Recent from talks
Nothing was collected or created yet.
Hysterectomy
View on Wikipedia| Hysterectomy | |
|---|---|
 Diagram showing what is removed with a radical hysterectomy | |
| Specialty | Gynaecology |
| ICD-9-CM | 68.9 |
| MeSH | D007044 |
| MedlinePlus | 002915 |
Hysterectomy is the surgical removal of the uterus and cervix. Supracervical hysterectomy refers to the removal of the uterus while the cervix is spared. These procedures may also involve removal of the ovaries (oophorectomy), fallopian tubes (salpingectomy), and other surrounding structures. The terms "partial" or "total" hysterectomy are lay terms that incorrectly describe the addition or omission of oophorectomy at the time of hysterectomy. These procedures are usually performed by a gynecologist. Removal of the uterus is a form of sterilization, rendering the patient unable to bear children (as does removal of ovaries and fallopian tubes) and has surgical risks as well as long-term effects, so the surgery is normally recommended only when other treatment options are not available or have failed. It is the second most commonly performed gynecological surgical procedure, after cesarean section, in the United States.[1] Nearly 68 percent were performed for conditions such as endometriosis, irregular bleeding, and uterine fibroids.[1] It is expected that the frequency of hysterectomies for non-malignant indications will continue to fall, given the development of alternative treatment options.[2]
Medical uses
[edit]
Hysterectomy is a major surgical procedure that has risks and benefits. It affects the hormonal balance and overall health of patients. Because of this, hysterectomy is normally recommended as a last resort after pharmaceutical or other surgical options have been exhausted to remedy certain intractable and severe uterine/reproductive system conditions. There may be other reasons for a hysterectomy to be requested. Such conditions and/or indications include, but are not limited to:[3]
- Endometriosis: growth of the uterine lining outside the uterine cavity. This inappropriate tissue growth can lead to pain and bleeding.[4]
- Adenomyosis: a form of endometriosis, where the uterine lining has grown into and sometimes through the uterine wall musculature. This can thicken the uterine walls and also contribute to pain and bleeding.[5]
- Heavy menstrual bleeding: irregular or excessive menstrual bleeding for greater than a week. It can disturb the regular quality of life and may be indicative of a more serious condition.
- Uterine fibroids: benign growths on the uterus wall. These muscular noncancerous tumors can grow in single form or in clusters and can cause extreme pain and bleeding.[6]
- Uterine prolapse: when the uterus sags down due to weakened or stretched pelvic floor muscles potentially causing the uterus to protrude out of the vagina in more severe cases.
- Reproductive system cancer prevention: especially if there is a strong family history of reproductive system cancers (especially breast cancer in conjunction with BRCA1 or BRCA2 mutation), or as part of recovery from such cancers.[7]
- Gynecologic cancer: depending on the type of hysterectomy, can aid in the treatment of cancer or precancer of the endometrium, cervix, or uterus. To protect against or treat cancer of the ovaries, one would need an oophorectomy.
- Transgender (trans) male affirmation: aids in gender dysphoria, prevention of future gynecologic problems, and transition to obtaining new legal gender documentation.[8]
- Severe developmental disabilities: this treatment is controversial at best. In the United States, specific cases of sterilization due to developmental disabilities have been found by state-level Supreme Courts to violate the patient's constitutional and common-law rights.[9]
- Postpartum: to remove either a severe case of placenta praevia (a placenta that has either formed over or inside the birth canal) or placenta percreta (a placenta that has grown into and through the wall of the uterus to attach itself to other organs), as well as a last resort in case of excessive obstetrical haemorrhage.[10]
- Chronic pelvic pain: should try to obtain the pain etiology, although it may have no known cause.[11]
- PMS and menstrual pain and other psychic and physical conditions caused by the menstrual period and causing suffering and diminishing life quality.
Risks and adverse effects
[edit]In 1995, the short-term mortality (within 40 days of surgery) was reported at 0.38 cases per 1000 when performed for benign causes. Risks for surgical complications were the presence of fibroids, younger age (vascular pelvis with higher bleeding risk and larger uterus), dysfunctional uterine bleeding and parity.[12]
The mortality rate is several times higher when performed in patients who are pregnant, have cancer or other complications.[13]
The long-term effect on all case mortality is relatively small. Women under the age of 45 years have a significantly increased long-term mortality that is believed to be caused by the hormonal side effects of hysterectomy and prophylactic oophorectomy.[14][15] This effect is not limited to pre-menopausal women; even women who have already entered menopause were shown to have experienced a decrease in long-term survivability post-oophorectomy.[16]
Approximately 35% of women after hysterectomy undergo another related surgery within 2 years.[17]
Ureteral injury is not uncommon and occurs in 0.2 per 1,000 cases of vaginal hysterectomy and 1.3 per 1,000 cases of abdominal hysterectomy.[18] The injury usually occurs in the distal ureter close to the infundibulopelvic ligament or as a ureter crosses below the uterine artery, often from blind clamping and ligature placement to control hemorrhage.[19]
Recovery
[edit]Hospital stay is 3 to 5 days or more for the abdominal procedure and between 1 and 2 days (but possibly longer) for vaginal or laparoscopically assisted vaginal procedures.[20] After the procedure, the American College of Obstetricians and Gynecologists recommends not inserting anything into the vagina for the first 6 weeks (including inserting tampons or having sex).[21]
Unintended oophorectomy and premature ovarian failure
[edit]Removal of one or both ovaries is performed in a substantial number of hysterectomies that were intended to be ovary sparing.[22]
The average onset age of menopause after hysterectomy with ovarian conservation is 3.7 years earlier than average.[23] This has been suggested to be due to the disruption of blood supply to the ovaries after a hysterectomy or due to missing endocrine feedback of the uterus. The function of the remaining ovaries is significantly affected in about 40% of people, and some of them even require hormone replacement therapy. Surprisingly, a similar and only slightly weaker effect has been observed for endometrial ablation which is often considered as an alternative to hysterectomy.[24]
A substantial number of women develop benign ovarian cysts after a hysterectomy.[25]
Effects on sexual life and pelvic pain
[edit]After hysterectomy for benign indications, the majority of patients report improvement in sexual life and pelvic pain. A smaller share of patients report worsening of their sexual life and other problems. The picture is significantly different for hysterectomy performed for malignant reasons; the procedure is often more radical with substantial side effects.[26][27] A proportion of patients who undergo a hysterectomy for chronic pelvic pain continue to have pelvic pain after a hysterectomy and develop dyspareunia (painful sexual intercourse).[28]
Premature menopause and its effects
[edit]Hysterectomies that also include the surgical removal of the ovaries (i.e. an oophorectomy) result in significant hormonal changes to the body. Estrogen levels fall sharply when the ovaries are removed, removing the protective effects of estrogen on the cardiovascular and skeletal systems. This condition is often referred to as "surgical menopause", although it is substantially different from a naturally occurring menopausal state; the former is a sudden hormonal shock to the body that causes rapid onset of menopausal symptoms such as hot flashes, while the latter is a gradually occurring decrease of hormonal levels over a period of years with uterus intact and ovaries able to produce hormones even after the cessation of menstrual periods.[29]
One study showed that the risk of subsequent cardiovascular disease is substantially increased for women who had a hysterectomy at age 50 or younger. No association was found for women undergoing the procedure after age 50. The risk is higher when ovaries are removed, but still noticeable even when ovaries are preserved.[30]
Several other studies have found that osteoporosis (decrease in bone density) and increased risk of bone fractures are associated with hysterectomies.[31][32] This has been attributed to the modulatory effect of estrogen on calcium metabolism and the drop in serum estrogen levels after menopause can cause excessive loss of calcium leading to bone wasting.
Hysterectomies have also been linked with higher rates of heart disease and weakened bones. Those who have undergone a hysterectomy with both ovaries removed typically have reduced testosterone levels as compared to those left intact.[22] Reduced levels of testosterone in women are predictive of height loss, which may occur as a result of reduced bone density,[33] while increased testosterone levels in women are associated with a greater sense of sexual desire.[34]
Oophorectomy before the age of 45 is associated with a fivefold mortality from neurologic and mental disorders.[35]
Urinary incontinence and vaginal prolapse
[edit]Urinary incontinence and vaginal prolapse are well known adverse effects that develop with high frequency a very long time after the surgery. Typically, those complications develop 10–20 years after the surgery.[36] For this reason exact numbers are not known, and risk factors are poorly understood. It is also unknown if the choice of surgical technique has any effect. It has been assessed that the risk for urinary incontinence is approximately doubled within 20 years after hysterectomy. One long-term study found a 2.4-fold increased risk for surgery to correct urinary stress incontinence following hysterectomy.[37][38]
The risk for vaginal prolapse depends on factors such as number of vaginal deliveries, the difficulty of those deliveries, and the type of labor.[39] Overall incidence is approximately doubled after hysterectomy.[40]
Adhesion formation and bowel obstruction
[edit]The formation of postoperative adhesions is a particular risk after hysterectomy because of the extent of dissection involved as well as the fact the hysterectomy wound is in the most gravity-dependent part of the pelvis into which a loop of bowel may easily fall.[41] In one review, incidence of small bowel obstruction due to intestinal adhesion was found to be 15.6% in non-laparoscopic total abdominal hysterectomies vs. 0.0% in laparoscopic hysterectomies.[42]
Wound infection
[edit]Wound infection occurs in approximately 3% of cases of abdominal hysterectomy. The risk is increased by obesity, diabetes, immunodeficiency disorder, use of systemic corticosteroids, smoking, wound hematoma, and preexisting infection such as chorioamnionitis and pelvic inflammatory disease.[43] Such wound infections mainly take the form of either incisional abscess or wound cellulitis. Typically, both confer erythema, but only an incisional abscess confers purulent drainage. The recommended treatment of an incisional abscess after hysterectomy is by incision and drainage, and then coverage by a thin layer of gauze followed by sterile dressing. The dressing should be changed and the wound irrigated with normal saline at least twice each day. In addition, it is recommended to administer an antibiotic active against staphylococci and streptococci, preferably vancomycin, when there is a risk of MRSA.[43] The wound can be allowed to close by secondary intention. Alternatively, if the infection is cleared and healthy granulation tissue is evident at the base of the wound, the edges of the incision may be reapproximated, such as by using butterfly stitches, staples or sutures.[43] Sexual intercourse remains possible after hysterectomy. Reconstructive surgery remains an option for women who have experienced benign and malignant conditions.[44] : 1020–1348
Other rare problems
[edit]Hysterectomy may cause an increased risk of the relatively rare renal cell carcinoma. The increased risk is particularly pronounced for young women; the risk was lower after vaginally performed hysterectomies.[45] Hormonal effects or injury of the ureter were considered as possible explanations.[46][47] In some cases, the renal cell carcinoma may be a manifestation of an undiagnosed hereditary leiomyomatosis and renal cell cancer syndrome.
Removal of the uterus without removing the ovaries can produce a situation that, on rare occasions, can result in ectopic pregnancy due to an undetected fertilization that had yet to descend into the uterus before surgery. Two cases have been identified and profiled in an issue of the Blackwell Journal of Obstetrics and Gynecology; over 20 other cases have been discussed in additional medical literature.[48] On very rare occasions, sexual intercourse after hysterectomy may cause a transvaginal evisceration of the small bowel.[49] The vaginal cuff is the uppermost region of the vagina that has been sutured closed. A rare complication, it can dehisce and allow the evisceration of the small bowel into the vagina.[50]
Alternatives
[edit]

Depending on the indication, there are alternatives to hysterectomy:
Heavy bleeding
[edit]Levonorgestrel intrauterine devices are highly effective at controlling dysfunctional uterine bleeding (DUB) or menorrhagia and should be considered before any surgery.[51]
Menorrhagia (heavy or abnormal menstrual bleeding) may also be treated with the less invasive endometrial ablation, which is an outpatient procedure in which the lining of the uterus is destroyed with heat, mechanically, or by radio frequency ablation.[52] Endometrial ablation greatly reduces or eliminates monthly bleeding in ninety percent of patients with DUB. It is not effective for patients with very thick uterine lining or uterine fibroids.[53]
Uterine fibroids
[edit]Levonorgestrel intrauterine devices are highly effective in limiting menstrual blood flow and improving other symptoms. Side effects are typically very moderate because the levonorgestrel (a progestin) is released in low concentration locally. There is now substantial evidence that Levonorgestrel-IUDs provide good symptomatic relief for women with fibroids.[54]
Uterine fibroids may be removed and the uterus reconstructed in a procedure called "myomectomy". A myomectomy may be performed through an open incision, laparoscopically, or through the vagina (hysteroscopy).[55]
Uterine artery embolization (UAE) is a minimally invasive procedure for treatment of uterine fibroids. Under local anesthesia, a catheter is introduced into the femoral artery at the groin and advanced under radiographic control into the uterine artery. A mass of microspheres or polyvinyl alcohol (PVA) material (an embolus) is injected into the uterine arteries to block the flow of blood through those vessels.[56] The restriction in blood supply usually results in a significant reduction of fibroids and improvement of heavy bleeding tendency. The 2012 Cochrane review comparing hysterectomy and UAE did not find any major advantage for either procedure. While the UAE procedure is associated with shorter hospital stay and a more rapid return to normal daily activities, it was also associated with a higher risk for minor complications later on. There were no differences between UAE and hysterectomy with regards to major complications.[57]
Uterine fibroids can be removed with a non-invasive procedure called Magnetic Resonance guided Focused Ultrasound (MRgFUS).
Uterine prolapse
[edit]Prolapse may also be corrected surgically without removal of the uterus.[58] There are several strategies that can be utilized to help strengthen pelvic floor muscles and prevent the worsening of prolapse. These include, but are not limited to, use of "kegel exercises", vaginal pessary, constipation relief, weight management, and care when lifting heavy objects.[59]
Types
[edit]
Hysterectomy, in the literal sense of the word, means merely the removal of the uterus. However, other organs such as the ovaries, fallopian tubes, and the cervix are very frequently removed as part of the surgery.[60]
- Radical hysterectomy: complete removal of the uterus, cervix, upper vagina, and parametrium. Indicated for cancer. Lymph nodes, ovaries, and fallopian tubes are also usually removed in this situation, such as in Wertheim's hysterectomy.[61]
- Total hysterectomy: complete removal of the uterus and cervix, with or without oophorectomy.
- Subtotal hysterectomy: removal of the uterus, leaving the cervix in situ.
Subtotal (supracervical) hysterectomy was originally proposed with the expectation that it may improve sexual functioning after hysterectomy, it has been postulated that removing the cervix causes excessive neurologic and anatomic disruption, thus leading to vaginal shortening, vaginal vault prolapse, and vaginal cuff granulations.[62] These theoretical advantages were not confirmed in practice, but other advantages over total hysterectomy emerged. The principal disadvantage is that the risk of cervical cancer is not eliminated, and women may continue cyclical bleeding (although substantially less than before the surgery). These issues were addressed in a systematic review of total versus supracervical hysterectomy for benign gynecological conditions, which reported the following findings:[63]
- There was no difference in the rates of incontinence, constipation, measures of sexual function, or alleviation of pre-surgery symptoms.
- Length of surgery and amount of blood lost during surgery were significantly reduced during supracervical hysterectomy compared to total hysterectomy, but there was no difference in post-operative transfusion rates.[64]
- Febrile morbidity was less likely and ongoing cyclic vaginal bleeding one year after surgery was more likely after supracervical hysterectomy.
- There was no difference in the rates of other complications, recovery from surgery, or readmission rates.
In the short term, randomized trials have shown that cervical preservation or removal does not affect the rate of subsequent pelvic organ prolapse.[65]
Supracervical hysterectomy does not eliminate the possibility of having cervical cancer since the cervix itself is left intact and may be contraindicated in women with increased risk of this cancer; regular pap smears to check for cervical dysplasia or cancer are still needed.[66][67]
Technique
[edit]Hysterectomy can be performed in different ways. The oldest known technique is vaginal hysterectomy. The first planned hysterectomy was performed by Konrad Johann Martin Langenbeck - Surgeon General of the Hannovarian army, although there are records of vaginal hysterectomy for prolapse going back as far as 50BC.[68]
The first abdominal hysterectomy recorded was by Ephraim McDowell. He performed the procedure in 1809 for a mother of five with a large ovarian mass on her kitchen table.[69]
In modern medicine today, laparoscopic vaginal (with additional instruments passing through ports in small abdominal incisions, close or in the navel) and total laparoscopic techniques have been developed.
Abdominal hysterectomy
[edit]Most hysterectomies in the United States are done via laparotomy (abdominal incision, not to be confused with laparoscopy). A transverse (Pfannenstiel) incision is made through the abdominal wall, usually above the pubic bone, as close to the upper hair line of the individual's lower pelvis as possible, similar to the incision made for a caesarean section. This technique allows physicians the greatest access to the reproductive structures and is normally done for the removal of the entire reproductive complex.[70] The recovery time for an open hysterectomy is 4–6 weeks and sometimes longer due to the need to cut through the abdominal wall. Historically, the biggest problem with this technique was infections, but infection rates are well-controlled and not a major concern in modern medical practice. An open hysterectomy provides the most effective way to explore the abdominal cavity and perform complicated surgeries. Before the refinement of the vaginal and laparoscopic vaginal techniques, it was also the only possibility to achieve subtotal hysterectomy; meanwhile, the vaginal route is the preferable technique in most circumstances.[71][72]
Vaginal hysterectomy
[edit]Vaginal hysterectomy is performed entirely through the vaginal canal and has clear advantages over abdominal surgery such as fewer complications, shorter hospital stays and shorter healing time.[73][74] Abdominal hysterectomy, the most common method, is used in cases such as after caesarean delivery, when the indication is cancer, when complications are expected, or when surgical exploration is required.
Laparoscopic-assisted vaginal hysterectomy
[edit]With the development of laparoscopic techniques in the 1970s and 1980s, the "laparoscopic-assisted vaginal hysterectomy" (LAVH) has gained great popularity among gynecologists because compared with the abdominal procedure it is less invasive and the post-operative recovery is much faster. It also allows better exploration and slightly more complicated surgeries than the vaginal procedure. LAVH begins with laparoscopy and is completed such that the final removal of the uterus (with or without removing the ovaries) is via the vaginal canal. Thus, LAVH is also a total hysterectomy; the cervix is removed with the uterus.[75] If the cervix is removed along with the uterus, the upper portion of the vagina is sutured together and called the vaginal cuff.[76]
Laparoscopic-assisted supracervical hysterectomy
[edit]The "laparoscopic-assisted supracervical hysterectomy" (LASH) was later developed to remove the uterus without removing the cervix using a morcellator which cuts the uterus into small pieces that can be removed from the abdominal cavity via the laparoscopic ports.[77]
Total laparoscopic hysterectomy
[edit]Total laparoscopic hysterectomy (TLH) was developed in the early 90s by Prabhat K. Ahluwalia in Upstate New York.[78] TLH is performed solely through the laparoscopes in the abdomen, starting at the top of the uterus, typically with a uterine manipulator. The entire uterus is disconnected from its attachments using long, thin instruments through the "ports". Then, all the tissue to be removed is passed through the small abdominal incisions.
Other techniques
[edit]Supracervical (subtotal) laparoscopic hysterectomy (LSH) is performed similarly to total laparoscopic surgery, but the uterus is amputated between the cervix and fundus.[79]
Dual-port laparoscopy is a form of laparoscopic surgery using two 5 mm midline incisions: the uterus is detached through the two ports and removed through the vagina.[80][81]
"Robotic hysterectomy" is a variant of laparoscopic surgery using special, remotely controlled instruments that allow the surgeon finer control as well as three-dimensional magnified vision.[82]
-
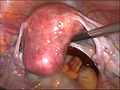
Uterus prior to hysterectomy
-
Laparoscopical hysterectomy
-
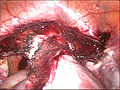
Cervical stump (white) after removal of the uterine corpus at laparoscopic supracervical hysterectomy
-
Transvaginal extraction of the uterus in total laparoscopical hysterectomy
-
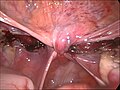
End of a laparoscopical hysterectomy
Comparison of techniques
[edit]Patient characteristics such as the reason for needing a hysterectomy, uterine size, descent of the uterus, presence of diseased tissues surrounding the uterus, previous surgery in the pelvic region, obesity, history of pregnancy, the possibility of endometriosis, or the need for an oophorectomy, will influence a surgeon's surgical approach when performing a hysterectomy.[83][needs update]
Vaginal hysterectomy is recommended over other variants where possible for women with benign diseases.[71][72][83] Vaginal hysterectomy was shown to be superior to LAVH and some types of laparoscopic surgery causing fewer short- and long-term complications, more favorable effect on sexual experience with shorter recovery times and fewer costs.[84][85][86]
Laparoscopic surgery offers certain advantages when vaginal surgery is not possible but also has the disadvantage of significantly longer time required for the surgery.[83][73]
In one 2004 study conducted in the UK comparing abdominal (laparotomic) and laparoscopic techniques, laparoscopic surgery was found to cause longer operation time and a higher rate of major complications while offering much quicker healing.[87] In another study conducted in 2014, laparoscopy was found to be "a safe alternative to laparotomy" in patients receiving total hysterectomy for endometrial cancer. Researchers concluded the procedure "offers markedly improved perioperative outcomes with a lower reoperation rate and fewer postoperative complications when the standard of care shifts from open surgery to laparoscopy in a university hospital".[88]
The abdominal technique is very often applied in difficult circumstances or when complications are expected. Given these circumstances, the complication rate and time required for surgery compares very favorably with other techniques; however time required for healing is much longer.[83]
Hysterectomy by abdominal laparotomy is correlated with a much higher incidence of intestinal adhesions than other techniques.[42]
Time required for completion of surgery in the eVAL trial is reported as follows:[87]
- abdominal 55.2 minutes average, range 19–155
- vaginal 46.6 minutes average, range 14–168
- laparoscopic (all variants) 82.5 minutes average, range 10–325 (combined data from both trial arms)
Morcellation has been widely used especially in laparoscopic techniques and sometimes for the vaginal technique, but now appears to be associated with a considerable risk of spreading benign or malignant tumors.[89][90] In April 2014, the FDA issued a memo alerting medical practitioners to the risks of power morcellation.[91]
Robotic-assisted surgery is presently used in several countries for hysterectomies. Additional research is required to determine the benefits and risks involved, compared to conventional laparoscopic surgery.[92]
A 2014 Cochrane review found that robotic-assisted surgery may have a similar complication rate when compared to conventional laparoscopic surgery. In addition, there is evidence to suggest that although the surgery may take longer, robotic-assisted surgery may result in shorter hospital stays.[92] More research is necessary to determine if robotic-assisted hysterectomies are beneficial for people with cancer.[92]
Previously reported marginal advantages of robotic-assisted surgery could not be confirmed; only differences in hospital stay and cost remain statistically significant.[93][94][95] In addition, concerns over widespread misleading marketing claims have been raised.[96]
| Technique | Benefits | Disadvantages |
|---|---|---|
| Abdominal hysterectomy |
|
|
| Vaginal hysterectomy | ||
| Laparoscopic supracervical hysterectomy (subtotal hysterectomy) | ||
| Laparoscopic-assisted vaginal hysterectomy (LAVH) |
|
|
| Total laparoscopic hysterectomy |
|
|
| Single-port laparoscopic hysterectomy / mini laparoscopic hysterectomy |
| |
| Robotic-assisted hysterectomy |
Incidence
[edit]Canada
[edit]In Canada, the number of hysterectomies between 2008 and 2009 was almost 47,000. The national rate for the same timeline was 338 per 100,000 population, down from 484 per 100,000 in 1997. The reasons for hysterectomies differed depending on whether the woman was living in an urban or rural location. Urban women opted for hysterectomies due to uterine fibroids and rural women had hysterectomies mostly for menstrual disorders.[101]
United States
[edit]Hysterectomy is the second most common major surgery among women in the United States (the first is cesarean section). In the 1980s and 1990s, this statistic was the source of concern among some consumer rights groups and puzzlement among the medical community,[102] and brought about informed choice advocacy groups like Hysterectomy Educational Resources and Services (HERS) Foundation, founded by Nora W. Coffey in 1982.
According to the National Center for Health Statistics, of the 617,000 hysterectomies performed in 2004, 73% also involved the surgical removal of the ovaries. There are currently an estimated 22 million women in the United States who have undergone this procedure. Nearly 68 percent were performed for benign conditions such as endometriosis, irregular bleeding, and uterine fibroids.[1] Such rates being highest in the industrialized world has led to the controversy that hysterectomies are being largely performed for unwarranted reasons.[103] More recent data suggests that the number of hysterectomies performed has declined in every state in the United States. From 2010 to 2013, there were 12 percent fewer hysterectomies performed, and the types of hysterectomies were more minimally invasive in nature, reflected by a 17 percent increase in laparoscopic procedures.[104]
United Kingdom
[edit]In the UK, 1 in 5 women is likely to have a hysterectomy by the age of 60, and ovaries are removed in about 20% of hysterectomies.[105]
Germany
[edit]The number of hysterectomies in Germany has been constant for many years. In 2006, 149,456 hysterectomies were performed. Additionally, of these, 126,743 (84.8%) successfully benefited the patient without incident. Women between the ages of 40 and 49 accounted for 50 percent of hysterectomies, and those between the ages of 50 and 59 accounted for 20 percent.[106] In 2007, the number of hysterectomies decreased to 138,164.[64] In recent years, the technique of laparoscopic or laparoscopically assisted hysterectomies has been raised into the foreground.[107][108]
Denmark
[edit]In Denmark, the number of hysterectomies from the 1980s to the 1990s decreased by 38 percent. In 1988, there were 173 such surgeries per 100,000 women, and by 1998, this number had been reduced to 107. The proportion of abdominal supracervical hysterectomies in the same period grew from 7.5 to 41 percent. A total of 67,096 women underwent hysterectomy during these years.[109]
See also
[edit]References
[edit]- ^ a b c "Plotting the downward trend in traditional hysterectomy | Institute for Healthcare Policy & Innovation". ihpi.umich.edu. Retrieved 2019-08-06.
- ^ Bahamondes L, Bahamondes MV, Monteiro I (July 2008). "Levonorgestrel-releasing intrauterine system: uses and controversies". Expert Review of Medical Devices. 5 (4): 437–445. doi:10.1586/17434440.5.4.437. PMID 18573044. S2CID 659602.
- ^ "Hysterectomy". womenshealth.gov. 2017-02-21. Retrieved 2019-08-06.
- ^ Parasar P, Ozcan P, Terry KL (March 2017). "Endometriosis: Epidemiology, Diagnosis and Clinical Management". Current Obstetrics and Gynecology Reports. 6 (1): 34–41. doi:10.1007/s13669-017-0187-1. PMC 5737931. PMID 29276652.
- ^ "Uterine Adenomyosis". Yale Medicine. Retrieved 2022-10-31.
- ^ Uterine fibroids: Overview. Institute for Quality and Efficiency in Health Care (IQWiG). 2017-11-16.
- ^ Temkin SM, Bergstrom J, Samimi G, Minasian L (December 2017). "Ovarian Cancer Prevention in High-risk Women". Clinical Obstetrics and Gynecology. 60 (4): 738–757. doi:10.1097/GRF.0000000000000318. PMC 5920567. PMID 28957949.
- ^ "Hysterectomy | Transgender Care". transcare.ucsf.edu. Retrieved 2019-08-02.
- ^ Washington (state) Protection and Advocacy System. "Growth Attenuation and Sterilization Procedures – "The Ashley Treatment"". Washington, DC: National Disabilities Rights Network. Archived from the original on July 27, 2011. Retrieved 2011-03-10.
- ^ Roopnarinesingh R, Fay L, McKenna P (May 2003). "A 27-year review of obstetric hysterectomy". Journal of Obstetrics and Gynaecology. 23 (3): 252–254. doi:10.1080/0144361031000098352. PMID 12850853. S2CID 28253372.
- ^ Lamvu G (May 2011). "Role of hysterectomy in the treatment of chronic pelvic pain". Obstetrics and Gynecology. 117 (5): 1175–1178. doi:10.1097/AOG.0b013e31821646e1. PMID 21508759.
- ^ McPherson K, Metcalfe MA, Herbert A, Maresh M, Casbard A, Hargreaves J, et al. (July 2004). "Severe complications of hysterectomy: the VALUE study". BJOG. 111 (7): 688–694. doi:10.1111/j.1471-0528.2004.00174.x. PMID 15198759. S2CID 38391308.
- ^ Wingo PA, Huezo CM, Rubin GL, Ory HW, Peterson HB (August 1985). "The mortality risk associated with hysterectomy". American Journal of Obstetrics and Gynecology. 152 (7 Pt 1): 803–808. doi:10.1016/s0002-9378(85)80067-3. PMID 4025434.
- ^ Shuster LT, Gostout BS, Grossardt BR, Rocca WA (September 2008). "Prophylactic oophorectomy in premenopausal women and long-term health". Menopause International. 14 (3): 111–116. doi:10.1258/mi.2008.008016. PMC 2585770. PMID 18714076.
- ^ American Urogynecologic Society (May 5, 2015), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, American Urogynecologic Society, retrieved June 1, 2015, which cites: * Blank SV (February 2011). "Prophylactic and risk-reducing bilateral salpingo-oophorectomy: recommendations based on risk of ovarian cancer". Obstetrics and Gynecology. 117 (2 Pt 1): 404. doi:10.1097/AOG.0b013e3182083189. PMID 21252760.
- ^ Shoupe D, Parker WH, Broder MS, Liu Z, Farquhar C, Berek JS (2007). "Elective oophorectomy for benign gynecological disorders". Menopause. 14 (3 Pt 2): 580–585. doi:10.1097/gme.0b013e31803c56a4. PMID 17476148. S2CID 37549821.
- ^ Madueke-Laveaux OS, Elsharoud A, Al-Hendy A (November 2021). "What We Know about the Long-Term Risks of Hysterectomy for Benign Indication-A Systematic Review". Journal of Clinical Medicine. 10 (22): 5335. doi:10.3390/jcm10225335. PMC 8622061. PMID 34830617.
- ^ Burks FN, Santucci RA (June 2014). "Management of iatrogenic ureteral injury". Therapeutic Advances in Urology. 6 (3): 115–124. doi:10.1177/1756287214526767. PMC 4003841. PMID 24883109.
- ^ Ureteral Trauma at eMedicine
- ^ "Abdominal hysterectomy". Mayo Clinic.
- ^ "Hysterectomy". www.acog.org. Retrieved 2021-12-28.
- ^ a b Laughlin GA, Barrett-Connor E, Kritz-Silverstein D, von Mühlen D (February 2000). "Hysterectomy, oophorectomy, and endogenous sex hormone levels in older women: the Rancho Bernardo Study". The Journal of Clinical Endocrinology and Metabolism. 85 (2): 645–651. doi:10.1210/jcem.85.2.6405. PMID 10690870. S2CID 8195493.
- ^ Farquhar CM, Sadler L, Harvey SA, Stewart AW (July 2005). "The association of hysterectomy and menopause: a prospective cohort study". BJOG. 112 (7): 956–962. doi:10.1111/j.1471-0528.2005.00696.x. PMID 15957999. S2CID 21619116.
- ^ Bofill Rodriguez M, Lethaby A, Grigore M, Brown J, Hickey M, Farquhar C (January 2019). "Endometrial resection and ablation techniques for heavy menstrual bleeding". The Cochrane Database of Systematic Reviews. 1 (1) CD001501. doi:10.1002/14651858.CD001501.pub5. PMC 7057272. PMID 30667064.
- ^ Petri Nahás EA, Pontes A, Nahas-Neto J, Borges VT, Dias R, Traiman P (February 2005). "Effect of total abdominal hysterectomy on ovarian blood supply in women of reproductive age". Journal of Ultrasound in Medicine. 24 (2): 169–174. doi:10.7863/jum.2005.24.2.169. hdl:11449/224482. PMID 15661947. S2CID 30259666.
- ^ Maas CP, Weijenborg PT, ter Kuile MM (2003). "The effect of hysterectomy on sexual functioning". Annual Review of Sex Research. 14: 83–113. PMID 15287159.
- ^ Komisaruk BR, Frangos E, Whipple B (2011). "Hysterectomy improves sexual response? Addressing a crucial omission in the literature". Journal of Minimally Invasive Gynecology. 18 (3): 288–295. doi:10.1016/j.jmig.2011.01.012. PMC 3090744. PMID 21545957.
- ^ Gunter J (September 2003). "Chronic pelvic pain: an integrated approach to diagnosis and treatment". Obstetrical & Gynecological Survey. 58 (9): 615–623. doi:10.1097/01.OGX.0000083225.90017.01. PMID 12972837. S2CID 25101266.
- ^ Dalal PK, Agarwal M (July 2015). "Postmenopausal syndrome". Indian Journal of Psychiatry. 57 (Suppl 2): S222 – S232. doi:10.4103/0019-5545.161483. PMC 4539866. PMID 26330639.
- ^ Ingelsson E, Lundholm C, Johansson AL, Altman D (March 2011). "Hysterectomy and risk of cardiovascular disease: a population-based cohort study". European Heart Journal. 32 (6): 745–750. doi:10.1093/eurheartj/ehq477. PMID 21186237.
- ^ Kelsey JL, Prill MM, Keegan TH, Quesenberry CP, Sidney S (November 2005). "Risk factors for pelvis fracture in older persons". American Journal of Epidemiology. 162 (9): 879–886. doi:10.1093/aje/kwi295. PMID 16221810.
- ^ van der Voort DJ, Geusens PP, Dinant GJ (2001). "Risk factors for osteoporosis related to their outcome: fractures". Osteoporosis International. 12 (8): 630–638. doi:10.1007/s001980170062. PMID 11580076. S2CID 9421669.
- ^ Jassal SK, Barrett-Connor E, Edelstein SL (April 1995). "Low bioavailable testosterone levels predict future height loss in postmenopausal women". Journal of Bone and Mineral Research. 10 (4): 650–654. doi:10.1002/jbmr.5650100419. PMID 7610937. S2CID 30094806.
- ^ Segraves R, Woodard T (May 2006). "Female hypoactive sexual desire disorder: History and current status". The Journal of Sexual Medicine. 3 (3): 408–418. doi:10.1111/j.1743-6109.2006.00246.x. PMID 16681466. S2CID 39458239.
- ^ Rivera CM, Grossardt BR, Rhodes DJ, Rocca WA (2009). "Increased mortality for neurological and mental diseases following early bilateral oophorectomy". Neuroepidemiology. 33 (1): 32–40. doi:10.1159/000211951. PMC 2697609. PMID 19365140.
- ^ Brown JS, Sawaya G, Thom DH, Grady D (August 2000). "Hysterectomy and urinary incontinence: a systematic review". Lancet. 356 (9229): 535–539. doi:10.1016/S0140-6736(00)02577-0. PMID 10950229. S2CID 213231.
- ^ Altman D, Granath F, Cnattingius S, Falconer C (October 2007). "Hysterectomy and risk of stress-urinary-incontinence surgery: nationwide cohort study". Lancet. 370 (9597): 1494–1499. doi:10.1016/S0140-6736(07)61635-3. PMID 17964350. S2CID 27132159.
- ^ McPherson K, Herbert A, Judge A, Clarke A, Bridgman S, Maresh M, Overton C (July 2005). "Self-reported bladder function five years post-hysterectomy". Journal of Obstetrics and Gynaecology. 25 (5): 469–475. doi:10.1080/01443610500235170. PMID 16183583. S2CID 38512052.
- ^ Lukanovic A, Drazic K (July 2010). "Risk factors for vaginal prolapse after hysterectomy". International Journal of Gynaecology and Obstetrics. 110 (1): 27–30. doi:10.1016/j.ijgo.2010.01.025. PMID 20362288. S2CID 24322065.
- ^ Altman D, Falconer C, Cnattingius S, Granath F (May 2008). "Pelvic organ prolapse surgery following hysterectomy on benign indications". American Journal of Obstetrics and Gynecology. 198 (5): 572.e1–572.e6. doi:10.1016/j.ajog.2008.01.012. PMID 18355787.
- ^ Wiseman DM (July 2008). "Disorders of adhesions or adhesion-related disorder: monolithic entities or part of something bigger--CAPPS?". Seminars in Reproductive Medicine. 26 (4): 356–368. CiteSeerX 10.1.1.628.2706. doi:10.1055/s-0028-1082394. PMID 18756413. S2CID 260317149.
- ^ a b Barmparas G, Branco BC, Schnüriger B, Lam L, Inaba K, Demetriades D (October 2010). "The incidence and risk factors of post-laparotomy adhesive small bowel obstruction". Journal of Gastrointestinal Surgery. 14 (10): 1619–1628. doi:10.1007/s11605-010-1189-8. PMID 20352368. S2CID 22720831.
- ^ a b c Duff P (2009). "Diagnosis and Management of Postoperative Infection". The Global Library of Women's Medicine. doi:10.3843/GLOWM.10032. ISSN 1756-2228.
- ^ Hoffman B (2012). Williams gynecology, 2nd edition. New York: McGraw-Hill Medical. p. 65. ISBN 978-0-07-171672-7.
- ^ Altman D, Yin L, Johansson A, Lundholm C, Grönberg H (December 2010). "Risk of renal cell carcinoma after hysterectomy". Archives of Internal Medicine. 170 (22): 2011–2016. doi:10.1001/archinternmed.2010.425. PMID 21149759.
- ^ Gago-Dominguez M, Castelao JE, Yuan JM, Ross RK, Yu MC (November 1999). "Increased risk of renal cell carcinoma subsequent to hysterectomy". Cancer Epidemiology, Biomarkers & Prevention. 8 (11): 999–1003. PMID 10566555.
- ^ Zucchetto A, Talamini R, Dal Maso L, Negri E, Polesel J, Ramazzotti V, et al. (November 2008). "Reproductive, menstrual, and other hormone-related factors and risk of renal cell cancer". International Journal of Cancer. 123 (9): 2213–2216. doi:10.1002/ijc.23750. PMID 18711701. S2CID 45289039.
- ^ Cocks PS (May 1980). "Early ectopic pregnancy after vaginal hysterectomy. Two case reports". British Journal of Obstetrics and Gynaecology. 87 (5): 363–365. doi:10.1111/j.1471-0528.1980.tb04559.x. PMID 7387935. S2CID 27101990.
- ^ Quiróz-Guadarrama CD, Martínez-Ordaz JL, Rojano-Rodríguez ME, Beristain-Hernández JL, Moreno-Portillo M (June 2013). "[Vaginal evisceration. Report of a case and a literature review]". Ginecologia y Obstetricia de Mexico. 81 (6): 349–352. PMID 23837301.
- ^ Jelovsek JE, Maher C, Barber MD (March 2007). "Pelvic organ prolapse". Lancet. 369 (9566): 1027–1038. doi:10.1016/s0140-6736(07)60462-0. PMID 17382829. S2CID 38561018.
- ^ Milsom I (June 2007). "The levonorgestrel-releasing intrauterine system as an alternative to hysterectomy in peri-menopausal women". Contraception. 75 (6 Suppl): S152 – S154. doi:10.1016/j.contraception.2007.01.003. PMID 17531608.
- ^ Bofill Rodriguez M, Lethaby A, Fergusson RJ (February 2021). "Endometrial resection and ablation versus hysterectomy for heavy menstrual bleeding". The Cochrane Database of Systematic Reviews. 2021 (2) CD000329. doi:10.1002/14651858.CD000329.pub4. PMC 8095059. PMID 33619722.
- ^ Pesmen C (July 27, 2007). "5 operations you don't want to get – and what to do instead". CNN.
- ^ Zapata LB, Whiteman MK, Tepper NK, Jamieson DJ, Marchbanks PA, Curtis KM (July 2010). "Intrauterine device use among women with uterine fibroids: a systematic review". Contraception. 82 (1): 41–55. doi:10.1016/j.contraception.2010.02.011. PMID 20682142.
- ^ Parker WH, Parker RL (2002). A Gynecologist's Second Opinion: The Questions & Answers You Need to Take Charge of Your Health (revised ed.). Plume. pp. 89–92, 105–150. Archived from the original on 2007-06-09.
- ^ Laios A, Baharuddin N, Iliou K, Gubara E, O'Sullivan G (2014). "Uterine artery embolization for treatment of symptomatic fibroids; a single institution experience". Hippokratia. 18 (3): 258–261. PMC 4309148. PMID 25694762.
- ^ Gupta JK, Sinha A, Lumsden MA, Hickey M (December 2014). "Uterine artery embolization for symptomatic uterine fibroids". The Cochrane Database of Systematic Reviews (12) CD005073. doi:10.1002/14651858.CD005073.pub4. PMC 11285296. PMID 25541260.
- ^ Jelovsek FR. "Having Prolapse, Cystocele and Rectocele Fixed Without Hysterectomy". Women's Health. wdxcyber.com.
- ^ "Hysterectomy". womenshealth.gov. 2017-02-21. Retrieved 2019-08-01.
- ^ "Complete Hysterectomy". www.cancer.gov. 2011-02-02. Retrieved 2022-10-31.
- ^ encyclopedia.com > Wertheim's hysterectomy Citing: "Wertheim's hysterectomy". A Dictionary of Nursing. 2008. Encyclopedia.com. (October 13, 2010).
- ^ Greer WJ, Richter HE, Wheeler TL, Varner RE, Szychowski JM, Kuppermann M, Learman LA (January 2010). "Long-Term Outcomes of the Total or Supracervical Hysterectomy (TOSH) Trial". Female Pelvic Medicine & Reconstructive Surgery. 16 (1): 49–57. doi:10.1097/SPV.0b013e3181cec343. PMC 3252027. PMID 22229107.
- ^ Lethaby A, Mukhopadhyay A, Naik R (April 2012). Lethaby A (ed.). "Total versus subtotal hysterectomy for benign gynaecological conditions". The Cochrane Database of Systematic Reviews. 4 (4) CD004993. doi:10.1002/14651858.CD004993.pub3. PMID 22513925.
- ^ a b Müller A, Thiel FC, Renner SP, Winkler M, Häberle L, Beckmann MW (May 2010). "Hysterectomy-a comparison of approaches". Deutsches Ärzteblatt International. 107 (20): 353–359. doi:10.3238/arztebl.2010.0353. PMC 2883234. PMID 20539807.
- ^ Thakar R, Ayers S, Clarkson P, Stanton S, Manyonda I (October 2002). "Outcomes after total versus subtotal abdominal hysterectomy". The New England Journal of Medicine. 347 (17): 1318–1325. doi:10.1056/NEJMoa013336. PMID 12397189.
- ^ American Academy of Family Physicians (April 2012). "Five Things Physicians and Patients Should Question" (PDF). Choosing Wisely: An Initiative of the ABIM Foundation. American Academy of Family Physicians. Archived from the original (PDF) on June 24, 2012. Retrieved August 14, 2012.
- ^ Consumer Reports; American Academy of Family Physicians (May 2012). "Pap tests: When you need them—and when you don't" (PDF). Choosing Wisely: An Initiative of the ABIM Foundation. Consumer Reports. Archived from the original (PDF) on June 24, 2012. Retrieved August 17, 2012.
- ^ Pillarisetty LS, Mahdy H (2022). "Vaginal Hysterectomy". StatPearls. Treasure Island (FL): StatPearls Publishing. PMID 32119369. Retrieved 2022-10-31.
- ^ "Hysterectomy: Then and Now - Oxford Women's Health". www.oxfordwomenshealth.co.nz. Archived from the original on 2023-10-17. Retrieved 2020-01-27.
- ^ Alazzam M, Khalifa MA, Al-Ani A (May 2022). "The transverse-vertical incision (Alazzam hybrid incision)". Langenbeck's Archives of Surgery. 407 (3): 1303–1309. doi:10.1007/s00423-021-02404-5. PMC 9151581. PMID 35226178.
- ^ a b Thomas B, Magos A (April 2011). "Subtotal hysterectomy and myomectomy - vaginally". Best Practice & Research. Clinical Obstetrics & Gynaecology. 25 (2): 133–152. doi:10.1016/j.bpobgyn.2010.11.003. PMID 21185235.
- ^ a b Sheth SS, Paghdiwalla KP, Hajari AR (April 2011). "Vaginal route: a gynaecological route for much more than hysterectomy". Best Practice & Research. Clinical Obstetrics & Gynaecology. 25 (2): 115–132. doi:10.1016/j.bpobgyn.2010.12.005. PMID 21349773.
- ^ a b Johnson N, Barlow D, Lethaby A, Tavender E, Curr L, Garry R (June 2005). "Methods of hysterectomy: systematic review and meta-analysis of randomised controlled trials". BMJ. 330 (7506): 1478. doi:10.1136/bmj.330.7506.1478. PMC 558455. PMID 15976422.
- ^ Pickett CM, Seeratan DD, Mol BW, Nieboer TE, Johnson N, Bonestroo T, Aarts JW (August 2023). "Surgical approach to hysterectomy for benign gynaecological disease". The Cochrane Database of Systematic Reviews. 2023 (8) CD003677. doi:10.1002/14651858.CD003677.pub6. PMC 10464658. PMID 37642285.
- ^ "Surgery - Foundation for Women's Cancer". Foundation for Women's Cancer. Archived from the original on 2018-11-07. Retrieved 2018-11-07.
- ^ Cronin B, Sung VW, Matteson KA (April 2012). "Vaginal cuff dehiscence: risk factors and management". American Journal of Obstetrics and Gynecology. 206 (4): 284–288. doi:10.1016/j.ajog.2011.08.026. PMC 3319233. PMID 21974989.
- ^ Devassy R, Cezar C, Krentel H, Verhoeven HC, Devassy R, de Wilde MS, et al. (February 2019). "Feasibility of myomatous tissue extraction in laparoscopic surgery by contained in - bag morcellation: A retrospective single arm study". International Journal of Surgery. 62: 22–27. doi:10.1016/j.ijsu.2018.12.013. PMID 30639472. S2CID 58564050.
- ^ Ahluwalia PK (August 1996). "Total Laparoscopic Hysterectomy". The Journal of the American Association of Gynecologic Laparoscopists. 3 (4, Supplement): S1 – S2. doi:10.1016/S1074-3804(96)80129-0. PMID 9074073.
- ^ Cipullo L, De Paoli S, Fasolino L, Fasolino A (2009). "Laparoscopic supracervical hysterectomy compared to total hysterectomy". JSLS. 13 (3): 370–375. PMC 3015973. PMID 19793479.
- ^ Lii SJ, Becker SF, Danilyants NE, MacKoul PJ (2010). "A Novel Approach to Total Laparoscopic Hysterectomy Using Only Two 5mm Ports: Initial Clinical Experience". Journal of Minimally Invasive Gynecology. 17 (6): S87. doi:10.1016/j.jmig.2010.08.381.
- ^ Moawad G, Robinson JK (2012). "Dual Port Hysterectomy: A Novel Technique and Initial Experience". Journal of Minimally Invasive Gynecology. 19 (6): S86. doi:10.1016/j.jmig.2012.08.620.
- ^ "Robotic surgery: MedlinePlus Medical Encyclopedia". medlineplus.gov.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z Aarts JW, Nieboer TE, Johnson N, Tavender E, Garry R, Mol BW, Kluivers KB (August 2015). "Surgical approach to hysterectomy for benign gynaecological disease". The Cochrane Database of Systematic Reviews. 2015 (8) CD003677. doi:10.1002/14651858.CD003677.pub5. hdl:2066/154037. PMC 6984437. PMID 26264829.
- ^ Stovall TG, Summitt RL (August 1996). "Laparoscopic hysterectomy--is there a benefit?". The New England Journal of Medicine. 335 (7): 512–513. doi:10.1056/NEJM199608153350712. PMID 8672159.
- ^ "Laparoscopic Hysterectomy and Health Care in America – Finding the Balance Between Costs and Outcomes". Archived from the original on 2010-07-17. Retrieved 2010-01-24.
- ^ Debodinance P (April 2001). "[Hysterectomy for benign lesions in the north of France: epidemiology and postoperative events]". Journal de Gynécologie, Obstétrique et Biologie de la Reproduction. 30 (2): 151–159. PMID 11319467.
- ^ a b Garry R, Fountain J, Mason S, Hawe J, Napp V, Abbott J, et al. (January 2004). "The eVALuate study: two parallel randomised trials, one comparing laparoscopic with abdominal hysterectomy, the other comparing laparoscopic with vaginal hysterectomy". BMJ. 328 (7432): 129–0. doi:10.1136/bmj.37984.623889.F6. PMC 314503. PMID 14711749.
- ^ Boosz A, Haeberle L, Renner SP, Thiel FC, Mehlhorn G, Beckmann MW, Mueller A (December 2014). "Comparison of reoperation rates, perioperative outcomes in women with endometrial cancer when the standard of care shifts from open surgery to laparoscopy". Archives of Gynecology and Obstetrics. 290 (6): 1215–1220. doi:10.1007/s00404-014-3347-9. PMID 25009071. S2CID 25074196.
- ^ Seidman MA, Oduyebo T, Muto MG, Crum CP, Nucci MR, Quade BJ (2012). Sullivan DJ (ed.). "Peritoneal dissemination complicating morcellation of uterine mesenchymal neoplasms". PLOS ONE. 7 (11) e50058. Bibcode:2012PLoSO...750058S. doi:10.1371/journal.pone.0050058. PMC 3506532. PMID 23189178.
- ^ Cucinella G, Granese R, Calagna G, Somigliana E, Perino A (August 2011). "Parasitic myomas after laparoscopic surgery: an emerging complication in the use of morcellator? Description of four cases". Fertility and Sterility. 96 (2): e90 – e96. doi:10.1016/j.fertnstert.2011.05.095. hdl:10447/77912. PMID 21719004.
- ^ "Safety Communications – UPDATED Laparoscopic Uterine Power Morcellation in Hysterectomy and Myomectomy: FDA Safety Communication". Center for Devices and Radiological Health. U.S. Food and Drug Administration. Archived from the original on December 4, 2014. Retrieved 2017-01-10.
- ^ a b c d e f g h Lawrie TA, Liu H, Lu D, Dowswell T, Song H, Wang L, Shi G (April 2019). "Robot-assisted surgery in gynaecology". The Cochrane Database of Systematic Reviews. 4 (4) CD011422. doi:10.1002/14651858.CD011422.pub2. PMC 6464707. PMID 30985921.
- ^ a b Wright JD, Ananth CV, Lewin SN, Burke WM, Lu YS, Neugut AI, et al. (February 2013). "Robotically assisted vs laparoscopic hysterectomy among women with benign gynecologic disease". JAMA. 309 (7): 689–698. doi:10.1001/jama.2013.186. PMID 23423414.
- ^ Soto E, Lo Y, Friedman K, Soto C, Nezhat F, Chuang L, Gretz H (December 2011). "Total laparoscopic hysterectomy versus da Vinci robotic hysterectomy: is using the robot beneficial?". Journal of Gynecologic Oncology. 22 (4): 253–259. doi:10.3802/jgo.2011.22.4.253. PMC 3254844. PMID 22247802.
- ^ Ridgeway BM, Buechel M, Nutter B, Falcone T (December 2015). "Minimally Invasive Hysterectomy: An Analysis of Different Techniques". Clinical Obstetrics and Gynecology. 58 (4): 732–739. doi:10.1097/GRF.0000000000000149. PMID 26457851. S2CID 42747985.
- ^ Schiavone MB, Kuo EC, Naumann RW, Burke WM, Lewin SN, Neugut AI, et al. (September 2012). "The commercialization of robotic surgery: unsubstantiated marketing of gynecologic surgery by hospitals". American Journal of Obstetrics and Gynecology. 207 (3): 174.e1–174.e7. doi:10.1016/j.ajog.2012.06.050. PMID 22835493.
- ^ Blikkendaal MD, Schepers EM, van Zwet EW, Twijnstra AR, Jansen FW (October 2015). "Hysterectomy in very obese and morbidly obese patients: a systematic review with cumulative analysis of comparative studies". Archives of Gynecology and Obstetrics. 292 (4): 723–738. doi:10.1007/s00404-015-3680-7. PMC 4560773. PMID 25773357.
- ^ a b c d e f g h King CR, Giles D (September 2016). "Total Laparoscopic Hysterectomy and Laparoscopic-Assisted Vaginal Hysterectomy". Obstetrics and Gynecology Clinics of North America. 43 (3): 463–478. doi:10.1016/j.ogc.2016.04.005. PMID 27521879.
- ^ a b c d e Nesbitt-Hawes EM, Maley PE, Won HR, Law KS, Zhang CS, Lyons SD, et al. (2013). "Laparoscopic subtotal hysterectomy: evidence and techniques". Journal of Minimally Invasive Gynecology. 20 (4): 424–434. doi:10.1016/j.jmig.2013.01.009. PMID 23510954.
- ^ a b Lethaby A, Mukhopadhyay A, Naik R (April 2012). "Total versus subtotal hysterectomy for benign gynaecological conditions". The Cochrane Database of Systematic Reviews. 4 (4) CD004993. doi:10.1002/14651858.CD004993.pub3. PMID 22513925.
- ^ "Hysterectomy rates falling: report". CBC News. 2010-05-27. Retrieved 2010-05-28.
- ^ Kolata, Gina (1988-09-20). "Rate of Hysterectomies Puzzles Experts". The New York Times.
- ^ Masters C (2006-07-01). "Are Hysterectomies Too Common?". Time. Retrieved 2007-07-17.
- ^ "Hysterectomy Procedures Pacing A Downward Trend". labblog.uofmhealth.org. 23 January 2018. Retrieved 2019-08-06.
- ^ Khastgir G, Studd J (1998). Hysterectomy and HRT. Taylor & Francis. p. 3. ISBN 978-1-85317-408-7.
- ^ Wolfrum C (1 June 2008). "Vorschnelle Schnitte". Apotheken Umschau. Baierbrunn: Wort & Bild Verlag.
- ^ Mettler L, Ahmed-Ebbiary N, Schollmeyer T (2005). "Laparoscopic hysterectomy: challenges and limitations". Minimally Invasive Therapy & Allied Technologies. 14 (3): 145–159. doi:10.1080/13645700510034010. PMID 16754157. S2CID 25696299.
- ^ Jäger C, Sauer G, Kreienberg R (2007). "Die laparoskopisch assistierte vaginale Hysterektomie – Sinn oder Unsinn?". Geburtshilfe und Frauenheilkunde. 67 (6): 628–632. doi:10.1055/s-2007-965243. S2CID 260157584.
- ^ Gimbel H, Settnes A, Tabor A (March 2001). "Hysterectomy on benign indication in Denmark 1988-1998. A register based trend analysis". Acta Obstetricia et Gynecologica Scandinavica. 80 (3): 267–272. doi:10.1080/j.1600-0412.2001.080003267.x. PMID 11207494.
External links
[edit]Hysterectomy
View on GrokipediaHistory
Ancient and Pre-Modern Procedures
The earliest documented attempts at hysterectomy occurred in ancient Greece via the vaginal route, primarily for severe uterine prolapse or inversion, though prior mentions in medical texts around 2000 years ago lack confirmatory evidence of execution.[13] Themison of Athens reportedly conducted the first such procedure circa 50 BCE on a patient with a gangrenous prolapsed uterus, involving direct vaginal extirpation without anesthesia or hemostatic measures.[14] Similarly, Soranus of Ephesus (c. 98–138 CE), a Greek physician practicing in Rome, performed vaginal hysterectomies for inverted uteri, describing techniques that relied on traction and incision but yielded high mortality from uncontrolled bleeding, sepsis, and shock due to the absence of antisepsis or vascular ligation.[15][16] These interventions were exceptional, reserved for life-threatening conditions, as the uterus was viewed in Hippocratic and Galenic traditions as a mobile organ prone to displacement causing hysteria-like symptoms, yet surgical removal was deemed too hazardous for routine application.[13] In the pre-modern era, spanning the medieval and early modern periods through the 18th century, vaginal hysterectomies remained sporadic and largely limited to cases of traumatic uterine inversion during childbirth or necrotic prolapse, with procedures entailing crude manual disarticulation or knifing through vaginal tissues.[17] European and Islamic medical texts, such as those by 11th-century Arabic surgeon Albucasis (Abū al-Qāsim al-Zahrāwī), alluded to uterine surgeries but provided no verified hysterectomy successes, emphasizing conservative treatments like cauterization or pessaries instead.[18] Survival rates were negligible, often below 10% in recorded attempts, attributable to peritonitis from fecal contamination, exsanguination without clamps or ligatures, and postoperative exhaustion, rendering the operation a last-resort measure performed by midwives or barber-surgeons rather than formalized gynecology.[15] Abdominal approaches were entirely precluded until the 19th century, as peritoneal cavity exposure invited fatal infections without Listerian principles.[2] Overall, pre-modern hysterectomies exemplified the era's surgical constraints, prioritizing palliation over excision amid profound risks.[19]19th-Century Advancements
The first planned vaginal hysterectomy was performed by Conrad Langenbeck in Göttingen, Germany, in 1813, on a patient with cervical cancer; the procedure involved prolapsing the uterus and ligating vessels, though early vaginal approaches had been attempted sporadically since ancient times with limited success due to hemorrhage and infection risks.[15] Abdominal hysterectomy emerged later in the century, with Charles Clay conducting the first recorded procedure in Manchester, England, on November 17, 1843, for what was believed to be an ovarian fibroid but proved to be a misdiagnosis; the patient succumbed postoperatively, highlighting the era's challenges including uncontrolled bleeding and sepsis.[15] [20] Subsequent attempts in the 1840s and 1850s yielded variable outcomes, as surgeons like Ellis Burnham achieved the first successful abdominal hysterectomy in Lowell, Massachusetts, in 1853, removing a uterine fibroid while preserving the uterus initially, though full extirpation remained perilous with mortality rates exceeding 70% in many series due to inadequate hemostasis and lack of antisepsis.[21] Vaginal techniques advanced concurrently, with Paul F. Eve performing the first documented successful vaginal hysterectomy in the United States in Augusta, Georgia, around 1843 for inversion of the uterus, yet overall survival hinged on case selection for benign conditions like prolapse rather than malignancy.[19] Key enablers included the introduction of ether anesthesia in 1846, which facilitated longer intra-abdominal operations, and James Simpson's use of chloroform in 1847, reducing patient trauma during procedures.[22] By mid-century, surgeons such as Nathan Bozeman and Washington L. Atlee refined ligation methods for ovarian vessels, improving hemostasis in abdominal approaches, while Thomas Keith in Edinburgh reported over 100 vaginal hysterectomies by the 1870s with mortality dropping to around 10% through meticulous technique.[13] Late-19th-century progress accelerated with Joseph Lister's antisepsis principles from 1867, which drastically curbed postoperative infections; Emil Freund standardized abdominal hysterectomy in the 1880s, emphasizing en bloc removal for fibroids and incorporating silver clamps for vessel control, contributing to mortality reductions below 20% in specialized centers by 1890.[23] [24] These advancements shifted hysterectomy from experimental desperation—often for uncontrollable uterine hemorrhage or large myomas—to a more viable intervention, though it remained controversial owing to ethical debates over elective removal in non-life-threatening cases and persistent risks compared to conservative therapies.[15]20th-Century Milestones and Modern Refinements
In the early 20th century, subtotal abdominal hysterectomy, which removes the uterine corpus while preserving the cervix, remained the predominant technique due to technical challenges in safely excising the cervix and concerns over postoperative complications. By the 1950s, total abdominal hysterectomy—encompassing removal of both the corpus and cervix—superseded it, driven by evidence of reduced risk for cervical stump carcinoma and advancements in surgical precision and anesthesia.[13][25] The discovery of antibiotics in the 1940s and maturation of blood transfusion techniques in the 1930s dramatically lowered infection and hemorrhage-related mortality, transforming hysterectomy from a high-risk procedure with rates exceeding 10% in the mid-19th century to safer operations with complication rates under 5% by mid-century.[26][27] Laparoscopic approaches emerged in the late 1980s, with Harry Reich performing the first laparoscopic hysterectomy in 1988, followed by the inaugural total laparoscopic hysterectomy in 1989, enabling visualization and dissection through small incisions and reducing recovery times compared to open surgery.[28][29] Modern refinements emphasize minimally invasive methods, including laparoscopically assisted vaginal hysterectomy (LAVH) and robotic-assisted techniques, which leverage articulated instruments and enhanced optics for improved dexterity in complex cases, though evidence shows comparable outcomes to conventional laparoscopy with potentially higher costs. Vaginal natural orifice transluminal endoscopic surgery (vNOTES), gaining traction since the 2010s, further minimizes scarring by accessing the uterus transvaginally without abdominal ports.[30][31]Indications and Benefits
Benign Conditions
Hysterectomy serves as a definitive treatment for several benign gynecological conditions, particularly when symptoms severely impair quality of life and conservative therapies—such as hormonal medications, minimally invasive procedures, or watchful waiting—prove inadequate or unsuitable. In the United States, benign indications account for the majority of the approximately 500,000 to 600,000 hysterectomies performed annually, with procedures increasingly favoring minimally invasive routes to reduce recovery time and complications.[32][8] Common benign pathologies include uterine leiomyomas, abnormal uterine bleeding, adenomyosis, endometriosis, and pelvic organ prolapse, each involving distinct pathophysiological mechanisms leading to pain, hemorrhage, or structural failure. Uterine leiomyomas, or fibroids, represent the leading benign indication, comprising about 51% of cases. These benign tumors arise from smooth muscle proliferation in the myometrium, affecting up to 80% of women by age 50, with symptomatic growth causing heavy menstrual bleeding, pelvic pressure, anemia, or infertility through distortion of the uterine cavity or vasculature compression. Hysterectomy is pursued when fibroids exceed 10-12 weeks' gestational size or fail response to alternatives like uterine artery embolization or myomectomy, providing complete resolution absent in conservative options.[8][33][34] Abnormal uterine bleeding (AUB), often structural or ovulatory dysfunction-related, accounts for roughly 42% of benign hysterectomies and manifests as prolonged or excessive menses leading to hemoglobin levels below 10 g/dL in severe instances. Guidelines recommend hysterectomy only after exhausting medical management with progestins, tranexamic acid, or endometrial ablation, as it eliminates the dysfunctional endometrium but forfeits fertility.[8][35] Adenomyosis, characterized by ectopic endometrial glands invading the myometrium, induces diffuse uterine enlargement and dysmenorrhea; hysterectomy offers curative removal of the affected organ, as partial treatments like levonorgestrel intrauterine devices provide only symptomatic palliation. Endometriosis, involving extrauterine endometrial implants causing adhesions and chronic inflammation, prompts hysterectomy in refractory deep infiltrating disease, though ovarian preservation may leave residual ovarian endometriomas risking symptom recurrence.[36][37] Pelvic organ prolapse, including uterine descent due to ligamentous laxity from childbirth trauma or collagen defects, necessitates hysterectomy when the uterus protrudes beyond the introitus (stage III-IV), often combined with vault suspension to avert enterocele formation. While uterine-sparing pessary or sacrohysteropexy exist, hysterectomy facilitates direct apical repair in postmenopausal women not desiring fertility preservation.[38][8]Malignant and Precancerous Conditions
Hysterectomy serves as the cornerstone of surgical management for endometrial cancer, particularly in stages I and II, where total hysterectomy with bilateral salpingo-oophorectomy enables both therapeutic removal and pathological staging to assess myometrial invasion and lymph node involvement.[39] For cases with cervical stromal invasion, radical hysterectomy extends resection to include parametrial tissues, upper vagina, and pelvic lymphadenectomy, improving locoregional control in early disease.[40] In advanced stages, hysterectomy may follow neoadjuvant chemotherapy if residual pelvic disease persists, though systemic therapy predominates.[41] For cervical cancer confined to the cervix (stages IA2 to IB3 per FIGO classification), radical hysterectomy with pelvic lymphadenectomy remains the standard for operable cases, removing the uterus, cervix, parametria, and upper vagina to achieve negative margins and reduce recurrence risk.[42] Simple hysterectomy suffices for microinvasive lesions (IA1 with lymphovascular invasion) or select low-risk early-stage tumors, preserving more anatomy while effectively treating disease.[43] Recent data indicate a shift toward open abdominal approaches over minimally invasive radical hysterectomy due to higher recurrence rates observed in laparoscopic or robotic series, prompting guideline updates favoring traditional surgery for optimal oncologic outcomes.[44] In epithelial ovarian cancer, hysterectomy forms part of comprehensive staging and cytoreductive surgery, routinely including total hysterectomy, bilateral salpingo-oophorectomy, and omentectomy to excise macroscopic disease and evaluate peritoneal spread, with studies confirming its role in initial management regardless of uterine involvement.[45] For germ cell tumors limited to one ovary in young patients desiring fertility preservation, unilateral salpingo-oophorectomy may suffice without hysterectomy, but bilateral involvement or advanced disease necessitates full uterine removal alongside adnexectomy.[46] Precancerous conditions warrant hysterectomy when conservative measures fail or risk of progression is high, such as in atypical endometrial hyperplasia or endometrial intraepithelial neoplasia unresponsive to progestin therapy, where total hysterectomy provides definitive cure and histopathological confirmation, averting up to 40% progression to endometrioid adenocarcinoma.[47] For high-grade cervical intraepithelial neoplasia (CIN 3), simple hysterectomy is indicated in women who have completed childbearing, especially with gland involvement or unsatisfactory colposcopy, eliminating persistent lesions that carry a 12-30% risk of invasive carcinoma if untreated.[42]Emergency and Other Indications
Emergency peripartum hysterectomy (EPH) is performed to address life-threatening obstetric hemorrhage, typically within 24 hours of delivery, when conservative measures such as uterotonics, balloon tamponade, or embolization fail.[48] The primary indications include uterine atony, which accounts for approximately 29% of cases, abnormal placentation such as placenta accreta spectrum disorders (up to 73% in some series), and uterine rupture (around 45%).[49][50] These procedures carry high maternal morbidity, with hysterectomy rates for severe postpartum hemorrhage varying from 0.2 to 2.6 per 1,000 deliveries globally, influenced by factors like cesarean delivery rates and access to blood products.[51] Cesarean hysterectomies predominate over those following vaginal delivery due to increased risks of atony and accreta in surgical births.[52] Non-obstetric emergencies requiring hysterectomy are rarer and include uncontrollable hemorrhage from ruptured ectopic pregnancies or traumatic uterine injury, though these often involve organ-preserving alternatives first.[53] In such scenarios, total abdominal hysterectomy may be lifesaving, but data emphasize rapid multidisciplinary intervention to minimize delays, as maternal mortality can reach 4-12% in EPH cohorts despite intervention.[54] Beyond emergent contexts, hysterectomy serves prophylactic roles in women with hereditary nonpolyposis colorectal cancer (Lynch syndrome), where it, combined with bilateral salpingo-oophorectomy, reduces endometrial and ovarian cancer risks by eliminating susceptible tissue.[55] This approach is recommended after childbearing for mutation carriers, given lifetime endometrial cancer risks exceeding 40% without surgery.[56] Hysterectomy is also conducted for individuals with gender dysphoria, particularly transgender males seeking alignment with male physiology, often alongside oophorectomy to halt menstrual cycles and estrogen production.[57] Guidelines from organizations like the World Professional Association for Transgender Health deem it medically necessary after hormone therapy and psychological evaluation, though long-term outcomes data remain limited, with primary diagnoses for such procedures frequently citing dysphoria rather than comorbid benign pathology.[58] Empirical evidence on causal efficacy for dysphoria resolution is derived largely from self-reported satisfaction, with potential confounders including selection bias in cohorts.[59]Surgical Techniques
Classification by Extent of Removal
Hysterectomies are classified primarily by the extent of removal of the uterus and adjacent structures, with the main types being subtotal (supracervical), total, and radical. This classification determines the surgical scope, potential preservation of anatomical function, and associated risks such as the need for ongoing cervical screening in subtotal cases. The choice depends on the underlying pathology, with subtotal and total procedures typically used for benign conditions and radical for malignancies.[1][7][60] Subtotal or supracervical hysterectomy entails excision of the uterine corpus (body) while leaving the cervix intact, often including removal of the fallopian tubes but preserving the ovaries unless separately indicated. This preserves the endocervical canal and may reduce operative time and blood loss in select cases, though evidence from randomized trials shows no significant long-term benefits in urinary, bowel, or sexual function compared to total hysterectomy, and it necessitates continued cervical cancer screening due to retained risk.[1][61][62] Total hysterectomy involves complete removal of the uterus, including the corpus and cervix, typically with closure of the vaginal cuff. It is the most common variant, performed in approximately 90% of cases for benign indications, eliminating the need for future cervical screening while addressing symptoms like heavy bleeding or fibroids without preserving potentially problematic cervical tissue. Oophorectomy or salpingectomy may accompany it but is not inherent to the uterine extent.[60][7][63] Radical hysterectomy extends beyond the uterus to include the cervix, upper one-third to half of the vagina, parametrial tissues, and often pelvic lymph nodes, classified further by systems such as Piver-Rutledge (types I-IV based on parametrial and vaginal resection depth). Reserved for cervical or endometrial cancers, it aims for oncologic clearance but carries higher risks of urinary and sexual dysfunction due to autonomic nerve disruption; type III (moderate radicality) remains standard for early-stage disease per surgical oncology guidelines.[64][63][65]Open Abdominal Hysterectomy
Open abdominal hysterectomy, also known as total abdominal hysterectomy when the cervix is removed, is a surgical procedure that removes the uterus through a large incision in the lower abdomen, allowing direct visualization and access to the pelvic cavity.[2][66] This approach is typically performed under general anesthesia and lasts 1-2 hours.[66] It is indicated primarily for benign conditions such as uterine fibroids, abnormal uterine bleeding, endometriosis, or uterine prolapse, particularly when the uterus is enlarged beyond 12 weeks' gestational size, severe adhesions from prior surgeries exist, or extrauterine pathology like adnexal masses requires exploration.[2][8] For malignant conditions, it facilitates staging and resection in cases where minimally invasive routes are infeasible or intraoperative conversion from laparoscopy occurs.[8] Contraindications include advanced cervical cancer beyond stage IB, where chemoradiation is preferred over surgery.[2] The procedure begins with a laparotomy incision, usually low transverse (Pfannenstiel) for better cosmesis or vertical midline for extensive access.[2] The peritoneal cavity is entered, adhesions lysed if present, and the bowel packed superiorly. Round ligaments are clamped, divided, and ligated bilaterally to enter the broad ligament spaces. The bladder is reflected inferiorly, exposing the uterine vessels, which are skeletonized, clamped, and ligated. If salpingo-oophorectomy is planned, ovarian vessels are divided. Cardinal and uterosacral ligaments are clamped and divided, followed by circumferential clamping of the vagina, amputation of the uterus, and closure of the vaginal cuff with absorbable sutures. The pelvis is irrigated, hemostasis confirmed, and the abdominal wall closed in layers.[2] Compared to minimally invasive alternatives like laparoscopy, open abdominal hysterectomy offers advantages in managing complex anatomy or large specimens but incurs disadvantages including longer operative times in some scenarios, increased blood loss (average 400 mL), higher rates of wound infections, and extended recovery periods of 4-6 weeks or more.[2][8] Patients typically require a 1-2 day hospital stay, with full recovery taking 6 weeks, during which heavy lifting and sexual activity are restricted to minimize dehiscence risk.[66] Specific risks include urinary tract injuries (e.g., ureteral or bladder damage in 1-2% of cases), bowel injury, vaginal cuff dehiscence, venous thromboembolism, and pelvic infections, with overall complication rates higher than vaginal or laparoscopic routes due to the open incision.[2][8] Evidence from guidelines emphasizes reserving this technique for situations where safer alternatives cannot ensure complete resection or adequate inspection.[8]Vaginal Hysterectomy
Vaginal hysterectomy involves the removal of the uterus through an incision made in the upper vagina, without requiring an abdominal incision. This approach is particularly suitable for cases involving uterine prolapse, smaller uteri (typically weighing less than 280 grams), or benign conditions such as fibroids that do not distort pelvic anatomy.[67][8] It is recommended as the preferred route for benign disease whenever feasible due to its association with reduced morbidity compared to abdominal hysterectomy.[8] The procedure begins with the patient under general or regional anesthesia, positioned in dorsal lithotomy. An incision is made in the posterior vaginal fornix to access the uterus, which is then clamped, cut, and ligated from surrounding ligaments, including the cardinal and uterosacral ligaments, while carefully dissecting the bladder from the lower uterine segment. The uterus is detached from the cervix and removed through the vaginal canal; the vaginal cuff is closed with absorbable sutures. Concomitant procedures, such as anterior/posterior colporrhaphy for prolapse repair, can be performed.[67][68] The success rate exceeds 95% in appropriately selected patients, with operative times averaging 60-90 minutes.[67] Advantages include shorter hospital stays (typically 1-2 days), reduced postoperative pain, lower infection rates, and faster recovery of 2-4 weeks compared to the 4-6 weeks or more for abdominal hysterectomy. Randomized trials demonstrate that vaginal hysterectomy results in less blood loss (mean 200-300 mL), fewer febrile episodes, and lower costs, with no difference in major complication rates when compared to laparoscopic approaches in select cases.[67][69][70] In a randomized comparison with abdominal hysterectomy for enlarged uteri, vaginal approach shortened hospital stay by one day and reduced overall costs without increasing complications.[70] Contraindications include large or adherent uteri, significant adhesions from prior surgeries, suspected malignancy requiring staging, or narrow vaginal access that limits visualization. Relative limitations involve difficulty with adnexal removal, succeeding in only 65-97.5% of cases.[8][67] Complications, occurring in 5-10% of cases, encompass urinary tract injury (1-2%, primarily bladder), vaginal cuff dehiscence (less than 1%), and hemorrhage, though rates are lower than abdominal methods due to avoidance of laparotomy. Long-term risks include vaginal shortening or dyspareunia in 5-10% of patients. Evidence from meta-analyses confirms vaginal hysterectomy's superior perioperative outcomes, including reduced hospital stay and quicker return to work, supporting its prioritization over more invasive routes.[67][71][72]Minimally Invasive Approaches
Minimally invasive approaches to hysterectomy utilize laparoscopic techniques, involving small abdominal incisions for inserting a camera and instruments, avoiding large open incisions (sometimes using laser). These methods include total laparoscopic hysterectomy (TLH), where the entire uterus is removed and extracted laparoscopically or vaginally; laparoscopic-assisted vaginal hysterectomy (LAVH), combining laparoscopy for initial dissection with vaginal removal; and laparoscopic supracervical hysterectomy (LSH), preserving the cervix.[73][8] In TLH, ports are placed in the abdomen, the uterus is detached from surrounding structures using laparoscopic tools, vessels are sealed, and the specimen is morcellated or extracted via the vagina or mini-incision. LAVH facilitates visualization of adhesions or large uteri, transitioning to vaginal completion, while LSH reduces operative time by avoiding full uterine removal. These approaches enable same-day or short-stay procedures for benign indications, with studies reporting hospital stays of 1-2 days compared to 3-5 for abdominal hysterectomy. Laparoscopic or vaginal methods typically allow recovery in 2-4 weeks, shorter than the 4-6 weeks or more for traditional open abdominal hysterectomy. These advantages are particularly beneficial for elderly patients, as videolaparoscopic hysterectomy is less invasive, offering quicker recovery and lower risk of complications compared to traditional methods.[29][74][75][76] For benign conditions, evidence from randomized controlled trials and meta-analyses shows laparoscopic methods yield less blood loss (mean 100-200 mL vs. 300-500 mL in open), reduced postoperative pain, and faster return to activity (2-4 weeks vs. 4-6 weeks). Operative times may be longer (120-180 minutes vs. 90-120 for open) but complication rates are comparable or lower, including fewer wound infections. Conversion to open occurs in 1-5% of cases due to adhesions or bleeding.[77][78][79] In early-stage cervical cancer, randomized trials indicate minimally invasive radical hysterectomy associates with higher recurrence rates (e.g., 4-year disease-free survival 82% vs. 96% for open) and possibly worse overall survival, attributed to potential tumor spillage or insufflation effects, leading to recommendations favoring open approaches for malignancy. For endometrial cancer, laparoscopic staging and hysterectomy show equivalent oncologic outcomes to open with better perioperative recovery.[80][81][82] Risks specific to these techniques include port-site hernias (0.5-2%), urinary tract injuries (1-2%, similar to open), and vaginal cuff dehiscence (1-4%, higher with energy devices). Patient selection is critical; obesity, prior surgeries, or large fibroids increase technical difficulty. Long-term data confirm reduced adhesion formation compared to open surgery.[73][75]Robotic-Assisted and Emerging Techniques
Robotic-assisted hysterectomy employs systems such as the da Vinci Surgical System, which provides surgeons with three-dimensional visualization, tremor filtration, and articulated instruments for enhanced precision during minimally invasive procedures.[83] First approved by the FDA for gynecologic use in 2005, this approach has been applied to both benign and malignant indications, facilitating procedures like total hysterectomy with salpingo-oophorectomy through small abdominal incisions.[84] Operative times typically range from 120 to 240 minutes, longer than conventional laparoscopy due to setup and docking requirements, but studies report reduced blood loss (median 50-100 mL) and hospital stays (1-2 days) compared to open surgery.[85] [86] Comparative analyses indicate no significant differences in major complications, readmission rates, or long-term outcomes between robotic-assisted and conventional laparoscopic hysterectomy for benign conditions, with robotic methods incurring higher costs (up to $2,000-3,000 more per case) without proportional benefits in conversion rates or postoperative pain.[87] [88] A 2024 systematic review of randomized trials confirmed equivalent surgical and patient-reported outcomes, attributing robotic adoption partly to shorter learning curves for complex cases, such as those involving obesity or adhesions, where conversion to open surgery drops below 5%.[87] [89] For malignant indications, robotic approaches yield similar oncologic efficacy to laparoscopy, with blood loss under 200 mL and lymph node yields exceeding 15 per procedure, though evidence from high-quality trials remains limited by surgeon experience variability.[90] Emerging techniques build on minimally invasive principles, including vaginal natural orifice transluminal endoscopic surgery (vNOTES), introduced clinically around 2012 and gaining traction post-2020 for its scarless access via the vaginal route.[91] vNOTES enables hysterectomy with uterine weights up to 500 g, reporting operative times of 60-90 minutes, negligible blood loss (<50 mL), and same-day discharge in over 80% of cases, with complication rates under 2% in initial series; it avoids abdominal trocars, reducing port-site hernias.[92] Single-port robotic systems, such as the da Vinci SP approved in 2018, further advance this by consolidating instruments into one incision, achieving low pain scores (VAS <3) and complication rates below 1% in early 2024 studies of benign hysterectomies.[93] [94] Other innovations like mini-laparoscopy (using 2-3 mm ports) show promise for reduced tissue trauma but lack large-scale outcome data beyond pilot feasibility trials.[95] These methods prioritize outpatient feasibility and cosmesis, though long-term comparative effectiveness trials are ongoing to validate advantages over established laparoscopy.[96]Risks and Complications
Perioperative and Immediate Risks
Perioperative risks of hysterectomy encompass intraoperative events such as hemorrhage and organ injury, as well as immediate postoperative issues including infection and thromboembolism.[97] Overall 30-day postoperative complication rates range from 4.4% to 6.2% across surgical approaches, with abdominal hysterectomy associated with higher composite rates (10.3%) compared to laparoscopic (5.3%) or vaginal (6.8%).[98] [99] Major complications, defined as those requiring intervention or prolonging hospital stay, occur in approximately 4.4% of laparoscopic cases and 4.9% of abdominal procedures.[99] Intraoperative bleeding is a primary concern, contributing to up to half of gynecological postoperative complications, though severe hemorrhage remains rare with median rates below 1%.[100] [97] Organ injuries, particularly to the genitourinary tract (e.g., bladder or ureter) or gastrointestinal tract (e.g., bowel), occur in 0.1-1% of cases, with ureteral injury rates around 0.02-1.4% depending on approach and surgeon experience.[97] These injuries often arise from dissection near pelvic structures and may necessitate immediate repair or conversion to open surgery.[101] Immediate postoperative anemia, resulting from surgical blood loss, can cause symptoms such as dyspnea (shortness of breath), fatigue, and weakness due to reduced oxygen delivery to tissues.[102] Infections, including wound, urinary tract, or pelvic abscesses, affect 4-25% of patients, with higher rates (6-25%) in abdominal hysterectomy versus 4-10% in vaginal approaches.[103] Urinary tract infections are the most frequent, occurring in up to 3.9% of cases.[104] Venous thromboembolism, including deep vein thrombosis or pulmonary embolism, represents another acute risk, with incidence influenced by prophylaxis protocols and patient factors like obesity.[97] Anesthesia-related complications, such as respiratory issues or cardiovascular events, are less common but elevated in patients with comorbidities.[105] Risk mitigation involves preoperative optimization, antibiotic prophylaxis, and thromboprophylaxis, which reduce infection and clot rates per guidelines from bodies like the American College of Obstetricians and Gynecologists (ACOG). ACOG strongly recommends antibiotic prophylaxis for all hysterectomies to lower infection risk, with regimens varying by procedure type (abdominal, vaginal, laparoscopic), patient factors (obesity, diabetes), or institutional protocols, administered preoperatively, intraoperatively, or postoperatively.[103][106] Patient-specific factors, including body mass index and surgical route, modulate these risks, with minimally invasive techniques generally lowering overall perioperative morbidity.[107]Long-Term Health Impacts
Hysterectomy, particularly when accompanied by bilateral salpingo-oophorectomy (BSO), is associated with elevated risks of cardiovascular disease (CVD), including coronary heart disease (CHD) and stroke, even in cases of ovarian conservation. A 2024 meta-analysis of observational studies found that hysterectomy increased the relative risk of CVD by approximately 1.17 (95% CI: 1.09-1.26), CHD by 1.20 (95% CI: 1.06-1.35), and stroke by 1.23 (95% CI: 1.08-1.40), though it did not significantly elevate all-cause or cardiovascular mortality.[108] These risks may stem from disruptions to ovarian blood supply, accelerated ovarian aging, or inflammatory responses, with hazard ratios for CVD reaching 1.09 after hysterectomy alone and 1.19 with BSO.[109] Hysterectomy is associated with an increased risk of type 2 diabetes, particularly when combined with bilateral oophorectomy, likely due to hormonal changes and premature menopause. Multiple studies confirm this association, with risks elevated by 13-40% depending on ovarian conservation.[110][111] There is no evidence linking hysterectomy to an increased risk of type 1 diabetes, an autoimmune condition typically diagnosed earlier in life. Skeletal health deteriorates post-hysterectomy, with heightened incidence of osteoporosis and fractures independent of oophorectomy status. A systematic review indicated that hysterectomy elevates fracture risk (OR 1.56-2.37 across studies) and reduces bone mineral density (BMD) at lumbar spine and hip sites, attributed to potential alterations in pelvic vascularization or hormonal microenvironments despite preserved ovaries.[112] Population-based cohorts report a 1.5- to 2-fold increase in osteoporosis diagnosis and fracture events among hysterectomized women followed for over a decade, with premenopausal procedures showing greater BMD loss (up to 5-10% at 2 years) compared to intact controls.[113][114] Pelvic floor disorders, including urinary incontinence and prolapse, persist or emerge long-term after hysterectomy due to denervation and structural support loss. Hysterectomy correlates with a 1.5- to 2-fold higher likelihood of stress urinary incontinence within 10 years, escalating to pelvic organ prolapse (OR 1.56; 95% CI: 1.35-1.78) beyond that duration in meta-analyzed cohorts.[115] Vaginal and abdominal approaches show comparable risks, with prospective data confirming sustained pelvic symptoms in 20-30% of women at 5-10 years post-surgery.[116] Sexual function outcomes vary, with systematic reviews reporting no overall decline in desire, arousal, or satisfaction for most women, though subsets experience dyspareunia or lubrication issues from vaginal shortening or nerve disruption. A 2023 meta-analysis of 22 studies (n>5,000) found neutral effects on composite sexual function scores (SMD 0.05; 95% CI: -0.12 to 0.22) across surgical routes, yet qualitative data highlight persistent dysfunction in 10-20% due to psychological factors or anatomical changes.[117] Oncologic profiles shift post-hysterectomy with BSO, reducing ovarian and breast cancer incidence (HR 0.4-0.7) but elevating colorectal cancer risk (HR 1.4-1.8), potentially from hormonal imbalances or microbiome alterations.[118] All-cause mortality remains comparable to non-hysterectomized peers when ovaries are conserved before age 50, though BSO before natural menopause shortens lifespan by 1-2 years via CVD and cancer pathways.[119] Comprehensive reviews emphasize individualized risk assessment, as procedure extent modulates these effects.[120]Psychological and Quality-of-Life Effects
Hysterectomy for benign conditions is associated with improvements in symptoms of depression and anxiety, as well as overall quality of life (QoL), in many patients, particularly when performed to alleviate severe gynecological symptoms such as heavy bleeding or chronic pain from fibroids.[121] A meta-analysis of 22 studies involving 5,978 participants found significant reductions in depression scores post-hysterectomy, alongside enhanced vitality, reduced discomfort, and better sexual activity metrics both short- and long-term.[121] These benefits are attributed to the resolution of underlying pathology, with minimally invasive approaches yielding superior short-term QoL gains compared to open abdominal methods, including better physical functioning and bodily pain control.[122] However, evidence indicates elevated long-term risks of de novo depression and anxiety following hysterectomy, even with ovarian conservation, with absolute risk increases of 6.6% for depression and 4.7% for anxiety over 30 years, particularly among women undergoing the procedure before age 45.[123][124] This risk escalates with bilateral oophorectomy, which induces surgical menopause and hormonal disruptions contributing to mood disorders, as opposed to natural menopause.[125] Some studies report higher postoperative depressive symptoms, such as diminished interest or persistent low mood, especially in younger patients or those without ovarian preservation.[126] Decisional regret occurs in a minority of cases but is more prevalent among younger women, with rates of surgical regret reaching 32.5% in those aged 30 or younger versus 9.1% in older groups, often tied to fertility loss.[127] Overall regret for the procedure itself remains low at approximately 2.8%, with over 95% of patients affirming their decision when adequately counseled preoperatively; however, 21-43% express persistent regret over infertility three years post-surgery.[128][129] Fertility-related regret may intensify initially but often diminishes over time for most, though it persists in a subset, underscoring the need for thorough preoperative discussions on reproductive implications.[130] Sexual function and body image exhibit mixed outcomes, with some meta-analyses noting preserved or improved libido and arousal due to pain relief, while others highlight potential declines in genital sensation from nerve disruption, leading to dyspareunia or reduced satisfaction in up to certain subgroups.[117] Psychological interventions post-surgery can mitigate pelvic floor-related distress and enhance coping, reducing anxiety prevalence from baseline levels.[131] Conflicting findings across studies may reflect selection biases in patient cohorts or variations in surgical extent, with pre-existing mental health conditions predicting poorer trajectories.[132] Long-term QoL improvements are more consistent in postmenopausal women, where anxiety and femininity perceptions tend to stabilize or enhance post-procedure.[133]Alternatives
Pharmacological and Conservative Options
Pharmacological interventions for uterine fibroids primarily target hormonal pathways to reduce symptoms such as heavy bleeding and pelvic pain, though evidence for long-term efficacy remains limited. GnRH agonists and antagonists, including elagolix, suppress ovarian estrogen production, leading to fibroid shrinkage of up to 50% in volume within three to six months, but their use is typically short-term (up to six months) due to side effects like bone density loss and menopausal symptoms.[134] [135] Selective progesterone receptor modulators (SPRMs), such as ulipristal acetate, have demonstrated reductions in fibroid volume by 20-40% and improved quality of life in randomized trials, yet concerns over rare liver toxicity have prompted regulatory restrictions in some regions.[136] Progestins, including oral or injectable forms, offer symptom relief by stabilizing the endometrium but do not consistently reduce fibroid size.[135] For heavy menstrual bleeding (menorrhagia), often a primary indication for hysterectomy consideration, first-line medical options include tranexamic acid, an antifibrinolytic that reduces blood loss by 30-60% during menses with minimal side effects in short-term use, supported by Cochrane reviews as superior to placebo.[137] [138] Levonorgestrel-releasing intrauterine systems (LNG-IUS) achieve amenorrhea in up to 50% of users after one year by providing local progestin, outperforming oral progestins in reducing bleeding volume per Cochrane analyses, though expulsion rates reach 5-10%.[139] Combined oral contraceptives and nonsteroidal anti-inflammatory drugs (NSAIDs) provide moderate relief (20-40% blood loss reduction) but are less effective for structural causes like fibroids.[140] In endometriosis and adenomyosis, where hysterectomy may be proposed for refractory pain or bleeding, hormonal suppression remains the cornerstone. NSAIDs alleviate dysmenorrhea acutely, while GnRH analogs reduce lesion size and pain scores by 40-70% in trials, albeit with hypoestrogenic risks necessitating add-back therapy.[141] [142] Progestins, particularly LNG-IUS for adenomyosis, yield sustained symptom improvement in 60-80% of cases over 12-24 months, with prospective studies favoring it over oral alternatives for bleeding control.[143] [144] Aromatase inhibitors show preliminary promise for pain reduction in adenomyosis but lack robust safety data for extended use.[145] Conservative management emphasizes symptom monitoring and lifestyle modifications when pathology is mild or asymptomatic, avoiding intervention unless quality of life is impaired. Regular exercise and weight management correlate with reduced bleeding severity, though causal evidence is observational and confounded by selection bias.[140] Watchful waiting is appropriate for small fibroids under 5 cm without symptoms, with serial ultrasound monitoring every 6-12 months to track growth rates averaging 1-2 mm per year.[146] Non-pharmacologic adjuncts like acupuncture yield inconsistent pain relief in small trials for endometriosis, insufficient for recommendation over evidence-based therapies.[147] Overall, these options delay or obviate surgery in 40-70% of cases depending on condition severity, per systematic reviews, but failure rates necessitate timely reassessment.[134]Uterine-Preserving Surgical Interventions
Uterine-preserving surgical interventions address symptomatic benign uterine conditions, including leiomyomas (fibroids), abnormal uterine bleeding, and pelvic organ prolapse, without requiring hysterectomy. These procedures prioritize symptom relief, fertility preservation, and uterine retention, often yielding comparable short-term outcomes to hysterectomy but with potential for recurrence or reintervention. Selection depends on patient age, desire for future pregnancy, fibroid characteristics, and prolapse severity, with evidence from randomized trials supporting their efficacy in appropriately selected cases.[148][149] Myomectomy, the excision of intramural or subserosal fibroids while conserving the uterus, serves as the primary surgical option for women with symptomatic leiomyomas seeking fertility. Performed via open abdominal, laparoscopic, or robotic approaches, laparoscopic myomectomy reduces blood loss and postoperative pain compared to abdominal methods, with operative times averaging 100-150 minutes for multiple fibroids. Health-related quality of life improves significantly post-procedure, though scores may lag slightly behind those after hysterectomy at one year; long-term fibroid recurrence approaches 22% over 11 years, necessitating reintervention in about 11% of cases. Myomectomy outperforms uterine artery embolization in fibroid-related quality of life at two years and is preferred for pregnancy-desiring patients due to lower miscarriage risks.[150][33][148][151] Endometrial ablation or resection targets heavy menstrual bleeding by destroying the endometrial lining via thermal, radiofrequency, or electrosurgical methods, often under hysteroscopic guidance. This outpatient procedure achieves amenorrhea or oligomenorrhea in 40-60% of patients, with satisfaction rates exceeding 80% at one year, and offers shorter recovery (days versus weeks) and fewer complications than hysterectomy. However, up to 25% of women may require hysterectomy within five years due to treatment failure or persistent symptoms, particularly in those with large uteri or adenomyosis; hysterectomy provides superior long-term bleeding control but at higher perioperative risk.[152][153][154] Uterine artery embolization (UAE), a catheter-based intervention, occludes fibroid blood supply using embolic particles, inducing ischemia and volume reduction of 30-50% within six months. Symptom improvement occurs in over 85% of patients, with sustained quality-of-life gains observed in observational studies up to five years; technical success exceeds 95%, though post-embolization syndrome affects most patients transiently. UAE yields lower pregnancy rates and higher miscarriage risks than myomectomy, limiting its use in fertility-focused cases, and meta-analyses confirm comparable efficacy to myomectomy for symptom control but with fewer major complications.[148][155][156] For uterine prolapse, preservation techniques like sacrohysteropexy or uterosacral ligament suspension use mesh or native tissue to restore anatomy laparoscopically or vaginally, avoiding hysterectomy. A 2024 meta-analysis reports lower recurrence (10-15% versus 20% with hysterectomy) and complication rates, with faster return to normal activity; uterine preservation maintains sexual function and avoids vaginal shortening risks associated with colpocleisis or hysterectomy. These approaches suit women rejecting hysterectomy for cultural, fertility, or menopausal status reasons, though long-term durability data remain limited beyond five years.[149][157][158]Management of Specific Conditions
Uterine FibroidsHysterectomy serves as a definitive treatment for symptomatic uterine leiomyomas when conservative options such as medication or myomectomy fail to alleviate heavy menstrual bleeding, pain, or pressure symptoms.[8][159] Symptomatic fibroids account for approximately 51.4% of benign hysterectomies, with the procedure eliminating fibroid-related symptoms in nearly all cases due to complete removal of the uterus.[8] The choice of approach—vaginal, laparoscopic, or abdominal—depends on fibroid size, number, and uterine mobility, with vaginal hysterectomy preferred for smaller uteri to minimize complications and recovery time.[32] Abnormal Uterine Bleeding
For nonpregnant reproductive-aged women with acute or chronic abnormal uterine bleeding unresponsive to hormonal or nonhormonal medical therapies, hysterectomy provides definitive control by eliminating the source of bleeding.[160] This indication represents about 41.7% of benign hysterectomies, often linked to ovulatory dysfunction or endometrial hyperplasia without atypia.[8] In cases with atypical hyperplasia, hysterectomy is prioritized to prevent progression to endometrial cancer, with total hysterectomy ensuring removal of at-risk tissue.[3] Adenomyosis
Hysterectomy is the definitive management for adenomyosis in women past childbearing age who experience persistent heavy bleeding, dysmenorrhea, or pelvic pain despite medical treatments like NSAIDs or hormonal therapies.[144] Adenomyosis is identified in up to 42% of hysterectomies performed for abnormal uterine bleeding, frequently coexisting with fibroids and contributing to diffuse uterine enlargement.[161] Total hysterectomy addresses the condition's intrauterine pathology comprehensively, though conservative surgical alternatives like excision may be considered in select cases to preserve fertility.[162] Endometriosis
In severe endometriosis refractory to excision, medical suppression, or conservative surgery, hysterectomy—often with bilateral salpingo-oophorectomy—relieves symptoms such as chronic pain and infertility by removing estrogen-dependent lesions, though extrauterine disease may persist.[163] Guidelines recommend this approach for women not seeking fertility preservation, with ovarian conservation possible if ovaries are unaffected to avoid surgical menopause.[164] Total hysterectomy outperforms subtotal in reducing recurrence for endometriosis-associated adenomyosis, based on comparative outcomes in symptom resolution.[165] Pelvic Organ Prolapse
Hysterectomy combined with vaginal vault suspension is indicated for symptomatic uterine prolapse, particularly when conservative measures like pessaries fail, to restore pelvic anatomy and alleviate bulge or pressure sensations.[166] Vaginal hysterectomy facilitates concurrent prolapse repair via uterosacral or sacrospinous ligament fixation, offering lower morbidity than abdominal routes for stage II or higher prolapse.[167] Uterine-preserving procedures like hysteropexy may suffice if fertility or uterine retention is desired, but hysterectomy is standard when coexisting conditions such as bleeding warrant uterine removal.[168] Gynecologic Malignancies
For early-stage endometrial cancer, standard management involves total hysterectomy with bilateral salpingo-oophorectomy and lymph node staging, achieving cure rates exceeding 90% in low-risk cases through minimally invasive approaches when feasible.[169] In stage IA cervical cancer, radical hysterectomy removes the uterus, cervix, and parametria, serving as primary therapy with adjuvant radiation for high-risk features.[170] These oncologic hysterectomies prioritize complete resection margins over benign techniques, with preoperative imaging guiding extent of surgery.[171]