Recent from talks
All channels
Be the first to start a discussion here.
Be the first to start a discussion here.
Be the first to start a discussion here.
Be the first to start a discussion here.
Welcome to the community hub built to collect knowledge and have discussions related to Cymene.
Nothing was collected or created yet.
Cymene
View on Wikipediafrom Wikipedia
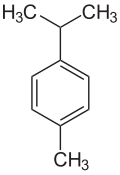
Cymene describes organic compounds with the formula CH3C6H4CH(CH3)2. Three isomers exist: 1,2- 1,3-, and 1,4-. All are colorless liquids, immiscible in water, with similar boiling points. They are classified are aromatic hydrocarbons. The bearing two substituents: an isopropyl (CH(CH3)2) group and a methyl group.[1]
| Cymenes | |||
| Name | o-Cymene | m-Cymene | p-Cymene |
|---|---|---|---|
| Structural formula |  |
 |
|
| CAS number | 527-84-4 | 535-77-3 | 99-87-6 |
| melting point (°C) | −71.54 | −63.75 | −67.94 |
| boiling point (°C) | 178.15 | 175.05 | 177.10 |
Production and reactions
[edit]m- and p-Cymene are prepared by alkylation of toluene with propylene:
- CH3C6H5 + 2 CH3CH=CH2 → CH3C6H4CH(CH3)2
These alkylations are catalyzed by various Lewis acids, such as aluminium trichloride.
m- and p-Cymene are mainly of interest as precursors to the respective cresols, which exploits the Hock rearrangements.[1]
References
[edit]- ^ a b Schmidt, Roland; Griesbaum, Karl; Behr, Arno; Biedenkapp, Dieter; Voges, Heinz-Werner; Garbe, Dorothea; Paetz, Christian; Collin, Gerd; Mayer, Dieter; Höke, Hartmut (2014). "Hydrocarbons". Ullmann's Encyclopedia of Industrial Chemistry. pp. 1–74. doi:10.1002/14356007.a13_227.pub3. ISBN 9783527306732.
