Chorioamnionitis
View on WikipediaThis article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these messages)
|
| Chorioamnionitis | |
|---|---|
 | |
| Micrograph showing acute chorioamnionitis, with neutrophils in the chorion. Also seen are fibrin thrombi, which indicate a severe fetal inflammatory response.[1] H&E stain. | |
| Specialty | Obstetrics and gynaecology |
Chorioamnionitis, also known as amnionitis and intra-amniotic infection (IAI), is inflammation of the fetal membranes (amnion and chorion), usually due to bacterial infection.[1] In 2015, a National Institute of Child Health and Human Development Workshop expert panel recommended use of the term "triple I" to address the heterogeneity of this disorder. The term triple I refers to intrauterine infection or inflammation or both and is defined by strict diagnostic criteria, but this terminology has not been commonly adopted although the criteria are used.[2]
Chorioamnionitis results from an infection caused by bacteria ascending from the vagina into the uterus and is associated with premature or prolonged labor.[3] It triggers an inflammatory response to release various inflammatory signaling molecules, leading to increased prostaglandin and metalloproteinase release. These substances promote uterine contractions and cervical ripening, causations of premature birth.[4] The risk of developing chorioamnionitis increases with number of vaginal examinations performed in the final month of pregnancy, including labor.[5][6] Tobacco and alcohol use also puts mothers at risk for chorioamnionitis development.[7]
Chorioamnionitis is caught early by looking at signs and symptoms such as fever, abdominal pain, or abnormal vaginal excretion.[8] Administration of antibiotics if the amniotic sac bursts prematurely can prevent chorioamnionitis occurrence.[9]
Signs and symptoms
[edit]The signs and symptoms of clinical chorioamnionitis include fever, leukocytosis (>15,000 cells/mm3), maternal (>100 bpm)[10] or fetal (>160 bpm) tachycardia, uterine tenderness and preterm rupture of membranes.[2]
Causes
[edit]Causes of chorioamnionitis stem from bacterial infection as well as obstetric and other related factors.[3][7]
Microorganisms
[edit]Bacterial, viral, and even fungal infections can cause chorioamnionitis. Most commonly from Ureaplasma, Fusobacterium, and Streptococcus bacteria species. Less commonly, Gardnerella, Mycoplasma, and Bacteroides bacteria species. Sexually transmitted infections, chlamydia and gonorrhea, can cause development of the condition as well.[7] Studies are continuing to identify other microorganism classes and species as infection sources.[11]
Obstetric and other
[edit]Birthing-related events, lifestyle, and ethnic background have been linked to an increase in the risk of developing chorioamnionitis apart from bacterial causation.[11] Premature deliveries, ruptures of the amniotic sac membranes, prolonged labor, and primigravida childbirth are associated with this condition.[12] At term mothers who experience a combination of pre-labor membrane ruptures and multiple invasive vaginal examinations, prolonged labor, or have meconium appear in the amniotic fluid are at higher risk than at term mothers experiencing just one of those events.[11] In other studies, smoking, alcohol use and drug use are noted as risk factors. Those of African American ethnicity are noted to be at higher risk.[7][12]
Anatomy
[edit]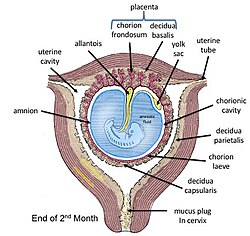
The amniotic sac consists of two parts:
- The outer membrane is the chorion. It is closest to the mother and physically supports the much thinner amnion.
- The chorion is the last and outermost of the membranes that make up the amniotic sac.[13]
- The inner membrane is the amnion. It is in direct contact with the amniotic fluid, which surrounds the fetus.
Diagnosis
[edit]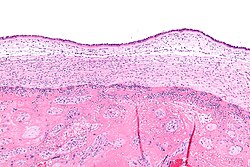

Pathologic
[edit]Chorioamnionitis is diagnosed from a histologic (tissue) examination of the fetal membranes.[12] Confirmed histologic chorioamnionitis without any clinical symptoms is termed subclinical chorioamnionitis and is more common than symptomatic clinical chorioamnionitis.[2]
Infiltration of the chorionic plate by neutrophils is diagnostic of (mild) chorioamnionitis. More severe chorioamnionitis involves subamniotic tissue and may have fetal membrane necrosis and/or abscess formation.[1]
Severe chorioamnionitis may be accompanied by vasculitis of the umbilical blood vessels due to the fetus' inflammatory cells. If very severe, funisitis, inflammation of the umbilical cord connective tissue, occurs.[12]
Suspected clinical diagnosis
[edit]The presence of fever between 38.0°C and 39.0°C alone is insufficient to indicate chorioamnionitis and is termed isolated maternal fever. Isolated maternal fever may not have an infectious cause and does not require antibiotic treatment.[2]
When intrapartum (during delivery) fever is higher than 39.0°C, suspected diagnosis of chorioamnionitis can be made. Alternatively, if intrapartum fever is between 38.0°C and 39.0°C, an additional risk factor must be present to make a presumptive diagnosis of chorioamnionitis. Additional risk factors include:[14]
- Fetal tachycardia
- Maternal leukocytosis (>15,000 cells/mm3)[15]
- Purulent cervical drainage
Confirmed diagnosis
[edit]Diagnosis is typically not confirmed until after delivery. However, people with confirmed diagnosis and suspected diagnosis have the same post-delivery treatment regardless of diagnostic status. Diagnosis can be confirmed histologically or through amniotic fluid tests such as gram staining, glucose levels, or other culture results consistent with infection.[14]
Prevention
[edit]If the amniotic sac breaks early into pregnancy, the potential of introducing bacteria in the amniotic fluid can increase. Administering antibiotics maternally can potentially prevent chorioamnionitis and allow for a longer pregnancy.[9] In addition, it has been shown that it is not necessary to deliver the fetus quickly after chorioamnionitis is diagnosed, so a C-section is not necessary unless maternal health concern is present.[12] However, research has found that beginning labor early at approximately 34 weeks can lessen the likelihood of fetal death, and reduce the potential for excessive infection within the mother.[12]
In addition, providers should interview people suspected to have chorioamnionitis about whether they are experiencing signs and symptoms at scheduled obstetrics visits during pregnancy, including whether the individual has experienced excretion vaginally, febrile, or abdominal pain.[8]
Treatment
[edit]The American College of Obstetricians and Gynecologists' Committee Opinion proposes the use of antibiotic treatment in intrapartum mothers with suspected or confirmed chorioamnionitis and maternal fever without an identifiable cause.[14]
Intrapartum antibiotic treatment consists of:[2]
- Standard
- Alternative
- Cesarean delivery
- Ampicillin and gentamicin plus either clindamycin or metronidazole
- Penicillin-allergy
- Vancomycin + gentamicin
- Gentamicin + clindamycin
However, there is not enough evidence to support the most efficient antimicrobial regimen.[16] Starting the treatment during the intrapartum period is more effective than starting it postpartum; it shortens the hospital stay for the mother and the neonate.[17] There is currently not enough evidence to dictate how long antibiotic therapy should last. Completion of treatment/cure is only considered after delivery.[2]
Supportive measures
[edit]Acetaminophen is often used for treating fevers and may be beneficial for fetal tachycardia. There can be increased likelihood for neonatal encephalopathy when mothers have intrapartum fever.[12]
Outcomes
[edit]Chorioamnionitis has possible associations with numerous neonatal conditions. Intrapartum (during labor) chorioamnionitis may be associated with neonatal pneumonia, meningitis, sepsis, and death. Long-term infant complications like bronchopulmonary dysplasia, cerebral palsy, and Wilson-Mikity syndrome have been associated to the bacterial infection.[14] Furthermore, histological chorioamnionitis may increase the likelihood of newborn necrotizing enterocolitis, where one or more sections of the bowel die. This occurs when the fetal gut barrier becomes compromised and is more susceptible to conditions like infection and sepsis.[18] In addition, chorioamnionitis can act as a risk factor for premature birth and periventricular leukomalacia.[19]
Complications
[edit]This section needs additional citations for verification. (July 2018) |
For mother and fetus, chorioamnionitis may lead to short-term and long-term issues when microbes move to different areas or trigger inflammatory responses due to infection.[12]
Maternal complications
[edit]- Higher risk for C-section
- Postpartum hemorrhage
- Endometritis[20]
- Bacteremia (often due to Group B streptococcus and Escherichia coli)[12]
- Pelvic abscess
Mothers with chorioamnionitis who undergo a C-section may be more likely to develop pelvic abscesses, septic pelvic thrombophlebitis, and infections at the surgical site.[11]
Fetal complications
[edit]Neonatal complications
[edit]- Perinatal death
- Asphyxia
- Early onset neonatal sepsis[21]
- Septic shock
- Neonatal pneumonia
- Infant respiratory distress
In the long-term, infants may be more likely to experience cerebral palsy or neurodevelopmental disabilities. Disability development is related to the activation of the fetal inflammatory response syndrome (FIRS) when the fetus is exposed to infected amniotic fluid or other foreign entities.[4][12] This systemic response results in neutrophil and cytokine release that can impair the fetal brain and other vital organs.[4][9] Compared to infants with clinical chorioamnionitis, it appears cerebral palsy may occur at a higher rate for those with histologic chorioamnionitis. However, more research needs to be done to examine this association.[22] There is also concern about the impact of FIRS on infant immunity as this is a critical time for growth and development. For instance, it may be linked to chronic inflammatory disorders, such as asthma.[23]
Epidemiology
[edit]Chorioamnionitis is approximated to occur in about 4%[8] of births in the United States. However, many other factors can increase the risk of chorioamnionitis. For example, in births with premature rupture of membranes (PROM), between 40 and 70% involve chorioamnionitis. Furthermore, clinical chorioamnionitis is implicated in 12% of all cesarean deliveries. Some studies have shown that the risk of chorioamnionitis is higher in those of African American ethnicity, those with immunosuppression, and those who smoke, use alcohol, or abuse drugs.[12]
See also
[edit]Notes
[edit]- ^ a b c d Kim CJ, Romero R, Chaemsaithong P, Chaiyasit N, Yoon BH, Kim YM (October 2015). "Acute chorioamnionitis and funisitis: definition, pathologic features, and clinical significance". American Journal of Obstetrics and Gynecology. 213 (4 Suppl): S29 – S52. doi:10.1016/j.ajog.2015.08.040. PMC 4774647. PMID 26428501.
- ^ a b c d e f Tita AT (2019). Berghella V, Barss BA (eds.). "Intra-amniotic infection (clinical chorioamnionitis or triple I)". UpToDate.
- ^ a b Cheng YW, Delaney SS, Hopkins LM, Caughey AB (November 2009). "The association between the length of first stage of labor, mode of delivery, and perinatal outcomes in women undergoing induction of labor". American Journal of Obstetrics and Gynecology. 201 (5): 477.e1–477.e7. doi:10.1016/j.ajog.2009.05.024. PMID 19608153.
- ^ a b c Galinsky R, Polglase GR, Hooper SB, Black MJ, Moss TJ (2013). "The consequences of chorioamnionitis: preterm birth and effects on development". Journal of Pregnancy. 2013 412831. doi:10.1155/2013/412831. PMC 3606792. PMID 23533760.
- ^ Seaward PG, Hannah ME, Myhr TL, Farine D, Ohlsson A, Wang EE, et al. (November 1997). "International Multicentre Term Prelabor Rupture of Membranes Study: evaluation of predictors of clinical chorioamnionitis and postpartum fever in patients with prelabor rupture of membranes at term". American Journal of Obstetrics and Gynecology. 177 (5): 1024–1029. doi:10.1016/s0002-9378(97)70007-3. PMID 9396886.
- ^ Gluck O, Mizrachi Y, Ganer Herman H, Bar J, Kovo M, Weiner E (April 2020). "The correlation between the number of vaginal examinations during active labor and febrile morbidity, a retrospective cohort study". BMC Pregnancy and Childbirth. 20 (1): 246. doi:10.1186/s12884-020-02925-9. PMC 7183634. PMID 32334543.
- ^ a b c d Sweeney EL, Dando SJ, Kallapur SG, Knox CL (January 2017). "The Human Ureaplasma Species as Causative Agents of Chorioamnionitis". Clinical Microbiology Reviews. 30 (1): 349–379. doi:10.1128/CMR.00091-16. PMC 5217797. PMID 27974410.
- ^ a b c Fowler JR, Simon LV (October 2019). "Chorioamnionitis.". StatPearls [Internet]. StatPearls Publishing. PMID 30335284.
- ^ a b c Ericson JE, Laughon MM (March 2015). "Chorioamnionitis: implications for the neonate". Clinics in Perinatology. 42 (1): 155–65, ix. doi:10.1016/j.clp.2014.10.011. PMC 4331454. PMID 25678002.
- ^ Sung JH, Choi SJ, Oh SY, Roh CR (March 2021). "Should the diagnostic criteria for suspected clinical chorioamnionitis be changed?". The Journal of Maternal-Fetal & Neonatal Medicine. 34 (5): 824–833. doi:10.1080/14767058.2019.1618822. PMID 31084245. S2CID 155091947.
- ^ a b c d Czikk MJ, McCarthy FP, Murphy KE (September 2011). "Chorioamnionitis: from pathogenesis to treatment". Clinical Microbiology and Infection. 17 (9): 1304–1311. doi:10.1111/j.1469-0691.2011.03574.x. PMID 21672080.
- ^ a b c d e f g h i j k Tita AT, Andrews WW (June 2010). "Diagnosis and management of clinical chorioamnionitis". Clinics in Perinatology. 37 (2): 339–354. doi:10.1016/j.clp.2010.02.003. PMC 3008318. PMID 20569811.
- ^ a b Betts JG, Young KA, Wise JA, Johnson E, Poe B, Kruse DH, et al. (25 April 2013). "Embryonic Development". Anatomy and Physiology. Houston, Texas: OpenStax. Retrieved 29 July 2020.
- ^ a b c d "Committee Opinion No. 712 Summary: Intrapartum Management of Intraamniotic Infection". Obstetrics and Gynecology. 130 (2): 490–492. August 2017. doi:10.1097/AOG.0000000000002230. PMID 28742671.
- ^ Zanella P, Bogana G, Ciullo R, Zambon A, Serena A, Albertin MA (June 2010). "[Chorioamnionitis in the delivery room]". Minerva Pediatrica. 62 (3 Suppl 1): 151–153. PMID 21090085.
- ^ Hopkins L, Smaill F (2002). "Antibiotic regimens for management of intraamniotic infection". The Cochrane Database of Systematic Reviews. 2014 (3) CD003254. doi:10.1002/14651858.CD003254. PMC 6669261. PMID 12137684.
- ^ Chapman E, Reveiz L, Illanes E, Bonfill Cosp X (December 2014). Reveiz L (ed.). "Antibiotic regimens for management of intra-amniotic infection". The Cochrane Database of Systematic Reviews. 2014 (12) CD010976. doi:10.1002/14651858.CD010976.pub2. PMC 10562955. PMID 25526426.
- ^ Been JV, Lievense S, Zimmermann LJ, Kramer BW, Wolfs TG (February 2013). "Chorioamnionitis as a risk factor for necrotizing enterocolitis: a systematic review and meta-analysis". The Journal of Pediatrics. 162 (2): 236–42.e2. doi:10.1016/j.jpeds.2012.07.012. PMID 22920508.
- ^ Wu YW, Colford JM (September 2000). "Chorioamnionitis as a risk factor for cerebral palsy: A meta-analysis". JAMA. 284 (11): 1417–1424. doi:10.1001/jama.284.11.1417. PMID 10989405.
- ^ Casey BM, Cox SM (March 1997). "Chorioamnionitis and endometritis". Infectious Disease Clinics of North America. 11 (1): 203–222. doi:10.1016/S0891-5520(05)70349-4. PMID 9067792.
- ^ Bersani I, Thomas W, Speer CP (April 2012). "Chorioamnionitis--the good or the evil for neonatal outcome?". The Journal of Maternal-Fetal & Neonatal Medicine. 25 (Suppl 1): 12–16. doi:10.3109/14767058.2012.663161. PMID 22309119. S2CID 11109172.
- ^ Shi Z, Ma L, Luo K, Bajaj M, Chawla S, Natarajan G, et al. (June 2017). "Chorioamnionitis in the Development of Cerebral Palsy: A Meta-analysis and Systematic Review". Pediatrics. 139 (6): e20163781. doi:10.1542/peds.2016-3781. PMC 5470507. PMID 28814548.
- ^ Sabic D, Koenig JM (January 2020). "A perfect storm: fetal inflammation and the developing immune system". Pediatric Research. 87 (2): 319–326. doi:10.1038/s41390-019-0582-6. PMC 7875080. PMID 31537013. S2CID 202702137.
References
[edit]External links
[edit]- Overview at Cleveland Clinic.
- Cerebral palsy inflammation link (29 November 2003) at BBC.
