Otolaryngol Head Neck Surg. 147 (5): 460–468. doi:10.1001/jamaoto.2021.0057. PMC 7953337. PMID 33704356.