Recent from talks
Nothing was collected or created yet.
Thelaziasis
View on Wikipedia| Thelaziasis | |
|---|---|
| Other names | Thelaziosis |
 | |
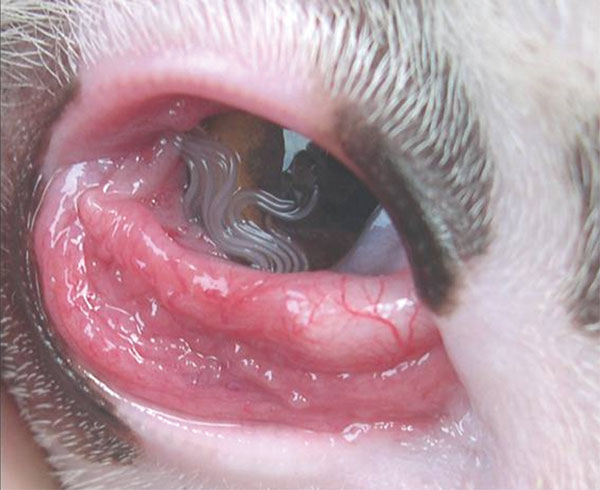
| Thelazia callipaeda infestation in a dog[1] | |
| Specialty | Infectious disease |
Thelaziasis is the term for infestation with parasitic nematodes of the genus Thelazia. The adults of all Thelazia species discovered so far inhabit the eyes and associated tissues (such as eyelids, tear ducts, etc.) of various mammal and bird hosts, including humans.[2] Thelazia nematodes are often referred to as "eyeworms".
Signs and symptoms
[edit]In animal and human hosts, infestation by Thelazia may be asymptomatic, though it frequently causes watery eyes (epiphora), conjunctivitis, corneal opacity, or corneal ulcers (ulcerative keratitis).[1] Infested humans have also reported "foreign body sensation" – the feeling that something is in the eye.[3][4]
Cause
[edit]Life cycle
[edit]In the uterus of the adult female, the embryos develop into first-stage larvae (L1), which remain in the eggshell (sheath).[5] The female deposits these sheathed larvae in the tears of the mammal or bird definitive host, and the larvae are ingested by tear-feeding flies. In the fly, the larvae "hatch" (exsheath), penetrate the gut wall, and migrate to either the fat body, testes or egg follicles (depending on the species). There they develop into third-stage larvae (L3), which migrate to the head of the fly. The infective L3 larvae wiggle out of the straw-like feeding apparatus of the fly when it feeds on the tears of another mammal or bird host. The L3 larvae develop into adults in the eye or surrounding tissues of the host, where they may live for over one year.
In the definitive host, Thelazia have been found in various tissues of the orbit (or socket) of the eye, including within the eyelids, in the tear glands, tear ducts, or the so-called "third eyelid" (nictitating membrane) or in the eyeball itself.[6]
Hosts and geographic range
[edit]While a few dozen species of Thelazia have been described in the literature, only three have been reported to infest humans, and only seven are commonly reported in veterinary contexts. The remaining species are occasionally found in birds or wild mammals.
In humans, dogs and cats, thelaziasis cases due to Thelazia callipaeda (Asia, Europe), and occasionally T. californiensis and T. gulosa (western North America), have been reported.[7]
Horses are infested by T. lacrymalis (worldwide) and, less frequently, by T. rhodesii (Africa, Asia, Europe).
In cattle, T. gulosa (Asia, Europe, North America), T. rhodesii (Africa, Asia, Europe) and T. skrjabini (Europe, North America) are the primary species of concern.
In camels, T. leesei infestations have been reported from the Post-Soviet states and India.
The intermediate hosts of several Thelazia species are known, and in each case they are tear-feeding flies of the genera Musca (family Muscidae), Phortica (family Drosophilidae), or Fannia (family Fanniidae).
Diagnosis
[edit]Diagnosis involves simply examining the eyes and nearby tissues for the worms. Adult Thelazia are very active; one author described T. californiensis as a "short lively piece of nylon fishing line about 10 mm long."[8]
Treatment
[edit]Because they live so close to the outside of the body, Thelazia is one of the few nematode infections which can be treated topically.
Topical treatment of livestock,[9] dogs and cats[10] with organophosphates (such as ecothiopate iodide or isofluorophate) and systemic treatment with anthelmintics (such as ivermectin, levamisole, and doramectin) are recommended by the Merck Veterinary Manual. Other sources have reported positive results treating dogs with moxidectin, imidacloprid, or milbemycin oxime.
For the treatment of human cases, removal of the worm is suggested. Topical treatment with cocaine or thiabendazole have also been reported to kill the worms in human cases.[8] Because most, if not all, species of Thelazia are spread by flies, sanitary practices which reduce the presence of flies will also reduce the spread of thelaziasis.
In canines, prevention against canine thelaziosis by monthly administrations of a combination of milbemycin oxime and afoxolaner (NexGard Spectra) has been found effective.[11]
Prevalence
[edit]By one author's count, 257 human cases of Thelazia callipaeda had been reported worldwide by the year 2000,[12] though thelaziasis is still considered to be a rare disease.
Various livestock and wildlife surveys suggest that thelaziasis is quite common among animals.
- A 1978 slaughterhouse survey in Guelph, Ontario, Canada found that about one-third (32%) of cattle over an eight-month period were infested with eyeworms.[13]
- A survey of horses in Kentucky revealed a 42% rate of infestation with Thelazia lacrymalis.[14]
- In Wyoming and Utah, a survey of hunter-harvested mule deer found 15% to be infested by Thelazia californiensis.[15]
- A survey of various sites in Italy found 23-60% of dogs, 5% of foxes and 4 out of 4 cats to be infested with Thelazia callipaeda.[16]
- In a study of dogs living in western Spain, 39.9% of the dogs were found to have Thelazia callipaeda worms living in their eyes.[17]
References
[edit]- ^ a b Otranto, D; Dutto, M (2008). "Human thelaziasis, Europe" (PDF). Emerging Infectious Diseases. 14 (4): 647–9. doi:10.3201/eid1404.071205. PMC 2570937. PMID 18394285.
- ^ Otranto, D; Traversa, D (2005). "Thelazia eyeworm: an original endo- and ecto-parasitic nematode". Trends in Parasitology. 21 (1): 1–4. doi:10.1016/j.pt.2004.10.008. PMID 15639731.
- ^ Sah, Ranjit; Khadka, Shusila; Adhikari, Mahesh; Niraula, Reema; Shah, Apoorva; Khatri, Anadi; Donovan, Suzanne (September 15, 2018). "Human Thelaziasis: Emerging Ocular Pathogen in Nepal". Open Forum Infectious Diseases. 5 (10) ofy237. doi:10.1093/ofid/ofy237. PMC 6189630. PMID 30349847.
- ^ "A Second Case of Human Conjunctival Infestation With Thelazia gulosa and a Review of T. gulosa in North America". academic.oup.com. 22 October 2019.
- ^ Anderson, R. C. (2000). "Thelazia". Nematode Parasites of Vertebrates: Their Development and Transmission (2nd ed.). Wallingford, England: CABI Publishing. pp. 404–407. ISBN 0-85199-421-0.
- ^ Shen, J; Gasser, RB; Chu, D; Wang, Z; Yuan, X; Cantacessi, C; Otranto, D (2006). "Human thelaziosis--a neglected parasitic disease of the eye". The Journal of Parasitology. 92 (4): 872–5. doi:10.1645/GE-823R.1. PMID 16995411. S2CID 22281032.
- ^ Bradbury; et al. (2018). "Case Report: Conjunctival Infestation with Thelazia gulosa: A Novel Agent of Human Thelaziasis in the United States". The American Journal of Tropical Medicine and Hygiene. 98 (4): 1171–1174. doi:10.4269/ajtmh.17-0870. PMC 5928835. PMID 29436343.
- ^ a b Chiapella, KJ (1974). "Letter: Verminous ophthalmia". The Western Journal of Medicine. 120 (6): 500. PMC 1130199. PMID 4546457.
- ^ "Merck Veterinary Manual".
- ^ "Merck Veterinary Manual".
- ^ Lebon, Wilfried; Guillot, Jacques; Álvarez, Maria-Jesús; Antonio Bazaga, José; Cortes-Dubly, Marie-Laure; Dumont, Pascal; Eberhardt, Marianne; Gómez, Héctor; Pennant, Olivier; Siméon, Noémie; Beugnet, Frederic; Halos, Lénaïg (2019). "Prevention of canine ocular thelaziosis (Thelazia callipaeda) with a combination of milbemycin oxime and afoxolaner (Nexgard Spectra®) in endemic areas in France and Spain". Parasite. 26: 1. doi:10.1051/parasite/2019001. ISSN 1776-1042. PMC 6333103. PMID 30644355.

- ^ Koyama, Y; Ohira, A; Kono, T; Yoneyama, T; Shiwaku, K (2000). "Five cases of thelaziasis". The British Journal of Ophthalmology. 84 (4): 441. doi:10.1136/bjo.84.4.439c. PMC 1723424. PMID 10777285.
- ^ Moolenbeek, WJ; Surgeoner, GA (1980). "Southern Ontario survey of eyeworms, Thelazia gulosa and Thelazia lacrymalis in cattle and larvae of Thelazia spp. in the face fly, Musca autumnalis". The Canadian Veterinary Journal. 21 (2): 50–2. PMC 1789681. PMID 7189135.
- ^ Lyons, ET; Swerczek, TW; Tolliver, SC; Bair, HD; Drudge, JH; Ennis, LE (2000). "Prevalence of selected species of internal parasites in equids at necropsy in central Kentucky (1995-1999)". Veterinary Parasitology. 92 (1): 51–62. doi:10.1016/S0304-4017(00)00266-1. PMID 10936545.
- ^ Dubay, SA; W, E; M, K; B, A (2000). "Bacteria and nematodes in the conjunctiva of mule deer from Wyoming and Utah". Journal of Wildlife Diseases. 36 (4): 783–7. doi:10.7589/0090-3558-36.4.783. PMID 11085445. S2CID 38137704.
- ^ Otranto, D; Ferroglio, E; Lia, RP; Traversa, D; Rossi, L (2003). "Current status and epidemiological observation of Thelazia callipaeda (Spirurida, Thelaziidae) in dogs, cats and foxes in Italy: a "coincidence" or a parasitic disease of the Old Continent?". Veterinary Parasitology. 116 (4): 315–25. doi:10.1016/j.vetpar.2003.07.022. PMID 14580802.
- ^ Miro, Guadalupe; Montoya, Ana; Hernandez, Leticia; Dado, Diana; Victoria Vazquez, Maria; Benito Manuel Villagrasa, Marta; Brianti, Emanuelle; Otranto, Domenico (27 July 2011). "Thelazia callipaeda: infection in dogs: a new parasite for Spain". Parasites & Vectors. 4 (148): 1756-3305. doi:10.1186/1756-3305-4-148. PMC 3158752. PMID 21791108.