Recent from talks
Nothing was collected or created yet.
Germinoma
View on Wikipedia| Germinoma | |
|---|---|
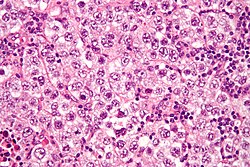 | |
| Micrograph of a germinoma, H&E stain | |
| Specialty | Oncology |
A germinoma is a type of germ-cell tumor,[1] which is not differentiated upon examination.[2] It may be benign or malignant.
Cause
[edit]Germinomas are thought to originate from an error of development, when certain primordial germ cells fail to migrate properly. Germinomas lack histologic differentiation, whereas nongerminomatous germ-cell tumors display a variety of differentiation. Like other germ-cell tumors, germinomas can undergo malignant transformation.[citation needed]
Histology
[edit]The tumor is uniform in appearance, consisting of large, round cells with vesicular nuclei and clear or finely granular cytoplasm that is eosinophilic. On gross examination, the external surface is smooth and bosselated (knobby), and the interior is soft, fleshy, and either cream-coloured, gray, pink, or tan. Microscopic examination typically reveals uniform cells that resemble primordial germ cells. Typically, the stroma contains lymphocytes, and about 20% of patients have sarcoid-like granulomas.[citation needed]
Diagnosis
[edit]Metastasis has been noted in about 22% of cases at time of diagnosis. Males are roughly twice as commonly affected in developing germinomas. They are most commonly diagnosed between the ages of 10 and 21.[citation needed]
Often, serum and spinal fluid tumor markers of alpha-fetoprotein and beta-HCG are tested. Pure germinomas are not associated with these markers. Nongerminomatous germ-cell tumors may be associated with increased markers such as alpha-fetoprotein with yolk sac tumors, as well as embryonic cell carcinomas and immature teratomas and beta-HCG, which occur in choriocarcinomas. In one to 15% of germinomas, a low level of beta-HCG may be produced. Although controversial, HCG-secreting germinomas may be more aggressive than nonsecreting ones.[citation needed]
Classification
[edit]The term "germinoma" most often refers to a tumor in the brain that has a histology identical to two other tumors, dysgerminoma in the ovary and seminoma in the testis.[3] Since 1994, MeSH has defined germinoma as "a malignant neoplasm of the germinal tissue of the gonads, mediastinum, or pineal region"[4] and within its scope included both dysgerminoma and seminoma. Collectively, these are the seminomatous or germinomatous tumors.[citation needed]
Locations
[edit]Ovary (dysgerminoma) and testis (seminoma)
[edit]Dysgerminoma is the most common type of malignant germ-cell ovarian cancer. Dysgerminoma usually occurs in adolescence and early adult life; about 5% occur in prepubertal children. Dysgerminoma is extremely rare after age 50. It occurs in both ovaries in 10% of patients and, in a further 10%, a microscopic tumor is in the other ovary.[citation needed]

Seminoma is the second-most common testicular cancer; the most common is mixed, which may contain seminoma.[citation needed]
Abnormal gonads (due to gonadal dysgenesis and androgen insensitivity syndrome) have a high risk of developing a dysgerminoma.[5] Most dysgerminomas are associated with elevated serum lactic dehydrogenase, which is sometimes used as a tumor marker. Metastases are most often present in the lymph nodes.[citation needed]
Intracranial germinoma
[edit]Intracranial germinoma occurs in 0.7 per million children.[6] As with other germ-cell tumors (GCTs) occurring outside the gonads, the most common location of intracranial germinoma is on or near the midline, often in the pineal or suprasellar areas; in 5-10% of patients with germinoma in either area, the tumor is in both areas. Like other GCTs, germinomas can occur in other parts of the brain. Within the brain, this tumor is most common in the hypothalamic or hypophyseal regions. In the thalamus and basal ganglia, germinoma is the most common GCT.[citation needed]
The diagnosis of an intracranial germinoma usually is based on biopsy, as the features on neuroimaging appear similar to other tumors.[citation needed]
Cytology of the cerebrospinal fluid often is studied to detect metastasis into the spine. This is important for staging and radiotherapy planning.
Intracranial germinomas have a reported 90% survival to five years after diagnosis.[7] Near total resection does not seem to influence the cure rate, so gross total resection is not necessary and can increase the risk of complications from surgery. The best results have been reported[citation needed] from craniospinal radiation with local tumor boost of greater than 4,000 centigray (cGy).
Treatment
[edit]Germinomas, like several other types of GCTs, are sensitive to both chemotherapy[8] and radiotherapy. For this reason, treatment with these methods can offer excellent chances of long-term survival, even cure.[citation needed]
Prognosis
[edit]Although chemotherapy can shrink germinomas, it is not generally recommended alone unless radiation has contraindications. In a study in the early 1990s, carboplatinum, etoposide, and bleomycin were given to 45 germinoma patients, and about half the patients relapsed. Most of these relapsed patients were then recovered with radiation or additional chemotherapy.[9]
See also
[edit]- Brown-Séquard syndrome (sections on cavernous malformation and germinoma)
- Spermatocytic tumor
References
[edit]- ^ "Germinoma" at Dorland's Medical Dictionary
- ^ Germinoma, Central Nervous System at eMedicine
- ^ "Pathology". Retrieved 2007-11-03.
- ^ Germinoma at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- ^ Sadler TW (2006). "Chapter 15". Langman's Medical Embryology (10th ed.). Lippincott, Williams & Wilkins. pp. 251–252.
- ^ Keene D, Johnston D, Strother D, Fryer C, Carret AS, Crooks B, et al. (May 2007). "Epidemiological survey of central nervous system germ cell tumors in Canadian children". Journal of Neuro-Oncology. 82 (3): 289–295. doi:10.1007/s11060-006-9282-2. PMID 17120159. S2CID 5872972.
- ^ Packer RJ, Cohen BH, Cooney K, Coney K (2000). "Intracranial germ cell tumors". The Oncologist. 5 (4): 312–320. doi:10.1634/theoncologist.2000-0312. PMID 10964999.
- ^ Ueba T, Yamashita K, Fujisawa I, Nakao S, Ooyama K, Yorihuji T, et al. (2007). "Long-term follow-up of 5 patients with intracranial germinoma initially treated by chemotherapy alone". Acta Neurochirurgica. 149 (9): 897–902, discussion 902. doi:10.1007/s00701-007-1268-0. PMID 17690837. S2CID 24074801.
- ^ Balmaceda C, Heller G, Rosenblum M, Diez B, Villablanca JG, Kellie S, et al. (November 1996). "Chemotherapy without irradiation--a novel approach for newly diagnosed CNS germ cell tumors: results of an international cooperative trial. The First International Central Nervous System Germ Cell Tumor Study". Journal of Clinical Oncology. 14 (11): 2908–2915. doi:10.1200/JCO.1996.14.11.2908. PMID 8918487.

