Recent from talks
Nothing was collected or created yet.
In organic chemistry, enols are a type of functional group or intermediate in organic chemistry containing a group with the formula C=C(OH) (R = many substituents). The term enol is an abbreviation of alkenol, a portmanteau deriving from "-ene"/"alkene" and the "-ol". Many kinds of enols are known.[1]
Keto–enol tautomerism refers to a chemical equilibrium between a "keto" form (a carbonyl, named for the common ketone case) and an enol. The interconversion of the two forms involves the transfer of an alpha hydrogen atom and the reorganisation of bonding electrons. The keto and enol forms are tautomers of each other.[2]
Enolization
[edit]Organic esters, ketones, and aldehydes with an α-hydrogen (C−H bond adjacent to the carbonyl group) often form enols. The reaction involves migration of a proton (H) from carbon to oxygen:[1]
- RC(=O)CHR′R′′ ⇌ RC(OH)=CR′R′′
In the case of ketones, the conversion is called a keto-enol tautomerism, although this name is often more generally applied to all such tautomerizations. Usually the equilibrium constant is so small that the enol is undetectable spectroscopically.
In some compounds with two (or more) carbonyls, the enol form becomes dominant. The behavior of 2,4-pentanedione illustrates this effect:[3]
| carbonyl | enol | Kenolization |
|---|---|---|
| Acetaldehyde CH3CHO |
CH2=CHOH | 5.8×10−7 |
| Acetone CH3C(O)CH3 |
CH3C(OH)=CH2 | 5.12×10−7 |
| Methyl acetate CH3CO2CH3 |
CH2=CH(OH)OCH3 | 4×10−20 |
| Acetophenone C6H5C(O)CH3 |
C6H5C(OH)=CH2 | 1×10−8 |
| Acetylacetone CH3C(O)CH2C(O)CH3 |
CH3C(O)CH=C(OH)CH3 | 0.27 |
| Trifluoroacetylacetone CH3C(O)CH2C(O)CF3 |
CH3C(O)CH=C(OH)CF3 | 32 |
| Hexafluoroacetylacetone CF3C(O)CH2C(O)CF3 |
CF3C(O)CH=C(OH)CF3 | ~104 |
| Cyclohexa-2,4-dienone | Phenol C6H5OH |
>1012 |
Enols are derivatives of vinyl alcohol, with a C=C−OH connectivity. Deprotonation of organic carbonyls gives the enolate anion, which are a strong nucleophile. A classic example for favoring the keto form can be seen in the equilibrium between vinyl alcohol and acetaldehyde (K = [enol]/[keto] ≈ 3×10−7). In 1,3-diketones, such as acetylacetone (2,4-pentanedione), the enol form is more favored.
The acid-catalyzed conversion of an enol to the keto form proceeds by proton transfer from O to carbon. The process does not occur intramolecularly, but requires participation of solvent or other mediators.
Stereochemistry of ketonization
[edit]If R1 and R2 (note equation at top of page) are different substituents, there is a new stereocenter formed at the alpha position when an enol converts to its keto form. Depending on the nature of the three R groups, the resulting products in this situation would be diastereomers or enantiomers.[citation needed]
Enediols
[edit]Enediols are alkenes with a hydroxyl group on each carbon of the C=C double bond. Normally such compounds are disfavored components in equilibria with acyloins. One special case is catechol, where the C=C subunit is part of an aromatic ring. In some other cases however, enediols are stabilized by flanking carbonyl groups. These stabilized enediols are called reductones. Such species are important in glycochemistry, e.g., the Lobry de Bruyn–Van Ekenstein transformation.[5]

Keto-enediol tautomerizations. Enediol in the center; acyloin isomers at left and right. Ex. is hydroxyacetone, shown at right.

Conversion of ascorbic acid (vitamin C) to an enolate. Enediol at left, enolate at right, showing movement of electron pairs resulting in deprotonation of the stable parent enediol. A distinct, more complex chemical system, exhibiting the characteristic of vinylogy.
Ribulose-1,5-bisphosphate is a key substrate in the Calvin cycle of photosynthesis. In the Calvin cycle, the ribulose equilibrates with the enediol, which then binds carbon dioxide. The same enediol is also susceptible to attack by oxygen (O2) in the (undesirable) process called photorespiration.

Keto-enediol equilibrium for ribulose-1,5-bisphosphate.
Phenols
[edit]Phenols represent a kind of enol. For some phenols and related compounds, the keto tautomer plays an important role. Many of the reactions of resorcinol involve the keto tautomer, for example. Naphthalene-1,4-diol exists in observable equilibrium with the diketone tetrahydronaphthalene-1,4-dione.[6]
Biochemistry
[edit]Keto–enol tautomerism is important in several areas of biochemistry.[citation needed]
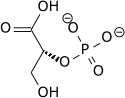
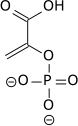
The high phosphate-transfer potential of phosphoenolpyruvate results from the fact that the phosphorylated compound is "trapped" in the less thermodynamically favorable enol form, whereas after dephosphorylation it can assume the keto form.[citation needed]
The enzyme enolase catalyzes the dehydration of 2-phosphoglyceric acid to the enol phosphate ester. Metabolism of PEP to pyruvic acid by pyruvate kinase (PK) generates adenosine triphosphate (ATP) via substrate-level phosphorylation.[7]
|
|

| ||||
| H2O | ADP | ATP | ||||

|

| |||||
| H2O | ||||||
Reactivity
[edit]Addition of electrophiles
[edit]The terminus of the double bond in enols is nucleophilic. Its reactions with electrophilic organic compounds is important in biochemistry as well as synthetic organic chemistry. In the former area, the fixation of carbon dioxide involves addition of CO2 to an enol.[citation needed]
Deprotonation: enolates
[edit]Deprotonation of enolizable ketones, aldehydes, and esters gives enolates.[8][9] Enolates can be trapped by the addition of electrophiles at oxygen. Silylation gives silyl enol ether.[10] Acylation gives esters such as vinyl acetate.[11]
Stable enols
[edit]In general, enols are less stable than their keto equivalents because of the favorability of the C=O double bond over C=C double bond. However, enols can be stabilized kinetically or thermodynamically.[citation needed]
Some enols are sufficiently stabilized kinetically so that they can be characterized.[citation needed]

Delocalization can stabilize the enol tautomer. Thus, very stable enols are phenols.[13] Another stabilizing factor in 1,3-dicarbonyls is intramolecular hydrogen bonding.[14] Both of these factors influence the enol-dione equilibrium in acetylacetone.
See also
[edit]- Alkenal
- Enolase
- Ketone
- Ynol
- Geminal diol, another form of ketones and aldehydes in water solutions
- Regioselectivity
References
[edit]- ^ a b Smith MB, March J (2001). Advanced Organic Chemistry (5th ed.). New York: Wiley Interscience. pp. 1218–1223. ISBN 0-471-58589-0.
- ^ Clayden, Jonathan; Greeves, Nick; Warren, Stuart (2012). Organic chemistry (2nd ed.). New York: Oxford University Press. pp. 450–451. ISBN 978-0-19-927029-3.
- ^ Manbeck, Kimberly A.; Boaz, Nicholas C.; Bair, Nathaniel C.; Sanders, Allix M. S.; Marsh, Anderson L. (2011). "Substituent Effects on Keto–Enol Equilibria Using NMR Spectroscopy". J. Chem. Educ. 88 (10): 1444–1445. Bibcode:2011JChEd..88.1444M. doi:10.1021/ed1010932.
- ^ Guthrie, J. Peter; Povar, Igor (2013). "Equilibrium constants for enolization in solution by computation alone". Journal of Physical Organic Chemistry. 26 (12): 1077–1083. doi:10.1002/poc.3168.
- ^ Schank, Kurt (1972). "Reductones". Synthesis. 1972 (4): 176–90. doi:10.1055/s-1972-21845. S2CID 260331550.
- ^ Kündig, E. Peter; Enríquez García, Alvaro; Lomberget, Thierry; Bernardinelli, Gérald (2006). "Rediscovery, Isolation, and Asymmetric Reduction of 1,2,3,4-Tetrahydronaphthalene-1,4-dione and Studies of Its [Cr(CO)3] Complex". Angewandte Chemie International Edition. 45 (1): 98–101. doi:10.1002/anie.200502588. PMID 16304647.
- ^ Berg, Jeremy M.; Tymoczko, Stryer (2002). Biochemistry (5th ed.). New York: W.H. Freeman and Company. ISBN 0-7167-3051-0.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, ISBN 978-0-471-72091-1
- ^ Manfred Braun (2015). Modern Enolate Chemistry: From Preparation to Applications in Asymmetric Synthesis. Wiley-VCH. doi:10.1002/9783527671069. ISBN 9783527671069.
- ^ Mukaiyama, T.; Kobayashi, S. Org. React. 1994, 46, 1. doi:10.1002/0471264180.or046.01
- ^ G. Roscher (2007). "Vinyl Esters". Ullmann's Encyclopedia of Chemical Technology. Weinheim: Wiley-VCH. doi:10.1002/14356007.a27_419. ISBN 978-3527306732. S2CID 241676899.
- ^ "Stable simple enols". Journal of the American Chemical Society. 1989. doi:10.1021/ja00203a019.
- ^ Clayden, Jonathan (2012). Organic Chemistry. Oxford University Press. pp. 456–459.
- ^ Zhou, Yu-Qiang; Wang, Nai-Xing; Xing, Yalan; Wang, Yan-Jing; Hong, Xiao-Wei; Zhang, Jia-Xiang; Chen, Dong-Dong; Geng, Jing-Bo; Dang, Yanfeng; Wang, Zhi-Xiang (2013-01-14). "Stable acyclic aliphatic solid enols: synthesis, characterization, X-ray structure analysis and calculations". Scientific Reports. 3 (1): 1058. Bibcode:2013NatSR...3E1058Z. doi:10.1038/srep01058. ISSN 2045-2322. PMC 3544012. PMID 23320139.
External links
[edit]Definition and Structure
General Formula and Nomenclature
An enol is an organic compound featuring a hydroxyl group (-OH) directly attached to a carbon atom involved in a carbon-carbon double bond, denoted as C=C-OH. This functional group distinguishes enols as tautomers of carbonyl compounds, particularly aldehydes and ketones, in the process known as keto-enol tautomerism.[1][8] The general molecular formula for enols derived from aldehydes is , where R represents a hydrogen atom or an alkyl substituent, while for those from ketones, it is , with R and R' being alkyl groups or hydrogen. In this structure, the enol moiety consists of planar sp²-hybridized carbon atoms: the double-bonded carbons and the carbon bearing the hydroxyl group adopt trigonal planar geometry, facilitating conjugation and resonance effects.[9][10] Under IUPAC nomenclature, the parent structure for the simplest enol, (also known as vinyl alcohol), is ethenol. More complex enols are named as alkenols, selecting the longest carbon chain that includes both the double bond and the hydroxyl group, with numbering starting from the carbon attached to the -OH to assign the lowest locants to the functional groups; for example, the enol form of acetone is prop-1-en-2-ol.[11][12] The term "enol" originated as a portmanteau of "ene" (from alkene) and "ol" (from alcohol), coined by Julius Wilhelm Brühl in 1894 in the context of tautomerism studies. Ludwig Knorr conducted pioneering investigations and isolated stable enol forms of β-dicarbonyl compounds in the 1880s and 1890s.[1][13][14]Relation to Keto-Enol Tautomerism
Keto-enol tautomerism refers to the reversible interconversion between a keto form, featuring a carbonyl group (C=O) and an alpha-hydrogen on an adjacent carbon, and an enol form, where a proton shifts from the alpha-carbon to the oxygen atom, resulting in a carbon-carbon double bond and a hydroxyl group.[2] This process is a classic example of tautomerism in carbonyl compounds such as aldehydes and ketones, driven by the migration of a hydrogen atom in a 1,3-position relative to the carbonyl.[15] The general equilibrium can be represented as: where the keto form predominates under typical conditions.[2] In most cases, enols represent the minor tautomer due to the greater thermodynamic stability of the keto form, which benefits from stronger C-O bond strength compared to the C=C and O-H bonds in the enol.[2] The equilibrium constant for this tautomerism typically favors the keto side by orders of magnitude for simple carbonyls, reflecting the lower energy of the carbonyl structure.[15] Spectroscopic techniques, particularly nuclear magnetic resonance (NMR), provide evidence for the presence of both tautomers by distinguishing their proton environments. Enol protons, specifically the hydroxyl group attached to the vinylic carbon (C=C-OH), exhibit characteristic chemical shifts in the range of 15-17 ppm in H NMR spectra, appearing downfield due to hydrogen bonding and the sp hybridization.[16] This deshielding contrasts with the alpha-protons in the keto form, which resonate around 2-3 ppm, allowing quantification of tautomer ratios through integration of peak areas.[17] A representative example is acetone, where the keto form (CHCOCH) vastly predominates over the enol (CH=C(OH)CH), with the enol content at equilibrium estimated at approximately (or 2.4 × 10^{-7}%) in the vapor phase at ambient temperature.[18] This low enol fraction underscores the instability of simple enols relative to their keto counterparts, though the tautomerism plays a crucial role in reactivity pathways.[19]Formation and Equilibrium
Enolization Mechanism
The acid-catalyzed enolization of ketones proceeds via protonation of the carbonyl oxygen by an acid catalyst, which enhances the electrophilicity of the carbon and increases the acidity of the alpha-hydrogen. This is followed by deprotonation at the alpha-carbon by a base, leading to the formation of the enol. The mechanism involves the following key steps:- Protonation: The carbonyl oxygen accepts a proton, yielding a protonated ketone intermediate, .
- Deprotonation: A base abstracts the alpha-proton, with concomitant formation of the C=C double bond and regeneration of the neutral oxygen, resulting in the enol .








