Recent from talks
Nothing was collected or created yet.
Sympathetic trunk
View on Wikipedia| Sympathetic trunk | |
|---|---|
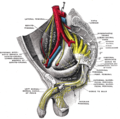
 Abdominal portion of the sympathetic trunk, with the celiac plexus and hypogastric plexus. (Sympathetic trunk labeled at center left.) | |
 Scheme showing pathways (white/grey rami are spatially reversed, possibly for clarity?) of a typical spinal nerve. 1. Somatic efferent. 2. Somatic afferent. 3,4,5. Sympathetic efferent. 6,7. Autonomic afferent. | |
| Details | |
| Identifiers | |
| Latin | truncus sympathicus |
| TA98 | A14.3.01.002 |
| TA2 | 6602 |
| FMA | 6258 |
| Anatomical terminology | |
The sympathetic trunk (sympathetic chain, gangliated cord) is a paired bundle of nerve fibers that run from the base of the skull to the coccyx. It is a major component of the sympathetic nervous system.
Structure
[edit]The sympathetic trunk lies just lateral to the vertebral bodies for the entire length of the vertebral column. It interacts with the anterior rami of spinal nerves by way of rami communicantes. The sympathetic trunk permits preganglionic fibers of the sympathetic nervous system to ascend to spinal levels superior to T1 and descend to spinal levels inferior to L2/3.[1][2][3][4][5][6][7]
The superior end of it is continued upward through the carotid canal into the skull, and forms a plexus on the internal carotid artery; the inferior part travels in front of the coccyx, where it converges with the other trunk at a structure known as the ganglion impar.
Along the length of the sympathetic trunk are sympathetic ganglia known as paravertebral ganglia.
-
The sympathetic trunk (yellow) can be seen just lateral to the vertebral column.
Relations
[edit]In the cervical region, the sympathetic trunk is situated upon the prevertebral fascia posterior to the carotid sheath.[8]: 600
Function
[edit]The sympathetic trunk is a fundamental part of the sympathetic nervous system, and part of the autonomic nervous system. It allows nerve fibres to travel to spinal nerves that are superior and inferior to the one in which they originated. Also, a number of nerves, such as most of the splanchnic nerves, arise directly from the trunks.
| Organ | Nerves[9] | Spinal column origin[9] |
|---|---|---|
| stomach | T5, T6, T7, T8, T9, sometimes T10 | |
| duodenum | T5, T6, T7, T8, T9, sometimes T10 | |
| jejunum and ileum | T5, T6, T7, T8, T9 | |
| spleen | T6, T7, T8 | |
| gallbladder and liver |
|
T6, T7, T8, T9 |
| colon |
| |
| pancreatic head | T8, T9 | |
| appendix |
|
T10 |
| bladder |
|
S2-S4 |
| kidneys and ureters |
|
T11, T12 |
Additional images
[edit]-
The formation of the spinal nerve from the dorsal and ventral roots.
-
Dissection of side wall of pelvis showing sacral and pudendal plexuses.
-
Sacral plexus of the right side.
-
Diagram of efferent sympathetic nervous system.
-
Sympathetic connections of the ciliary and superior cervical ganglia.
-
Sacral sympathetic
See also
[edit]References
[edit]![]() This article incorporates text in the public domain from page 976 of the 20th edition of Gray's Anatomy (1918)
This article incorporates text in the public domain from page 976 of the 20th edition of Gray's Anatomy (1918)
- ^ Mader S. S. (2000): Human biology. McGraw-Hill, New York, ISBN 0-07-290584-0; ISBN 0-07-117940-2.
- ^ Pritchard T. E., Alloway D. (1999): Medical neuroscience. Hayes Barton Press, ISBN 978-1-59377-200-0:https://books.google.com/books/about/Medical_neuroscience.html?id=m7Y80PcFHtsC.
- ^ Butler A. B., Hodos W. (2005): Comparative vertebrate neuroanatomy: evolution and adaptation. Wiley-Blackwell, ISBN 978-0-471-21005-4.
- ^ Butler, Ann B.; Hodos, William (2005-09-02). Comparative Vertebrate Neuroanatomy: Evolution and Adaptation. Wiley. ISBN 9780471733836.
- ^ Hall J. E., Guyton A. C. (2006): Textbook of medical physiology, 11th edition. Elsevier Saunders, St. Louis, Mo, ISBN 0-7216-0240-1.
- ^ Warrell D. A., Cox T. M., Firth J. D. (2010): The Oxford Textbook of Medicine Archived 2012-03-21 at the Wayback Machine (5th ed.). Oxford University Press
- ^ Greenstein B., Greenstein A. (2002): Color atlas of neuroscience – Neuroanatomy and neurophysiology. Thieme, Stuttgart – New York, ISBN 9783131081711.
- ^ Standring, Susan (2020). Gray's Anatomy: The Anatomical Basis of Clinical Practice (42nd ed.). [New York]. ISBN 978-0-7020-7707-4. OCLC 1201341621.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ a b Unless specified otherwise in the boxes, the source is: Moore, Keith L.; Agur, A. M. R. (2002). Essential Clinical Anatomy (2nd ed.). Lippincott Williams & Wilkins. p. 199. ISBN 978-0-7817-5940-3.
External links
[edit]- Anatomy figure: 21:04-04 at Human Anatomy Online, SUNY Downstate Medical Center - "The position of the right and left vagus nerves, and sympathetic trunks in the mediastinum."
- Anatomy photo:43:15-0102 at the SUNY Downstate Medical Center - "The Female Pelvis: The Posterolateral Pelvic Wall"
- Atlas image: n3a6p1 at the University of Michigan Health System - "Autonomic Connections of the Spinal Cord"
- Diagram at umm.edu