Recent from talks
Nothing was collected or created yet.
Savior sibling
View on WikipediaThis article needs additional citations for verification. (October 2025) |
A savior baby, savior sibling, or donor baby is a child who is conceived in order to provide a stem cell, bone marrow, or other transplant to a sibling that is affected with a fatal disease, such as cancer or Fanconi anemia, that can best be treated by hematopoietic stem cell, or other transplant.
Introduction
[edit]The savior sibling is conceived through in vitro fertilization. Fertilized zygotes are tested for genetic compatibility (human leukocyte antigen (HLA) typing), using preimplantation genetic diagnosis (PGD), and only zygotes that are compatible with the existing child are implanted. Zygotes are also tested to make sure they are free of the original genetic disease. The procedure is controversial.[1][2][3]
Indications
[edit]A savior sibling may be the solution for any disease treated by hematopoietic stem cell transplantation. It is effective against genetically detectable (mostly monogenic) diseases, e.g. Fanconi anemia,[4] Diamond–Blackfan anemia[5] and β-thalassemia, in the ailing sibling, since the savior sibling can be selected to not have inherited the disease. The procedure may also be used in children with leukemia, and in such cases HLA match is the only requirement, and not exclusion of any other obvious genetic disorder.
Procedure
[edit]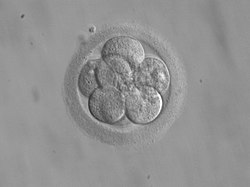
Multiple embryos are created and preimplantation genetic diagnosis is used to detect and select ones that are free of a genetic disorder and that are also a HLA match for an existing sibling who requires a transplant. Upon birth, umbilical cord blood is taken and used for hematopoietic stem cell transplantation.
Bioethical concerns
[edit]This section may require copy editing for grammar, style, cohesion, tone, or spelling. (October 2025) |
This section contains too many or overly lengthy quotations. (October 2025) |
The conception of a child in order to save another raises ethical issues, the sick eldest child being doomed, unless a compatible brother or sister can treat them.
This ethical debate is reflected in the terminology used, with opponents insisting on the term "baby‐medicine" to underline the "instrumentalization of the human body".[6] For these opponents, PGD is considered "utilitarianism taken to the extreme", "human procreation is totally diverted for the benefit of the project of creating a human being whose main "mission" is to be a medicine. Project carrying a radical alienation of one's freedom since its conception is only desired because of its therapeutic potential. They have no other choice than to take on the status of reservoir of cells for their sick elder, subject to a project predetermined by others, in this case society, the medical profession and their own parents."[citation needed]
The Jérome Lejeune Foundation, like other religious associations, which is anti-abortion, against research on embryos, but not against assisted procreation, speaks of a "double sorting baby", because this child is the survivor of a double sorting: the 1st in relation to the disease, the 2nd, in relation to compatibility,[7] thus wanting to criticize the selection of a large number of "healthy" embryos and therefore the "interruption of the life" of future babies, and therefore a eugenic drift. Finally, these same opponents refuse the term "double hope baby", because it means the same practice as the expression "baby-medicine" – therefore the same criticisms apply.[citation needed]
Specialists prefer to use "child of double hope": first hope of the birth of a healthy child, second hope of curing the eldest child, "the fact of being wanted for another does not exclude being wanted at the same time." "conceiving a child in the hope that it can cure someone is not in itself immoral, provided that it is not conceived exclusively for this purpose. We can reasonably hope that parents who love their child to the point of trying everything to save him will know how to love for himself the one by whom their first child was saved."[citation needed]
In 2002 the National Consultative Ethics Committee (CCNE) was reserved, recalling the "risk of instrumentalization of the unborn child" and that the "selection of an embryo and the initiation of a child conceived only as a potential donor, and not primarily for himself, is not thinkable in view of the values that he has always defended." However, he puts things into perspective by indicating that "allowing a desired child to represent, moreover, a hope of recovery for his eldest, is an acceptable objective, if he is second." The Council of State questions: "The use of "double IPR" has remained exceptional (7 requests since the end of 2009). The hope offered to certain families in the face of suffering from an illness without a therapeutic solution seems not to have been fully satisfied. Therefore, it is not certain that the weight that double PGD places on the child resulting from it has found sufficient medical justification."[8]
French law rejects the concept of "medicine baby" but authorizes that of "double hope baby".[9] In 2009, the CCNE recalled its 2002 opinion "allowing a desired child to represent hope of recovery for their eldest is an acceptable objective if he is second." As the current law provides, this extreme possibility should only be reserved for couples who have a child suffering from an illness leading to death. A savior baby or savior sibling is a child who is conceived in order to provide a stem cell transplant to a sibling that is affected with a fatal disease, such as cancer or Fanconi anemia, that can best be treated by hematopoietic stem cell transplantation.[citation needed]
Ethical critics argue that this practice instrumentalizes these children in two ways: first, by having them for instrumental reasons rather than for their own sake, and second, by valuing them solely as a means to cure their sick sibling, potentially violating ethical principles like beneficence and nonmaleficence. Supporters, on the other hand, contend that parents' motives should not be condemned, and the key question is how savior siblings are treated after birth. They argue that the Kantian principle prohibiting the use of individuals solely as means to an end does not apply if the savior sibling is also treated as an end in itself. The willingness of parents to go to great lengths to conceive such a child may indicate their dedication to the welfare of all their children.[10]
Legality
[edit]- In the United Kingdom, the House of Lords has ruled that it is lawful to use modern reproductive techniques to create a savior sibling.[11]
- In Victoria, Australia, use of PGD for HLA typing is reviewed by the Infertility Treatment Authority on a case-by-case basis.[1]
- In Spain, the Cortes Generales approved the Assisted Reproduction Law in 2006, which authorizes preimplantation diagnosis for therapeutic purposes.[citation needed]
- In France, the bioethics law of 7 July 2011 authorizes the practice of double PGD, going beyond the opinion of the Council of State and thus perpetuating a practice provisionally authorized by the bioethics law of August 2004, including the decree application published in the Official Journal of 23 December 2006.[12]
- In Switzerland, the law on medically assisted reproduction prohibits pre-implantation genetic diagnosis (PGD) and "savior siblings".[13]
History
[edit]Yury Verlinsky and collaborators described the first case in 2001:[14] that of Adam Nash, born 29 August 2000.[15]
Challenges and considerations
[edit]Concerns regarding savior siblings revolve around potential physical and psychological harm. Physically, there are inherent risks associated with the medical technologies involved in conceiving savior siblings, such as in vitro fertilization (IVF), preimplantation genetic diagnosis (PGD), and hematopoietic stem cell (HSC) donation. These risks appear to be relatively low, with IVF being a routine procedure and limited short-term harm observed with PGD and HLA typing. The risks associated with HSC transplantation depend on the source of HSC and collection method, with generally low risks.
However, there is a concern about the possibility of a savior sibling being repeatedly asked to donate HSC, which could potentially harm the child. To address this, legal safeguards can be put in place to limit the number of HSC donations by a child donor.
Psychologically, it has been suggested that savior siblings might experience harm if they discover they were conceived primarily to be an HSC donor for their sick sibling. However, there is insufficient empirical evidence to support this claim. On the contrary, it can be argued that savior siblings may experience psychological benefits from knowing they contributed to helping their ailing sibling, regardless of the transplantation's success. The balance between potential psychological benefits and medical risks remains a subject of debate.
Past studies on pediatric sibling donors of HSC transplantations indicate that the experience varies. In cases of unsuccessful transplantations, some siblings may experience negative emotions, particularly if the failure is linked to the sick sibling's death. Proper information, parental care, emotional support, and attention can help mitigate any potential negative psychological impacts on child donors, including savior siblings. Overall, addressing these concerns involves careful consideration of both the physical and psychological well-being of savior siblings during the decision-making process and throughout their lives.[16]
Popular culture
[edit]The novel My Sister's Keeper, later adapted into a film, is about a child who was born as a savior sibling to her sister Kate who is affected by acute promyelocytic leukemia.
In the British soap opera Emmerdale, Debbie Dingle gave birth to her son Jack, who would serve as a savior sibling to his older sister Sarah, who was suffering from Fanconi anemia.
On the popular American show CSI: Crime Scene Investigation, the episode "Harvest" deals with the reported abduction and later murder of a thirteen-year-old girl named Alycia who was later revealed to be a savior sibling for her brother Daniel.
In the American show Heroes, one of the protagonists, Mohinder Suresh, is revealed to have been conceived to cure his sister Shanti of a deadly disease known as the Shanti Virus, although he was ultimately born too late to save her life. His antibodies act as a cure for other patients with the disease throughout the show as well.
On Star Trek: Enterprise, the episode "Similitude" sees a clone created of Trip Tucker for the purpose of organ harvesting. "Sim" is born through the highly controversial use of an alien lifeform.
The novel Never Let Me Go, later adapted into a film, is centred around a dystopian future society where human clones are created and allowed to live to their teenage years before being used for organ harvesting. The films Parts: The Clonus Horror and The Island have a similar premise.
In the Grey's Anatomy episode "I Bet It Stung," the character Donna is a savior sibling to her older sister Reese.
In the 9-1-1 episode "Buck Begins", it is revealed that Evan "Buck" Buckley was conceived as a savior sibling for his older brother Daniel.
In the Korean drama The Penthouse, the character Anna is adopted by a Korean-American family to be a savior sibling to their son, Logan Lee, who was suffering from bone marrow cancer at the time.
In the light novels of Sword Art Online and their respective anime adaptations, the character Vassago Casals was born from an affair his Japanese father had with a Spanish woman he paid for sexual services. Although Vassago's mother desired to perform an abortion, his father convinced her not to go through with it, as his older son from his actual wife had a congenital kidney defect, hoping that the child could serve as a kidney donor.
See also
[edit]References
[edit]- ^ a b Liu, Crystal K. (2007). "'Saviour Siblings'? The Distinction between PGD with HLA Tissue Typing and Preimplantation HLA Tissue Typing". Journal of Bioethical Inquiry. 4: 65–70. doi:10.1007/s11673-007-9034-9. S2CID 54515037.
- ^ Sui, Suli; Sleeboom-Faulkner, Margaret (2010). "Choosing offspring: Prenatal genetic testing for thalassaemia and the production of a 'saviour sibling' in China". Culture, Health & Sexuality. 12 (2): 167–75. doi:10.1080/13691050902914110. PMID 19499399. S2CID 24115814.
- ^ Bennett, B (2005). "Symbiotic relationships: Saviour siblings, family rights and biomedicine". Australian Journal of Family Law. 19 (3): 195–212. PMID 17058348.
- ^ Moore, Pete (2007). The Debate About Genetic Engineering (Ethical Debates). New York, NY: Rosen Central. p. 29. ISBN 978-1-4042-3754-4.
- ^ "Saviour sibling". worldwidewords.org. 11 August 2007.
- ^ ""L'enfant du double espoir" n'est pas un "bébé médicament"". Le Monde.fr (in French). 15 February 2011. Retrieved 5 October 2023.
- ^ "Fondation Jérôme Lejeune - Déficience intellectuelle d'origine génétique". Fondation Jérôme Lejeune (in French). Retrieved 5 October 2023.
- ^ d'État, Le Conseil (5 October 2023). "Accueil - Conseil d'Etat". Conseil d'État (in French). Retrieved 5 October 2023.
- ^ Fagniez, P. -L.; Loriau, J.; Tayar, C. (1 October 2005). "Du " bébé médicament " au " bébé du double espoir "". Gynécologie Obstétrique & Fertilité. 33 (10): 828–832. doi:10.1016/j.gyobfe.2005.07.034. ISSN 1297-9589. PMID 16139550.
- ^ Kuek, Chee Ying; Singh, Sharon Kaur a/p Gurmukh; Tay, Pek San (June 2021). "Conception of Saviour Siblings: Ethical Perceptions of Selected Stakeholders in Malaysia". Asian Bioethics Review. 13 (2): 167–178. doi:10.1007/s41649-021-00166-2. PMC 8079567. PMID 33968213.
- ^ [2005] UKHL 28>
- ^ "Décret n° 2006-1660 du 22 décembre 2006 relatif au don de gamètes et à l'assistance médicale à la procréation et modifiant le code de la santé publique (Dispositions réglementaires)".
- ^ Jacolet, Thierry (16 February 2011). ""Bébé-médicament" : une procédure impossible en Suisse". Rev Med Suisse (in Swiss French). 282 (6): 423.
- ^ "Preimplantation Diagnosis for Fanconi Anemia Combined With HLA Matching", Yury Verlinsky et al. The Journal of the American Medical Association.
- ^ "What Are Savior Siblings & How Are They Created? - Cases & Ethics". inviTRA. 25 July 2018. Retrieved 18 April 2023.
- ^ Kuek, Chee Ying; Gurmukh Singh, Sharon Kaur a/p; Tay, Pek San (17 March 2021). "Conception of Saviour Siblings: Ethical Perceptions of Selected Stakeholders in Malaysia". Asian Bioethics Review. 13 (2): 167–178. doi:10.1007/s41649-021-00166-2. ISSN 1793-8759. PMC 8079567. PMID 33968213.
External links
[edit]- "Made to Order Savior" New York Times Magazine
- "Select a Baby’s Health, Not Eye Color" Los Angeles Times Op-ed
