Recent from talks
Nothing was collected or created yet.
Sardinian dhole
View on Wikipedia
| Sardinian dhole Temporal range: Middle Pleistocene-Holocene
| |
|---|---|

| |
| Drawing of the skeleton of C. sardous scale bar = 20 cm | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Chordata |
| Class: | Mammalia |
| Order: | Carnivora |
| Family: | Canidae |
| Subfamily: | Caninae |
| Tribe: | Canini |
| Genus: | †Cynotherium Studiati, 1857 |
| Type species | |
| Cynotherium sardous Studiati, 1857
| |
| Other species | |
| |
| |
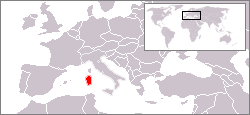
| Former range (in red) | |
The Sardinian dhole (genus Cynotherium especially C. sardous) is an extinct insular canid which was endemic to what is now the Mediterranean islands of Sardinia and Corsica during the Middle-Late Pleistocene. It went extinct at the end of the Pleistocene around the time of human settlement of the islands.
Its scientific name means "dog-beast of Sardinia", the genus name from the Ancient Greek: θήρ, romanized: thḗr, lit. 'beast', 'wild animal' and κύων, kyōn, 'dog' and the specific name from the Latin: Sardous, lit. 'of Sardinia', alt. form of Latin: Sardus.
Around the size of a jackal, morphological studies indicate it was a specialised stalking predator of small mammals and birds, with a probable preference for the endemic Sardinian pika.
Evolution
[edit]The oldest remains of Cynotherium on Sardinia date to around the early-Middle Pleistocene transition around 800,000 years ago, associated with a faunal turnover event on Sardinia likely caused low sea levels allowing dispersal to Sardinia-Corsica from mainland Italy. Cynotherium is suggested to have originated from the species Xenocyon lycaonoides of mainland Europe. Some of the older Cynotherium remains are assigned to the separate species Cynotherium malatestai, which is larger than C. sardous and shows intermediate features between it and X. lycaonoides.[1] A 2021 genetic study found that its closest living relative is the dhole, from which it diverged approximately 885,000 years ago. The study found that the lineage of the Sardinan dhole and dhole emerged from the hybridisation between a lineage closely related to the genus Canis and a lineage related to the African wild dog (Lycaon pictus) in proportions of roughly 65% and 35% respectively. The modern dhole was also found to have additional African wild dog related ancestry (making up around 25% of its genome) not found in the Sardinan dhole.[2]
Description
[edit]

C. sardous was relatively small in size, with a weight around 10 kilograms (22 lb), comparable to a jackal.[3] The skull is slender and wedge-shaped in lateral view. The snout is narrower than in extant dholes, but broader than those of foxes. The postorbital region of the skull is broad, and the zygomatic arches only modestly project outwards. The mastoid is very enlarged and projects outwards. The sagittal crest is weakly developed. The mandible is slender.[4] The attachment sites for the triceps on the scapula, ulna and humerus are large, indicating the muscle, along with the anconeus, was well developed. The scar for the deltoid muscle at the posterior of the deltoid ridge on the humerus is high, large and rugose, indicating this muscle was also well developed.[3] Analysis of its ear morpology, including the significant reduction in the number of cochlear turns, suggests a specialisation towards the hearing of high-frequency sounds, but suggests it was poor at detecting low frequency sounds, and completely unable to detect sounds lower than 250 Hz.[5]
Paleobiology and paleoecology
[edit]
When the ancestor of this canid became confined to the island, its diet became limited to small prey. Cynotherium as a predator specializing in small, fast moving prey is supported by an examination of the animal's anatomy. The evolution of short, powerful limbs, a low neck carriage and increased head and neck mobility suggests an animal specialised for stalking low to the ground, and then quickly pouncing on or running down prey.[4][3] A suggested preferred prey item of the Sardinian dhole is the Sardinian pika (Prolagus sardus), a large lagomorph that was also endemic and abundant on the island, with birds also being likely targets.[4]
During the Middle and Late Pleistocene Corsica and Sardinia had their own highly endemic depauperate terrestrial mammal fauna which besides the Sardinian dhole and Sardinian pika, included the Tyrrhenian field rat, (Rhagamys orthodon) the Tyrrhenian vole (Microtus henseli), a shrew (Asoriculus similis), a mole (Talpa tyrrhenica), a dwarf mammoth (Mammuthus lamarmorai), a galictine mustelid (Enhydrictis galictoides), three species of otter (Algarolutra majori, Sardolutra ichnusae, Megalenhydris barbaricina) and a deer (Praemegaceros cazioti).[6]
Extinction
[edit]The youngest radiocarbon dates for the Sardinan dhole are around 11,500 years BC/13,500 years Before Present (BP), only a few thousand years before the first confirmed human presence on Sardian-Corsica around 8,000 BC/10,000 years BP, and it is assumed that the species was alive when humans arrived on the island. The causes of its extinction are uncertain.[7] The low genetic diversity of sampled individuals suggests that the population size had been small but stable for a long period of time prior to extinction.[2]
See also
[edit]References
[edit]- ^ Madurell-Malapeira, Joan; Palombo, Maria Rita; Sotnikova, Marina (2015-07-04). "Cynotherium malatestai, sp. nov. (Carnivora, Canidae) from the early middle Pleistocene deposits of Grotta dei Fiori (Sardinia, Western Mediterranean)". Journal of Vertebrate Paleontology. 35 (4) e943400. Bibcode:2015JVPal..35E3400M. doi:10.1080/02724634.2014.943400. ISSN 0272-4634. S2CID 129741290.
- ^ a b Ciucani, Marta Maria; Jensen, Julie Kragmose; Sinding, Mikkel-Holger S.; Smith, Oliver; Lucenti, Saverio Bartolini; Rosengren, Erika; Rook, Lorenzo; Tuveri, Caterinella; Arca, Marisa; Cappellini, Enrico; Galaverni, Marco; Randi, Ettore; Guo, Chunxue; Zhang, Guojie; Sicheritz-Pontén, Thomas (December 2021). "Evolutionary history of the extinct Sardinian dhole". Current Biology. 31 (24): 5571–5579.e6. Bibcode:2021CBio...31E5571C. doi:10.1016/j.cub.2021.09.059. hdl:2158/1252046. PMID 34655517. S2CID 238996621.
- ^ a b c Lyras G.; Van der Geer A. (2006). "Adaptations of the Pleistocene island canid Cynotherium sardous (Sardinia, Italy) for hunting small prey". Cranium. 23 (1): 51–60. PDF copy
- ^ a b c Lyras G.A.; Van der Geer A.E.; Dermitzakis M.; De Vos J. (2006). "Cynotherium sardous, an insular canid (Mammalia: Carnivora) from the Pleistocene of Sardinia (Italy), and its origin". Journal of Vertebrate Paleontology. 26 (3): 735–745. doi:10.1671/0272-4634(2006)26[735:CSAICM]2.0.CO;2. S2CID 84448363.
- ^ Zedda, Marco; Brunetti, Antonio; Palombo, Maria Rita (2022-03-25). "First Attempt to Infer Sound Hearing and Its Paleoenvironmental Implications in the Extinct Insular Canid Cynotherium sardous Studiati, 1857 (Sardinia, Italy)". Animals. 12 (7): 833. doi:10.3390/ani12070833. ISSN 2076-2615. PMC 8996844. PMID 35405823.
- ^ Palombo, Maria Rita; Rozzi, Roberto (April 2014). "How correct is any chronological ordering of the Quaternary Sardinian mammalian assemblages?". Quaternary International. 328–329: 136–155. Bibcode:2014QuInt.328..136P. doi:10.1016/j.quaint.2013.09.046.
- ^ Valenzuela, Alejandro; Torres-Roig, Enric; Zoboli, Daniel; Pillola, Gian Luigi; Alcover, Josep Antoni (March 2022). "Asynchronous ecological upheavals on the Western Mediterranean islands: New insights on the extinction of their autochthonous small mammals". The Holocene. 32 (3): 137–146. Bibcode:2022Holoc..32..137V. doi:10.1177/09596836211060491. hdl:11584/322952. ISSN 0959-6836. S2CID 244763779.
External links
[edit]- ISPRA Photo of Cynotherium sardous











