Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
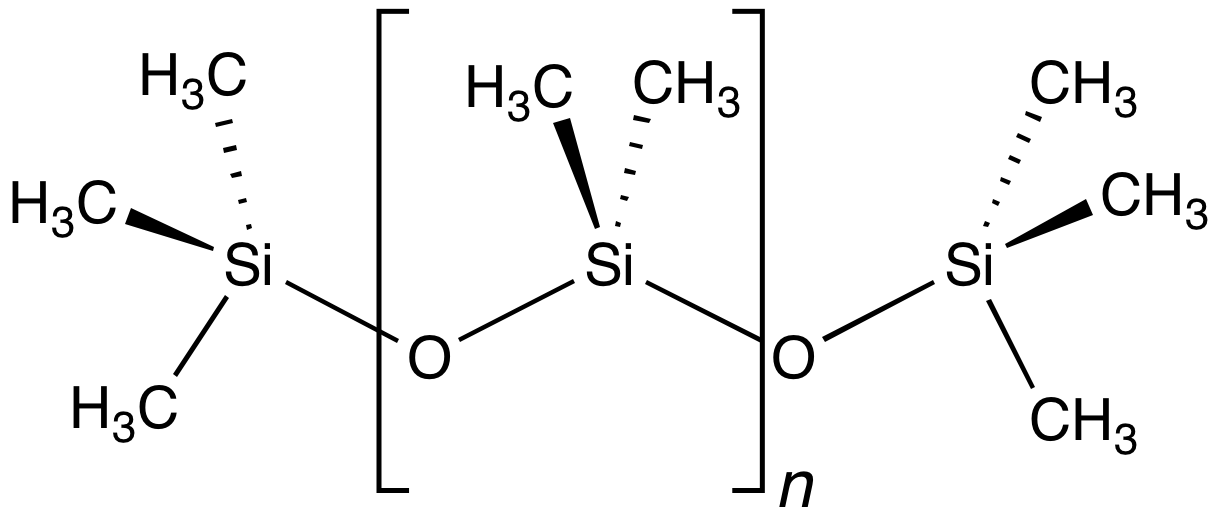
Siloxane
In organosilicon chemistry, a siloxane is an organic compound containing a functional group of two silicon atoms bound to an oxygen atom: Si−O−Si. The parent siloxanes include the oligomeric and polymeric hydrides with the formulae H[OSiH2]nOH and [OSiH2]n. Siloxanes also include branched compounds, the defining feature of which is that each pair of silicon centres is separated by one oxygen atom. The siloxane functional group forms the backbone of silicones [−R2Si−O−SiR2−]n, the premier example of which is polydimethylsiloxane (PDMS). The functional group R3SiO− (where the three Rs may be different) is called siloxy. Siloxanes are manmade and have many commercial and industrial applications because of the compounds’ hydrophobicity, low thermal conductivity, and high flexibility.
Siloxanes generally adopt structures expected for linked tetrahedral ("sp3-like") centers. The Si−O bond length is 1.64 Å (vs Si–C distance of 1.92 Å) and the Si-O-Si angle is rather open at 142.5°. By contrast, the C−O distance in a typical dialkyl ether is much shorter at 1.414(2) Å with a more acute C−O−C angle of 111°. It can be appreciated that the siloxanes would have low barriers for rotation about the Si−O bonds as a consequence of low steric hindrance. This geometric consideration is the basis of the useful properties of some siloxane-containing materials, such as their low glass transition temperatures.
The main route to siloxane functional group is by hydrolysis of silicon chlorides:
The reaction proceeds via the initial formation of silanols (R3Si−OH):
The siloxane bond can then form via a silanol + silanol pathway or a silanol + chlorosilane pathway:
Hydrolysis of a silyldichloride can afford linear or cyclic products. Linear products are terminated with silanol groups:
Cyclic products have no silanol termini:
The linear products, polydimethylsiloxane (PDMS), are of great commercial value. Their production requires the production of dimethylsilicon dichloride.
Hub AI
Siloxane AI simulator
(@Siloxane_simulator)
Siloxane
In organosilicon chemistry, a siloxane is an organic compound containing a functional group of two silicon atoms bound to an oxygen atom: Si−O−Si. The parent siloxanes include the oligomeric and polymeric hydrides with the formulae H[OSiH2]nOH and [OSiH2]n. Siloxanes also include branched compounds, the defining feature of which is that each pair of silicon centres is separated by one oxygen atom. The siloxane functional group forms the backbone of silicones [−R2Si−O−SiR2−]n, the premier example of which is polydimethylsiloxane (PDMS). The functional group R3SiO− (where the three Rs may be different) is called siloxy. Siloxanes are manmade and have many commercial and industrial applications because of the compounds’ hydrophobicity, low thermal conductivity, and high flexibility.
Siloxanes generally adopt structures expected for linked tetrahedral ("sp3-like") centers. The Si−O bond length is 1.64 Å (vs Si–C distance of 1.92 Å) and the Si-O-Si angle is rather open at 142.5°. By contrast, the C−O distance in a typical dialkyl ether is much shorter at 1.414(2) Å with a more acute C−O−C angle of 111°. It can be appreciated that the siloxanes would have low barriers for rotation about the Si−O bonds as a consequence of low steric hindrance. This geometric consideration is the basis of the useful properties of some siloxane-containing materials, such as their low glass transition temperatures.
The main route to siloxane functional group is by hydrolysis of silicon chlorides:
The reaction proceeds via the initial formation of silanols (R3Si−OH):
The siloxane bond can then form via a silanol + silanol pathway or a silanol + chlorosilane pathway:
Hydrolysis of a silyldichloride can afford linear or cyclic products. Linear products are terminated with silanol groups:
Cyclic products have no silanol termini:
The linear products, polydimethylsiloxane (PDMS), are of great commercial value. Their production requires the production of dimethylsilicon dichloride.