Recent from talks
Nothing was collected or created yet.
Discectomy
View on Wikipedia| Discectomy | |
|---|---|
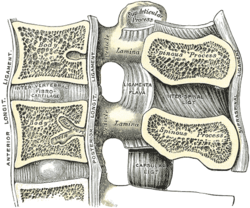 Median sagittal section of two lumbar vertebrae and their ligaments. ("Intervertebral fibrocartilage", an old name for the intervertebral disc, labeled at center left.) | |
| ICD-9-CM | 80.51 |
| MeSH | D017586 |
| MedlinePlus | 007250 |

A discectomy (also called open discectomy, if done through a 1/2 inch or larger skin opening) is the surgical removal of abnormal disc material that presses on a nerve root or the spinal cord. The procedure involves removing a portion of an intervertebral disc, which causes pain, weakness or numbness by stressing the spinal cord or radiating nerves. The traditional open discectomy, or Love's technique, was published by Ross and Love in 1971. Advances have produced visualization improvements to traditional discectomy procedures (e.g. microdiscectomy, an open discectomy using an external microscope typically done through a 1-inch or larger skin opening), or endoscopic discectomy (the scope passes internally and typically done through a 2 mm skin opening or larger, up to 12 mm). In conjunction with the traditional discectomy or microdiscectomy, a laminotomy is often involved to permit access to the intervertebral disc. Laminotomy means a significant amount of typically normal bone (the lamina) is removed from the vertebra, allowing the surgeon to better see and access the area of disc herniation.
Endoscopic discectomy
[edit]Small or ultra-small endoscopic discectomy (called Nano Endoscopic Discectomy) does not have internal cutting or bone removal and, due to the small size, is not called "open". These procedures do not cause post-laminectomy syndrome (failed back syndrome).[1][2]
Microdiscectomy
[edit]Microdiscectomy is a spine operation with a smaller incision than traditional discectomy, in which a portion of a herniated nucleus pulposus is removed by way of a surgical instrument, while using an external operating microscope for lighting and magnification. They may be "open", i.e., with a larger incision, or minimally invasive, i.e., with a 1.5 to 2.0 cm surgical incision.[3]
Indications
[edit]Microdiscectomy may be a surgical option for patients with a single-level disc herniation and evidence of nerve root compression with residual unremitting radicular symptoms after failed conservative treatment. Cauda equina syndrome and progressive or new motor deficits are among the urgent surgical indications for microdiscectomy.[3]
Contraindications
[edit]Contraindications include additional pathologies, including infection, tumor, or segmental instability or vertebral fracture where fusion or instrumentation may be required. However, segmental instability and spondylolisthesis may be considered relative contraindications by some physicians.[3]
Discectomy for athletes
[edit]Degeneration caused by years of repetitive mechanical stress can cause the disc of an athlete to be herniated. Lumbar disc herniation (LDH) is a critical injury for elite athletes that could cause extreme pain and significantly hinder performance. To relieve the pain, athletes usually go through microdiscectomy. However, the results of treatments in elite athletes differ due to the demand for optimal treatment, short recovery period, and high performance after the operation.[citation needed]
Most athletes return to their pre-surgery level after a discectomy. A systematic review of 450 athletes shows that 75–100% of athletes return to play after surgery.[4] The average recovery period ranged from 2.8 to 8.7 months. Athletes recovered an average of 64.4% to 103.6% of their preoperative performance and had reported career longevity of 2.6 to 4.8 years post-return.[4]
There are unsuccessful cases of discectomy for certain athletes like Tiger Woods, a world-famous PGA Tour golfer. Woods underwent three microdiscectomy procedures from 2014 to 2015 which failed to alleviate his pain.[5] The removal of disc material due to discectomy meant that Woods eventually had to go through spinal fusion to recover.
In the US
[edit]In the U.S., it has been estimated that the Medicare system spends over $300 million annually on lumbar discectomies.[6]
See also
[edit]References
[edit]- ^ Anderson, Greg; Vaccaro, Alexander R., eds. (2013). "Percutaneous Discogenic Pain Treatment". Decision Making in Spinal Care. doi:10.1055/b-0034-84365. ISBN 978-1-60406-417-9.
- ^ Ditsworth, D.; Geer, G.; Lombardi, L. "#455 Nano Endoscopic Approach for Central Lumbar Disc Herniations". ISASS17. Archived from the original on 20 July 2018.
- ^ a b c Dowling, Thomas J.; Dowling, Thomas J. (2021), "Microdiscectomy", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 32310444, retrieved 2021-05-07
- ^ a b Nair, Rueben; Kahlenberg, Cynthia A.; Hsu, Wellington K. (June 2015). "Outcomes of Lumbar Discectomy in Elite Athletes: The Need for High-level Evidence". Clinical Orthopaedics & Related Research. 473 (6): 1971–1977. doi:10.1007/s11999-014-3762-z. PMC 4419017. PMID 25002213.
- ^ O'Sullivan, Peter (July 2015). "Common misconceptions about back pain in sport: Tiger Woods' case brings five fundamental questions into sharp focus". British Journal of Sports Medicine. 49 (14): 905–907. doi:10.1136/bjsports-2014-094542. PMID 25807161.
- ^ Schoenfeld, A. J.; Weiner, B. K. (2010). "Treatment of lumbar disc herniation: Evidence-based practice". International Journal of General Medicine. 3: 209–214. doi:10.2147/ijgm.s12270. PMC 2915533. PMID 20689695.
Further reading
[edit]- Emmett, J. L.; Love, J. G. (1971). "Vesical dysfunction caused by protruded lumbar disk". The Journal of Urology. 105 (1): 86–91. doi:10.1016/s0022-5347(17)61466-6. PMID 5100876.
Discectomy
View on GrokipediaOverview
Definition and Purpose
A discectomy is a surgical procedure that involves the removal of part or all of an intervertebral disc, most commonly the herniated or damaged portion of the nucleus pulposus, to address disorders of the spine. This intervention targets the cushion-like structures between the vertebrae that can protrude or rupture, leading to compression of nearby neural structures.[3][2] The primary purpose of discectomy is to relieve pressure on spinal nerves or the spinal cord caused by herniated disc material, which can result in conditions such as radiculopathy, sciatica, or myelopathy. By decompressing these neural elements, the procedure aims to alleviate associated symptoms including pain, numbness, and weakness that radiate to the limbs or affect motor function. It is typically considered after conservative treatments, such as physical therapy, medications, or epidural injections, have failed to provide relief.[3][4][5] Discectomy was first conceptualized in the early 20th century as a targeted approach to treat intervertebral disc ruptures, with the landmark description by William J. Mixter and Joseph S. Barr in their 1934 publication, which established the procedure's role in managing sciatica and related neural compression unresponsive to non-surgical methods. Over time, its purpose has evolved to emphasize minimally disruptive techniques while maintaining the core goal of neural decompression without altering spinal stability. Unlike spinal fusion, which joins adjacent vertebrae to limit motion and stabilize the spine, discectomy preserves natural segmental movement. In contrast to laminectomy, which removes portions of the vertebral lamina to broaden the spinal canal, discectomy specifically focuses on excising offending disc tissue.[6][7][8][9]Relevant Anatomy
The intervertebral disc is a fibrocartilaginous structure located between adjacent vertebral bodies, consisting of two primary components: the outer annulus fibrosus and the inner nucleus pulposus. The annulus fibrosus is a tough, concentric ring of fibrocartilage and collagen fibers that encases the nucleus pulposus, providing structural integrity and resisting tensile forces during spinal motion. The nucleus pulposus is a gel-like core composed primarily of water, proteoglycans, and collagen, which functions to absorb shock, distribute loads evenly across the vertebral endplates, and allow flexibility in the spinal column. Together, these components enable the spine to withstand compressive forces while maintaining mobility.[10] Discectomy procedures most commonly target the lumbar spine, particularly the L4-L5 and L5-S1 levels, where approximately 95% of lumbar disc herniations occur due to the higher mechanical stress and mobility at these segments. Cervical discectomies are also performed, often at C5-C6 or C6-C7, to address radiculopathy or myelopathy, while thoracic discectomies are rarer, typically involving mid-to-lower thoracic levels (T8-T12) due to the relative stability of that region. These levels are selected based on the prevalence of degenerative changes and herniations that compress neural elements.[11] Key anatomical structures surrounding the intervertebral discs include the vertebral bodies, which form the bony framework; the spinal cord, which occupies the central canal up to the L1-L2 level; and the cauda equina, a bundle of lumbosacral nerve roots descending below the conus medullaris in the lumbar and sacral regions. Nerve roots emerge laterally through intervertebral foramina, with dorsal root ganglia located just proximal to the foramina, making them vulnerable to compression. The posterior longitudinal ligament runs along the posterior aspect of the vertebral bodies and discs within the spinal canal, reinforcing the disc posteriorly and limiting excessive flexion.[10][12][11] Pathophysiological changes in discectomy-relevant anatomy often involve disc herniation, classified into types such as protrusion (or prolapse), where the nucleus pulposus bulges through the annulus fibrosus without breaching it fully; extrusion, where nuclear material penetrates the annulus but remains connected to the disc; and sequestration, where a free fragment detaches entirely. These herniations can lead to central canal stenosis by compressing the spinal cord or cauda equina in the midline, or foraminal stenosis by impinging on exiting nerve roots laterally, thereby disrupting neural function.[11][13]Types of Discectomy
Microdiscectomy
Microdiscectomy, a minimally invasive surgical approach to lumbar disc herniation, was developed in the 1970s by neurosurgeons M. Gazi Yaşargil and Wolfhard Caspar, who independently introduced the use of an operating microscope to enhance precision in disc removal.[14][15] This technique gained widespread adoption as the standard for treating lumbar disc herniations by the 1980s, building on earlier open discectomy methods but incorporating magnification to minimize tissue disruption.[14][16] The procedure involves a small incision, typically 2-3 cm in length, made paramedian to the midline over the affected vertebral level to allow direct access to the interlaminar space.[15][5] Under magnification provided by an operating microscope or surgical loupes, a partial laminotomy is performed to create a limited bony window in the lamina, exposing the ligamentum flavum and nerve root without extensive muscle retraction.[15][5] The herniated disc fragment is then carefully removed while preserving the integrity of the remaining healthy disc material and annular structure to avoid postoperative instability.[15] Specialized instrumentation facilitates the delicate dissection and removal process, including microdiscectomes for precise cutting, Kerrison rongeurs for bone and ligament trimming, and pituitary forceps for grasping and extracting disc fragments.[15][17] Surgeons emphasize a conservative approach, avoiding aggressive removal of disc tissue beyond the herniated portion to maintain spinal stability and reduce the risk of recurrent herniation.[15] Compared to traditional open discectomy, microdiscectomy offers advantages such as reduced tissue trauma due to the smaller exposure, shorter operative times averaging 1-2 hours, and lower intraoperative blood loss, often negligible in volume.[18][15] These benefits contribute to less postoperative pain and faster initial mobilization, though microdiscectomy differs from endoscopic discectomy in its reliance on direct microscopic visualization rather than camera-guided tubular access.[18][19]Endoscopic Discectomy
Endoscopic discectomy represents an ultra-minimally invasive surgical technique for removing herniated disc material using an endoscope, allowing direct visualization through small portals without extensive tissue disruption. The procedure typically employs a percutaneous or transforaminal approach, where a 6-8 mm cannula is inserted through a small skin incision, guided by fluoroscopy to access the affected disc space. Once positioned, a rigid endoscope provides high-resolution imaging of the neural structures and herniation, enabling precise removal of disc fragments using specialized instruments such as forceps, rongeurs, or shavers; adjunctive methods like laser or radiofrequency ablation may be used for decompression in select cases.[20][21] Key variants include transforaminal endoscopic lumbar discectomy (TELD), which accesses the disc via Kambin's triangle in a posterolateral trajectory to target foraminal or extraforaminal herniations, and the interlaminar endoscopic approach, which provides posterior access similar to traditional methods but with reduced exposure. Another variant is unilateral biportal endoscopy (UBE), utilizing two small portals—one for the endoscope and one for working instruments—to facilitate decompression. These approaches are tailored to the herniation's location, with TELD often preferred for L4-L5 and higher levels due to anatomical feasibility.[20][22] The primary advantages of endoscopic discectomy stem from its minimal invasiveness, featuring incisions under 1 cm that preserve paraspinal muscles and ligaments, thereby reducing postoperative pain and accelerating recovery. It supports outpatient procedures in many cases, with patients often discharged within hours and returning to work sooner than with more invasive options; success rates range from 85-90% in carefully selected patients with contained herniations. Compared to microdiscectomy, it offers even less muscle disruption while similarly targeting symptomatic disc protrusions.[20][21] This technique evolved from early percutaneous nucleotomy concepts in the 1970s, pioneered by Kambin and Hijikata, and gained traction in the 1990s with fiberoptic endoscope advancements enabling arthroscopic discectomy. Refinements in the 2000s, including micro-endoscopic systems by Foley in 1997 and biportal innovations in 2016, expanded its applicability to a broader range of lumbar pathologies, driven by improvements in imaging and instrumentation.[20][22][21]Percutaneous Laser Disc Decompression
Percutaneous laser disc decompression (PLDD) is a minimally invasive procedure for treating contained lumbar disc herniations by reducing intradiscal pressure with laser energy. Under local anesthesia and fluoroscopic guidance, a hollow needle is inserted percutaneously into the nucleus pulposus, where the laser evaporates water content and denatures proteins, allowing the herniated disc material to retract and decompress the adjacent nerve root. This outpatient technique preserves surrounding tissues without requiring general anesthesia or incisions beyond the needle tract.[23] PLDD is ideal for contained, mild-to-moderate herniations without severe muscle weakness, whereas traditional discectomy is better suited for large, extruded herniations or severe symptoms such as weakness or cauda equina syndrome.[24]Open Discectomy
Open discectomy, also known as conventional or standard discectomy, represents the traditional surgical approach to removing herniated intervertebral disc material that compresses spinal nerves. First performed in 1934 by William J. Mixter and Joseph S. Barr, this procedure established the direct correlation between lumbar disc herniation and sciatica, revolutionizing treatment for radiculopathy by excising prolapsed disc fragments through a posterior approach.[25] Initially involving a full laminectomy to access the spinal canal, it became the gold standard for disc surgery for decades until the advent of less invasive techniques in the late 20th century.[26] The technique employs a larger midline incision, typically measuring 4 to 6 cm, centered over the affected vertebral level to allow broad exposure of the posterior spine.[27] Surgeons perform a full laminectomy or hemilaminectomy to remove portions of the lamina bone, retracting paraspinal muscles and ligaments for direct visualization of the thecal sac and nerve roots without the use of magnification or specialized optics.[28] Once the herniated disc is identified, a complete or subtotal discectomy is conducted by incising the posterior longitudinal ligament and annulus fibrosus, followed by extraction of the extruded nucleus pulposus and any free fragments using rongeurs or curettes.[28] This method ensures thorough decompression but involves greater muscle dissection and potential disruption to surrounding tissues compared to refined variants.[29] In contemporary practice, open discectomy is primarily reserved for complex cases, such as those involving large free disc fragments, multilevel herniations, or revision surgeries where extensive exploration of the spinal canal is necessary, including large extruded herniations or severe symptoms like weakness or cauda equina syndrome.[30] It facilitates comprehensive inspection and removal of pathology that may be challenging to access via narrower approaches, though it results in higher degrees of tissue trauma.[30] Drawbacks include increased postoperative pain due to extensive muscle retraction and incision size, often persisting for one to two weeks with associated soreness and swelling.[31] Recovery historically required 2 to 4 weeks of bed rest, though modern protocols have shortened this to about one month for return to normal activities, supported by enhanced pain management and early mobilization.[32]Indications and Patient Selection
Primary Indications
Discectomy is primarily indicated for patients experiencing severe radicular pain, such as sciatica persisting beyond 6 weeks despite conservative therapy, which arises from nerve root compression due to lumbar disc herniation.[3] Similarly, in the cervical spine, it is indicated for radiculopathy causing persistent arm pain, numbness, or weakness after 4-6 weeks of conservative management.[33] Neurological deficits, including motor weakness like foot drop or sensory loss in the affected dermatome, further support surgical intervention when these symptoms impair daily function and do not resolve with nonoperative measures.[25] In the cervical region, progressive myelopathy with gait disturbance or upper extremity weakness may warrant urgent surgery.[33] In emergency cases, cauda equina syndrome—characterized by saddle anesthesia, bowel or bladder dysfunction, and bilateral leg weakness—necessitates immediate discectomy to prevent permanent neurological damage.[34] Diagnostic confirmation relies on imaging studies that correlate clinical symptoms with anatomical pathology, such as magnetic resonance imaging (MRI) demonstrating disc herniation impinging on the nerve root, or computed tomography (CT) in cases where MRI is contraindicated.[35] Electromyography (EMG) may be employed to assess nerve involvement and confirm radiculopathy, particularly when symptoms are atypical or to differentiate from other neuropathies.[36] Patient selection emphasizes those who have failed 6-12 weeks of conservative management, including nonsteroidal anti-inflammatory drugs (NSAIDs) for pain and inflammation control, along with epidural steroid injections to reduce nerve root irritation.[37] For lumbar discectomy, individuals aged 20-50 years often represent the ideal cohort, as this demographic experiences peak incidence of symptomatic herniations and typically exhibits robust recovery potential post-surgery.[38] The Spine Patient Outcomes Research Trial (SPORT), a landmark randomized controlled study published in 2006, provides robust evidence supporting discectomy for these indications, demonstrating superior relief of leg pain and improved functional outcomes compared to nonoperative care at 2- and 4-year follow-ups, particularly in patients with persistent radiculopathy.[39]Contraindications
Discectomy, a procedure to remove herniated disc material, carries specific contraindications that must be evaluated to avoid excessive risk or poor outcomes. Absolute contraindications include active local or systemic infections, such as spinal osteomyelitis or epidural abscess, which increase the risk of postoperative sepsis.[3] Malignant tumors with dural involvement or segmental spinal instability, including unstable spondylolisthesis, also preclude the procedure due to the potential for neurological deterioration or need for immediate stabilization.[3][15] Severe osteoporosis with high fracture risk is another absolute barrier, as it compromises bone integrity during surgical manipulation and healing.[40] Relative contraindications encompass conditions that elevate perioperative risks but may not entirely rule out surgery with careful management. Coagulopathy, whether congenital or acquired (e.g., from anticoagulants), heightens bleeding risks and is typically addressed through correction prior to intervention, though persistent issues may defer the procedure.[25] Poor overall surgical candidacy, including uncontrolled diabetes mellitus or cardiovascular disease, impairs wound healing and increases complication rates, making patients suboptimal candidates.[41] Non-discogenic pain sources, such as facet joint arthropathy or vascular claudication, represent relative barriers when symptoms do not correlate with imaging findings of disc herniation, as surgery may fail to alleviate symptoms.[25] Bony spinal canal stenosis or vertebral fractures requiring fusion further complicate candidacy, often necessitating alternative approaches.[3] Type-specific contraindications vary by technique. For endoscopic discectomy, far-lateral or highly migrated herniations (≥3 mm), severe spinal stenosis, calcified discs, or significant preoperative scarring from prior surgery are inadvisable due to technical limitations in visualization and access.[42][43] Microdiscectomy is relatively contraindicated in revision cases with extensive epidural scarring or high-grade spondylolisthesis (>grade 2), where instability may worsen post-decompression without concurrent fusion.[15][44] When contraindications are present, alternatives prioritize non-surgical options like physical therapy, epidural steroid injections, or percutaneous discectomy for symptom management, while spinal fusion may be considered for instability-related cases to provide decompression and stabilization.[25][15]Surgical Procedure
Discectomy procedures vary by spinal region, with lumbar being the most common. The following primarily describes lumbar discectomy; cervical discectomy typically uses an anterior approach, often combined with fusion (ACDF) for stability, as detailed in the introduction.[2][45]Preoperative Evaluation
The preoperative evaluation for lumbar discectomy begins with comprehensive imaging to confirm the diagnosis and assess the extent of disc herniation. Magnetic resonance imaging (MRI) serves as the gold standard for evaluating soft tissue pathology, providing detailed visualization of the herniated disc, nerve root compression, and spinal cord involvement with high diagnostic accuracy.[46] Plain X-rays are routinely obtained to evaluate spinal alignment, bone structure, and any degenerative changes that may influence surgical planning. In cases where MRI is contraindicated, such as in patients with pacemakers or severe claustrophobia, computed tomography (CT) myelography is utilized as an alternative to delineate the thecal sac and nerve roots effectively. For cervical discectomy, imaging is similar, but additional emphasis may be placed on assessing spinal cord compression and vascular structures via MRI or CT angiography if needed. Neurological examination includes region-specific tests, such as the straight-leg raise for lumbar radiculopathy (reproducing pain at L4-L5 or L5-S1 levels to confirm nerve root irritation) or Spurling's maneuver for cervical nerve root compression.[3] Clinical assessment focuses on a thorough neurological examination and medical history review to identify deficits and comorbidities that could impact surgical safety. A detailed medical history is reviewed to screen for comorbidities, including cardiovascular disease, necessitating cardiac clearance via risk stratification guidelines that incorporate clinical predictors and functional status to minimize perioperative cardiac events. Patient education is integral, emphasizing informed consent that outlines the procedure's risks, benefits, and alternatives, such as conservative management, to ensure shared decision-making. Patients are advised on smoking cessation, as preoperative nicotine use increases the risk of surgical site infections, myocardial infarction, and prolonged recovery following discectomy, with cessation even four weeks prior reducing these complications. Anesthesia planning typically involves general anesthesia as the standard approach for discectomy due to its provision of secure airway management and patient tolerance during positioning, though regional anesthesia like spinal may be considered in select lumbar cases for potential benefits in pain control. For cervical procedures, general anesthesia is also standard, with attention to airway management due to the anterior neck approach. Preoperative laboratory work, including coagulation studies and infection screening (e.g., complete blood count and inflammatory markers), is performed to identify bleeding risks or subclinical infections that could elevate perioperative morbidity.Intraoperative Techniques
The intraoperative phase of lumbar discectomy begins with patient positioning, typically prone on a radiolucent operating table using a frame such as the Wilson or Allen bow to facilitate access to the lumbar spine.[3] This position involves transverse pads at the iliac crests and chest, with hips and knees slightly flexed to induce mild lumbar lordosis, optimizing the interlaminar window and reducing intra-abdominal pressure.[3] General anesthesia is administered, and prophylactic antibiotics, such as cefazolin, are given intravenously within 30-60 minutes prior to incision to prevent surgical site infections.[3] For cervical discectomy, patients are positioned supine with the neck slightly extended, using a shoulder roll for access via an anterior transverse incision along skin creases, typically 2-4 cm long.[45] The incision varies by technique to minimize tissue disruption. In open discectomy, a 3-4 cm midline longitudinal incision is made over the affected level, providing broad exposure of the paraspinal muscles and lamina.[3] Microdiscectomy employs a smaller 2-3 cm midline or paramedian incision, utilizing an operating microscope or surgical loupes for magnified visualization and precise dissection through the interlaminar space.[3] For endoscopic discectomy, a 1-1.5 cm incision is placed laterally (e.g., via the Kambin triangle approach), allowing insertion of an endoscope for navigation and removal under direct endoscopic guidance without extensive muscle retraction.[3] Retractors, such as self-retaining Gelpi or Caspar types, are applied in open and micro approaches to expose the ligamentum flavum, which is then partially resected to access the epidural space; tubular retractors are preferred in minimally invasive variants to maintain a narrow corridor.[3] In cervical procedures, the anterior approach involves dissecting through the anterior neck muscles to access the disc space, with removal of the herniated disc using rongeurs and curettes, followed by insertion of a graft or cage for fusion.[47] The core discectomy involves careful decompression of the neural elements. The dura and traversing nerve root are gently retracted using blunt probes or nerve root retractors to visualize the herniated fragment.[3] The posterior annulus fibrosus is incised with a scalpel or No. 11 blade, and the herniated disc material is removed piecemeal using pituitary rongeurs, Kerrison rongeurs, or curettes, ensuring complete decompression without aggressive disc space exploration to avoid iatrogenic instability.[3] In microdiscectomy, the microscope aids in hemostasis and fragment extraction under high magnification; endoscopic variants rely on the scope for real-time irrigation and visualization during removal.[3] The surgical field is copiously irrigated with saline to clear debris and loose fragments, followed by meticulous hemostasis using bipolar electrocautery to control epidural venous bleeding.[3] Intraoperative neuromonitoring is routinely employed to safeguard neural structures, particularly in cases with significant nerve root compression or foraminal involvement, for both lumbar and cervical procedures. Somatosensory evoked potentials (SSEPs) assess sensory pathways, while motor evoked potentials (MEPs) monitor motor integrity, with alerts triggered by signal changes exceeding 50% amplitude reduction or 10% latency increase.[48] The procedure typically lasts 1-2 hours, depending on the herniation complexity and surgical approach, with endoscopic techniques often shorter due to reduced exposure time.[2] Closure proceeds in layers to promote wound healing without routine drainage, as drains are generally unnecessary in uncomplicated discectomies. The paraspinal musculature and fascia are approximated with absorbable sutures (e.g., Vicryl), the subcutaneous layer closed with interrupted sutures, and the skin approximated using subcuticular stitches or staples.[3] For cervical closure, multilayer closure includes reapproximation of the platysma and skin, often with drains to prevent hematoma. Optionally, vancomycin powder may be applied to the wound bed prior to closure for additional antimicrobial prophylaxis.[3]Postoperative Management
Following lumbar discectomy, patients typically experience a short hospital stay of 1 to 2 days, allowing for initial recovery from anesthesia and monitoring for any immediate issues, though many are discharged the same day for minimally invasive procedures.[1][32] For cervical discectomy, hospital stay is similar, 1-2 days, with monitoring for dysphagia or airway issues due to the anterior approach.[45] Pain management in the immediate postoperative period often involves multimodal analgesia, starting with short-term opioids for severe discomfort in the first 24-48 hours, transitioning to nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen or acetaminophen to minimize opioid use and side effects like constipation or dependency.[49][50] Gabapentinoids like pregabalin may also be incorporated for neuropathic pain relief, with efficacy demonstrated in reducing overall pain scores without increasing complications.[51] Early mobilization is encouraged within 24 hours of surgery to promote circulation, prevent blood clots, and facilitate faster recovery, beginning with assisted walking in the hospital and progressing to short, frequent ambulation sessions.[52][53] Patients are typically out of bed on the day of surgery or the first postoperative day, with goals of 30 minutes of walking twice daily by discharge, under supervision to ensure safety. For cervical patients, mobilization includes neck precautions to avoid extension.[54] Wound care focuses on maintaining sterility to prevent infection, with the incision site covered by a sterile dressing for the first 3-5 days, during which it should remain dry and patients avoid submerging it in water.[55] Dressings are changed every other day after the initial period or as needed, using clean technique, and the site is monitored daily for signs of infection such as fever above 101°F (38.3°C), increasing redness (erythema), swelling, or purulent drainage, as well as other warning signs requiring immediate medical attention, particularly after lumbar procedures: severe increase in pain, redness/swelling/discharge from incision, new numbness or weakness in legs, or problems with bladder/bowel control.[55][56][57] Gentle cleaning with mild soap and water is permitted once the dressing is removed, patting dry without rubbing.[58] For cervical wounds, additional monitoring for hematoma or swallowing difficulties is required. Activity restrictions are implemented to protect the surgical site during the initial healing phase, generally prohibiting bending at the waist, twisting, or lifting more than 5-10 pounds for 4-6 weeks to reduce reherniation risk.[59][60] Prolonged sitting is limited to 20-30 minutes at a time, with encouragement to alternate positions and use lumbar support, while a back brace may be optionally used for lumbar procedures to provide stability during the first 4 weeks, though evidence for routine use is mixed.[59][56] Cervical patients follow neck-specific restrictions, avoiding heavy lifting and overhead activities for 4-6 weeks, often with a cervical collar for 1-2 weeks.[45] Follow-up care includes an initial clinic visit at 10-14 days postoperatively for wound assessment and suture or staple removal if non-absorbable materials were used, with imaging such as MRI considered only if symptoms like persistent radiculopathy or neurological deficits arise.[61][62] Subsequent visits occur at 6 weeks and 3 months to evaluate progress, adjusting restrictions as healing advances toward short-term recovery at home.[62] Follow-up for cervical procedures is similar but includes assessment of fusion progress via X-ray at 6-12 weeks.Outcomes and Recovery
Short-term Recovery
Short-term recovery following discectomy typically spans the initial weeks to months after surgery, with most patients experiencing gradual improvements in symptoms and function. Discectomy provides advantages including rapid leg pain relief and suitability for severe symptoms impacting quality of life. Leg pain, often caused by nerve compression, is frequently relieved immediately or within days post-procedure due to the decompression of the affected nerve root.[63] In contrast, residual back pain from surgical incision and tissue manipulation may persist for 2-4 weeks, gradually subsiding as inflammation decreases and mobility increases.[64] Rehabilitation plays a central role in short-term recovery, with physical therapy commonly initiated 1-2 weeks after surgery to promote healing and prevent complications. Early sessions focus on gentle core strengthening exercises, such as pelvic tilts and bridges, alongside posture education and low-impact activities like walking to restore spinal stability without straining the surgical site. Patients are advised to avoid high-impact or twisting movements during this phase to minimize re-injury risk.[65] For patients undergoing lumbar discectomy, postoperative recovery involves phased rehabilitation under medical supervision, individualized according to the type of surgery and patient-specific factors. The approach emphasizes gradual progression to restore function while avoiding excessive lumbar stress. Recommended activities include:- Early phase (starting from 1-2 days post-surgery): straight leg raise training (multiple times daily to prevent nerve root adhesion) and five-point support exercises (slowly raising the hips, holding briefly, and lowering, in 3-5 sets per day to strengthen waist and back muscles).
- Mid phase: low-impact exercises such as slow walking (20-30 minutes), swimming (breaststroke or freestyle, avoiding butterfly stroke), cycling, along with gradual enhancement of waist and back muscle strength and maintenance of correct posture.
- Bending, twisting, and lifting more than 5 kg for the first month.
- Running, jumping, and high-impact activities for 3 months.
- Twisting sports such as golf or bowling for 6 months.
- Prolonged sitting or standing, intense activities, butterfly stroke, or other movements increasing lumbar pressure.
- Use of a waist brace beyond 1 month to prevent waist and back muscle atrophy.
Long-term Outcomes
Studies on the long-term efficacy of discectomy reveal that 70-90% of patients experience sustained pain relief and functional improvement at 5-10 years post-surgery. For percutaneous laser disc decompression (PLDD), a minimally invasive variant, success rates range from 70-89% in most studies, with notable long-term (2+ years) improvements in pain and function; some studies show 30-74% pain reduction, with results depending on patient selection.[67][68] For instance, a comprehensive analysis of over 39,000 patients reported good or excellent outcomes in 78.9% at a mean follow-up of 6.1 years, with microdiscectomy achieving 84.3% success.[69] Similarly, in a 10-year follow-up study, approximately 80% of patients maintained significant pain reduction following lumbar microdiscectomy.[70] Reoperation rates are approximately 10-15% over this period (14.4% at 5 years in one nationwide cohort), particularly for select patients with persistent radicular symptoms, where discectomy outperforms conservative management by reducing the need for eventual surgical intervention compared to crossover rates in nonoperative groups.[71] Several factors influence these outcomes, with smoking and obesity associated with poorer prognosis and higher recurrence risk. Smokers exhibit a reoperation rate of 6.4% for recurrent lumbar disc herniation, compared to 4.0% in nonsmokers, due to impaired disc healing and increased degeneration.[72] Obesity similarly worsens results, with obese patients (BMI ≥30) showing 12 times higher odds of recurrent herniation and less favorable Oswestry Disability Index (ODI) improvements at long-term follow-up.[73] Recurrence risk is generally 5-15%, as multilevel disc disease can predispose to accelerated adjacent segment degeneration and instability.[74] Quality of life measures, such as the ODI, demonstrate durable gains, with mean scores improving from preoperative levels of around 67% to 10% at 5 years in endoscopic discectomy cohorts.[75] Data from the Swedish Spine Register corroborate this, indicating excellent to good results in 76-86% of adolescent and adult patients at 1 year, with significant reductions in disability.[76] Compared to conservative treatments, discectomy provides superior relief for persistent symptoms, though approximately 10% of cases may require subsequent fusion due to iatrogenic instability.[77]Complications and Risks
Intraoperative Risks
Intraoperative risks during discectomy primarily involve potential damage to neural, vascular, and anesthetic structures, though these are generally low with modern techniques. These complications can arise from direct surgical manipulation, positioning, or procedural factors, but most are identifiable and managed in real time to prevent long-term sequelae.[78] Nerve-related risks are among the most common intraoperative concerns. Dural tears occur in approximately 1-4% of lumbar discectomy cases, often due to inadvertent incision during decompression or retraction, and are typically repaired immediately with sutures or sealants to prevent cerebrospinal fluid leakage.[79] Nerve root injury is rarer, affecting less than 1% of procedures, and usually results from prolonged retraction or direct trauma, leading to transient sensory or motor deficits that may require immediate release of pressure.[25] In endoscopic approaches, the incidence of dural tears may be slightly higher (1.7-4.3%) due to limited visualization and instrument maneuvering.[80] Vascular complications, while infrequent, can be serious. Epidural bleeding can occur from venous sources in the epidural space and is typically controlled through hemostatic agents or bipolar cautery.[78] Major arterial injuries are exceedingly rare and more associated with anterior or lateral approaches, potentially causing massive hemorrhage.[81] Anesthesia-related risks under general anesthesia include airway management issues, such as difficult intubation or ventilation challenges in the prone position. These are mitigated by preoperative airway assessment.[82] Several strategies reduce these intraoperative risks. Intraoperative neuromonitoring, using somatosensory or motor evoked potentials, allows real-time detection of neural compromise from retraction or ischemia, enabling prompt adjustments.[83] Fluoroscopy provides precise guidance for instrument placement, particularly in minimally invasive or endoscopic discectomy, minimizing blind dissection and vascular or dural injury.[80] Preoperative screening for anatomical variants further supports these measures by identifying high-risk patients.[78]Postoperative Complications
Postoperative complications following discectomy, while relatively uncommon, can significantly impact patient recovery and may require additional interventions. These complications typically arise in the immediate to intermediate postoperative period and include infections, recurrent disc herniation, cerebrospinal fluid (CSF) leaks due to dural tears, epidural hematomas, scar adhesion or epidural fibrosis, and the onset of adjacent segment disease. Overall complication rates for lumbar microdiscectomy range from 10% to 16%, with most being manageable through conservative or targeted treatments.[84][85] Infection remains one of the most concerning postoperative issues, manifesting as superficial wound infections or deeper discitis. Superficial wound infections occur in approximately 1-2% of cases, presenting with erythema, drainage, or fever within the first few weeks after surgery.[25] Discitis, a more severe deep infection involving the disc space, affects 0.5-1% of patients and is often diagnosed through imaging and culture confirmation, typically 2-8 weeks postoperatively.[86] Risk factors include obesity, diabetes, and intraoperative contamination, though prophylactic antibiotics reduce incidence. Management involves early administration of targeted intravenous antibiotics based on culture results, with surgical debridement required in up to 50% of discitis cases to prevent chronic osteomyelitis or abscess formation.[86][25] Recurrent disc herniation, defined as re-protrusion of disc material at the same operative level, occurs in 5-15% of patients within the first two years, most commonly due to incomplete initial decompression or annular defects.[87][88][89] Symptoms such as radicular pain or weakness typically emerge within months, confirmed by MRI. This complication is more frequent at the L4-L5 level and can be mitigated by meticulous annular closure techniques during the primary procedure. Approximately 20% of recurrent cases necessitate further surgery, often a repeat discectomy, while others resolve with conservative measures like physical therapy or epidural injections.[87][88] Epidural scar adhesion or fibrosis, arising from postoperative inflammation and healing, is common in lumbar discectomy and can lead to neural tethering, chronic pain, or failed back surgery syndrome. While often asymptomatic, symptomatic cases contribute to persistent radiculopathy and complicate reoperations.[90] Other notable complications include CSF leaks secondary to intraoperative dural tears, which occur in 1-3% of discectomies and may present postoperatively as positional headaches, clear drainage from the wound, or pseudomeningocele formation.[91] These leaks, often stemming from unrecognized tears during surgery, are managed conservatively with bed rest, hydration, and caffeine in most cases, though persistent leaks (>7 days) may require epidural blood patching or surgical repair to avoid meningitis. Epidural hematomas, though rare (0.1-0.2% symptomatic), can cause acute neurological deficits due to cord compression and demand urgent evacuation if diagnosed via MRI.[92][91] Adjacent segment disease, involving degenerative changes at levels adjacent to the operated site, begins to manifest in 4-5% of patients within 3-5 years post-discectomy, driven by altered biomechanics and increased load transfer.[93] Early signs include back pain or stenosis, progressing to require reoperation in about 1-2% annually; prevention focuses on preserving motion through minimally invasive approaches. Overall, vigilant monitoring in the postoperative period, including serial imaging for high-risk patients, facilitates timely intervention and improves outcomes.[93][25]Cervical-Specific Risks
In cervical discectomy, particularly anterior cervical discectomy and fusion (ACDF), additional risks include dysphagia (difficulty swallowing, 10-50% incidence, often transient), hoarseness due to recurrent laryngeal nerve injury (1-5%), and esophageal perforation (rare, <1%). These are managed with careful surgical technique and postoperative monitoring.[94][95]Special Populations
Discectomy in Athletes
Discectomy is indicated in athletes for acute lumbar disc herniations often resulting from high-impact collisions in contact sports, such as those experienced by football linemen during blocking maneuvers. These injuries typically arise from sudden axial loading and torsional forces that compress the intervertebral disc, leading to extrusion of nucleus pulposus material and subsequent radiculopathy or cauda equina symptoms unresponsive to conservative management. In high-demand sports like baseball pitching or weightlifting, repetitive microtrauma accelerates disc degeneration, prompting surgical intervention when symptoms impair performance and imaging confirms focal herniation.[96][97] Among athletes, microdiscectomy is the preferred surgical approach due to its minimally invasive nature, allowing precise removal of herniated fragments through a small incision while preserving surrounding disc material and spinal stability. This technique minimizes disruption to the annulus fibrosus and facet joints, which is critical for maintaining biomechanical integrity under athletic loads, unlike more extensive discectomies that risk postoperative instability or accelerated degeneration. Surgeons emphasize limited resection to only the offending fragment, guided by intraoperative microscopy or endoscopy, to optimize long-term spinal function in dynamic environments.[77] Recovery protocols for athletes incorporate sport-specific physical therapy to restore core strength, proprioception, and neuromuscular control, typically spanning 3-6 months before clearance for NFL players, with gradual progression from isometric exercises to plyometrics and position-specific drills. Clearance criteria include absence of radicular pain, full lumbar range of motion, symmetric strength, and negative provocative tests like the straight-leg raise, often verified through functional assessments and imaging if needed. These adaptations extend general recovery timelines to accommodate high-intensity demands, focusing on injury prevention through biofeedback and bracing during reintegration.[98][99] Outcomes in athletes following discectomy show 70-85% successfully returning to pre-injury performance levels, with pooled return-to-play rates around 80% across elite sports, though career longevity may shorten by 1-2 years on average. Recurrence rates are elevated in elite athletes due to ongoing physical stresses, ranging 5-15% overall and higher in overhead sports like MLB pitching, where rotational forces contribute to re-herniation in up to 10-15% of cases within 5 years. Factors such as younger age and single-level surgery predict better results, underscoring the procedure's efficacy for select athletic populations.[100][98]Regional Variations in the United States
In the United States, lumbar discectomy remains one of the most common spinal procedures, with approximately 300,000 performed annually as of the early 2020s.[101] Regional prevalence shows significant variation, with higher procedure rates in southern and south-central states such as Texas and Florida, where large spine centers contribute to elevated volumes compared to national averages.[102][103] These disparities are partly attributed to demographic factors, including higher concentrations of manual labor occupations in these regions, which correlate with increased incidence of degenerative disc conditions necessitating surgery.[104] Healthcare coverage for discectomy is broadly available through Medicare and Medicaid, which reimburse for medically necessary procedures like microdiscectomy when conservative treatments fail.[105] The average cost ranges from $20,000 to $50,000, varying by state due to differences in facility fees, surgeon expertise, and regional pricing; for instance, costs are typically higher in high-volume states like Florida and Texas owing to specialized spine centers.[106][107] Access disparities persist, particularly between rural and urban areas, where rural patients face longer wait times for general surgical care—often exceeding urban averages by 10-20%—and must travel greater distances for care, exacerbating delays in elective procedures.[108][109] The American Association of Neurological Surgeons (AANS) and Congress of Neurological Surgeons (CNS) endorse microdiscectomy as a standard treatment for lumbar disc herniation in guidelines, recommending it for patients with radiculopathy unresponsive to nonoperative management.[110] Recent trends indicate a shift toward outpatient settings, with approximately 60% of discectomies performed ambulatory by 2025, driven by advancements in minimally invasive techniques and expansions under the Affordable Care Act (ACA) that improved insurance coverage and reduced barriers to elective spine care.[111][112] This transition has been particularly pronounced in states with robust ACA implementation, leading to a 17-23% increase in overall spine surgery volumes in expansion states.[113]History and Developments
Early Procedures
The concept of lumbar disc herniation as a pathological entity emerged in the early 20th century through autopsy findings. In 1911, Scottish physicians George S. Middleton and John H. Teacher described the first documented case of spinal cord injury due to rupture of an intervertebral disc in a 20-year-old mining engineer who developed sudden paraplegia while lifting heavy coal; at autopsy, they identified a herniated disc at the T10-T11 level compressing the spinal cord.[114] This report provided initial evidence of disc protrusion as a cause of neurological symptoms, though it was not widely recognized at the time.[26] Surgical intervention for presumed disc-related compression began tentatively in the 1910s, with limited understanding of the condition. German surgeon Fedor Krause likely performed the first successful removal of a ruptured disc in 1909, as described in his collaboration with Oppenheim, though the case was interpreted as tumor resection.[115] In 1911, American orthopedic surgeon Joel E. Goldthwaite first linked disc pathology to sciatica in a clinical case, suggesting mechanical compression of nerve roots.[115] However, adoption of intentional discectomy remained rare until the 1930s due to diagnostic limitations, including reliance on plain radiographs and exploratory surgery, as advanced imaging like MRI did not exist.[26] The landmark advancement came in 1934 when neurosurgeon William J. Mixter and orthopedic surgeon Joseph S. Barr published their seminal paper in the New England Journal of Medicine, reporting 19 cases of ruptured intervertebral discs causing sciatica and detailing surgical removal via open laminectomy.[6] Their work established the causal link between disc herniation and radiculopathy, shifting treatment from conservative nerve exploration to targeted disc excision. Initial techniques involved complete or hemilaminectomy to access the spinal canal, often through a transdural approach, followed by manual removal of the herniated nucleus pulposus.[26] These procedures carried high morbidity in the pre-antibiotic era, with infection rates reaching up to 20% due to open exposure and poor sterile conditions.[116] Early discectomies faced significant challenges, including frequent misdiagnosis of herniated discs as spinal tumors or chondromas, leading to unnecessary extensive resections.[26] Mortality rates ranged from 5% to 10%, primarily from intraoperative bleeding, cerebrospinal fluid leaks, and subsequent complications like meningitis.[117] Limited adoption persisted into the 1940s, as diagnosis depended on clinical symptoms and myelography (introduced in the 1920s but refined later), often resulting in delayed or incorrect interventions.[26] Despite these hurdles, Mixter and Barr's findings laid the foundation for discectomy as a standard procedure for relieving sciatica by decompressing nerve roots.Modern Advancements
The introduction of the operating microscope in the 1970s marked the onset of the minimally invasive era in discectomy, enabling smaller incisions and improved visualization of neural structures during lumbar procedures. Pioneered by surgeons such as M. Gazi Yasargil in 1970 and further refined by Robert W. Williams and Wolfgang Caspar in 1977, this advancement reduced tissue trauma compared to traditional open techniques, leading to shorter recovery times and lower complication rates.[116] By the 1990s, endoscopic approaches emerged, allowing percutaneous access through tubular retractors, while laser-assisted methods like percutaneous laser disc decompression (PLDD) gained traction for targeted disc ablation without extensive dissection; PLDD, first described by Peter Ascher and colleagues in the late 1980s and refined in the 1990s, uses laser energy to vaporize herniated material via a needle insertion.[118] These innovations built on earlier foundations but shifted focus toward outpatient feasibility and precision. In the cervical spine, advancements paralleled lumbar developments but emphasized anterior approaches due to anatomical constraints. The anterior cervical discectomy was first described in the 1950s, with Ralph Cloward introducing discectomy via an anterior route in 1958, often combined with fusion using bone grafts. This evolved into anterior cervical discectomy and fusion (ACDF), popularized by Robinson and Smith in 1955, becoming the standard for cervical radiculopathy and myelopathy from disc herniation. Microscopic and endoscopic techniques for cervical discectomy emerged in the 1980s and 1990s, reducing complications like dysphagia associated with open anterior exposures.[119] Technological aids have further enhanced discectomy accuracy since the 2000s, with intraoperative navigation systems like Medtronic's StealthStation providing real-time 3D imaging guidance to minimize radiation exposure and improve screw placement in adjacent fusion cases. Adopted widely in spine surgery from the early 2000s, StealthStation integrates CT or fluoroscopic data for dynamic tracking, reducing navigational errors by up to 50% in complex lumbar procedures.[120] Robotics, exemplified by the Mazor X system (introduced in 2016 by Mazor Robotics, now Medtronic), offers preoperative planning and robotic arm stabilization for precise trajectories, with adoption surging in the 2010s; clinical studies report accuracy rates exceeding 98% for pedicle screw insertion, potentially lowering revision rates in discectomy-augmented stabilizations.[121] Evidence-based validation of these advancements came through randomized controlled trials (RCTs), such as the 1983 Maine Lumbar Spine Study, which demonstrated superior outcomes for microdiscectomy over conservative management in patients with sciatica from disc herniation, with 80-90% reporting significant pain relief at one year.[122] More recent evidence includes 2020s applications of artificial intelligence (AI) for preoperative planning, where machine learning models analyze MRI scans to predict herniation severity and optimal surgical approaches, achieving over 85% accuracy in outcome forecasting and aiding personalized risk assessment.[123] Looking to future directions, biologics such as mesenchymal stem cells (MSCs) and platelet-rich plasma (PRP) are under investigation for post-discectomy disc repair to mitigate recurrence and degeneration; ongoing trials as of 2025, including those with autologous nucleus pulposus cells post-endoscopic discectomy, show promising preservation of disc height and reduced inflammation in early-phase results.[124] For instance, a 2025 study on MSC-seeded gels reported improved water retention and functional scores compared to controls, highlighting potential for regenerative integration in standard discectomy protocols.[125]References
- https://pubmed.ncbi.nlm.nih.gov/8855462/

