Recent from talks
Nothing was collected or created yet.
Mitochondrial disease
View on WikipediaThis article needs additional citations for verification. (February 2014) |
| Mitochondrial disease | |
|---|---|
| Other names | Mitochondrial cytopathy; mitochondriopathy (MCP) |
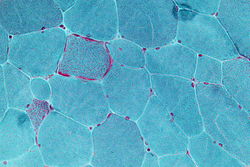 | |
| Micrograph showing ragged red fibers, a finding seen in various types of mitochondrial diseases. Muscle biopsy. Gomori trichrome stain. | |
| Specialty | Medical genetics |
Mitochondrial disease is a group of genetic disorders caused by mitochondrial dysfunction. Mitochondria are the organelles that generate energy for the cell and are found in every cell of the human body except red blood cells. They convert the energy of food molecules into the ATP that powers most cell functions.
Mitochondrial diseases take on unique characteristics both because of the way the diseases are often inherited and because mitochondria are so critical to cell function. A subclass of these diseases that have neuromuscular symptoms are known as mitochondrial myopathies.
Types
[edit]Mitochondrial disease can manifest in many different ways[1] whether in children[2] or adults.[3] Examples of mitochondrial diseases include:
- Mitochondrial myopathy[2][3]
- Maternally inherited diabetes mellitus and deafness (MIDD)[4]
- While diabetes mellitus and deafness can be found together for other reasons, at an early age this combination can be due to mitochondrial disease, as may occur in Kearns–Sayre syndrome and Pearson syndrome[2]
- Leber's hereditary optic neuropathy (LHON)[3]
- Leigh syndrome, subacute necrotizing encephalomyelopathy[6]
- after normal development the disease usually begins late in the first year of life, although onset may occur in adulthood
- a rapid decline in function occurs and is marked by seizures, altered states of consciousness, dementia, ventilatory failure
- Neuropathy, ataxia, retinitis pigmentosa, and ptosis (NARP)
- progressive symptoms as described in the acronym
- dementia
- Myoneurogenic gastrointestinal encephalopathy (MNGIE)
- gastrointestinal pseudo-obstruction
- neuropathy
- MERRF syndrome
- progressive myoclonic epilepsy
- "Ragged Red Fibers" are clumps of diseased mitochondria that accumulate in the subsarcolemmal region of the muscle fiber and appear when muscle is stained with modified Gömöri trichrome stain
- short stature
- hearing loss
- lactic acidosis
- exercise intolerance
- MELAS syndrome, mitochondrial encephalopathy, lactic acidosis, and stroke-like episodes
- Mitochondrial DNA depletion syndrome
- Cancer: While the cancer cells prefer Warburg effect however efficient mitochondrial maintenance remains crucial for tumor resilience, redox regulation, and avoidance of cell death. Tumor cells exhibit metabolic heterogeneity, shifting between glycolysis and mitochondrial oxidative phosphorylation to meet energy demands and support growth, especially during metastasis. Key bioenergetic parameters such as oxygen flux, proton leakage, membrane potential, extracellular acidification rate (ECAR), and proton-motive force are examined as “sub-domains” that tumors may regulate to promote survival and evade apoptosis.[7]
Conditions such as Friedreich's ataxia can affect the mitochondria but are not associated with mitochondrial proteins.
Presentation
[edit]Associated conditions
[edit]Acquired conditions in which mitochondrial dysfunction has been involved include:
- ALS[8]
- Alzheimer's disease,[9]
- Bipolar disorder,[10][11][12] schizophrenia, aging and senescence,[13] anxiety disorders[14]
- Cancer
- Cardiovascular disease
- Diabetes
- Huntington's disease
- Long Covid
- ME/CFS
- Parkinson's disease
- Sarcopenia[11]
The body, and each mutation, is modulated by other genome variants; the mutation that in one individual may cause liver disease might in another person cause a brain disorder. The severity of the specific defect may also be great or small. Some defects include exercise intolerance. Defects often affect the operation of the mitochondria and multiple tissues more severely, leading to multi-system diseases.[15]
It has also been reported that drug tolerant cancer cells have an increased number and size of mitochondria, which suggested an increase in mitochondrial biogenesis.[16] A recent study in Nature Nanotechnology has reported that cancer cells can hijack the mitochondria from immune cells via physical tunneling nanotubes.[17]
As a rule, mitochondrial diseases are worse when the defective mitochondria are present in the muscles, cerebrum, or nerves,[18] because these cells use more energy than most other cells in the body.
Although mitochondrial diseases vary greatly in presentation from person to person, several major clinical categories of these conditions have been defined, based on the most common phenotypic features, symptoms, and signs associated with the particular mutations that tend to cause them.[citation needed]
An outstanding question and area of research is whether ATP depletion or reactive oxygen species are in fact responsible for the observed phenotypic consequences.[citation needed]
Cerebellar atrophy or hypoplasia has sometimes been reported to be associated.[19]
Causes
[edit]Mitochondrial disorders may be caused by mutations (acquired or inherited), in mitochondrial DNA (mtDNA), or in nuclear genes that code for mitochondrial components. They may also be the result of acquired mitochondrial dysfunction due to adverse effects of drugs, infections, or other environmental causes.[20]

Nuclear DNA has two copies per cell (except for sperm and egg cells), one copy being inherited from the father and the other from the mother. Mitochondrial DNA, however, is inherited from the mother only (with some exceptions) and each mitochondrion typically contains between 2 and 10 mtDNA copies. During cell division the mitochondria segregate randomly between the two new cells. Those mitochondria make more copies, normally reaching 500 mitochondria per cell. As mtDNA is copied when mitochondria proliferate, they can accumulate random mutations, a phenomenon called heteroplasmy. If only a few of the mtDNA copies inherited from the mother are defective, mitochondrial division may cause most of the defective copies to end up in just one of the new mitochondria (for more detailed inheritance patterns, see human mitochondrial genetics). Mitochondrial disease may become clinically apparent once the number of affected mitochondria reaches a certain level; this phenomenon is called "threshold expression".
Mitochondria possess many of the same DNA repair pathways as nuclei do—but not all of them;[21] therefore, mutations occur more frequently in mitochondrial DNA than in nuclear DNA (see Mutation rate). This means that mitochondrial DNA disorders may occur spontaneously and relatively often. Defects in enzymes that control mitochondrial DNA replication (all of which are encoded for by genes in the nuclear DNA) may also cause mitochondrial DNA mutations.
Most mitochondrial function and biogenesis is controlled by nuclear DNA. Human mitochondrial DNA encodes 13 proteins of the respiratory chain, while most of the estimated 1,500 proteins and components targeted to mitochondria are nuclear-encoded. Defects in nuclear-encoded mitochondrial genes are associated with hundreds of clinical disease phenotypes including anemia, dementia, hypertension, lymphoma, retinopathy, seizures, and neurodevelopmental disorders.[22]
A study by Yale University researchers (published in the February 12, 2004, issue of the New England Journal of Medicine) explored the role of mitochondria in insulin resistance among the offspring of patients with type 2 diabetes.[23] Other studies have shown that the mechanism may involve the interruption of the mitochondrial signaling process in body cells (intramyocellular lipids). A study conducted at the Pennington Biomedical Research Center in Baton Rouge, Louisiana[24] showed that this, in turn, partially disables the genes that produce mitochondria.
Mechanisms
[edit]The effective overall energy unit for the available body energy is referred to as the daily glycogen generation capacity,[25][26][27] and is used to compare the mitochondrial output of affected or chronically glycogen-depleted individuals to healthy individuals.[26]
The glycogen generation capacity is entirely dependent on, and determined by, the operating levels of the mitochondria in all of the cells of the human body;[28] however, the relation between the energy generated by the mitochondria and the glycogen capacity is very loose and is mediated by many biochemical pathways.[25] The energy output of full healthy mitochondrial function can be predicted exactly by a complicated theoretical argument, but this argument is not straightforward, as most energy is consumed by the brain and is not easily measurable.
Diagnosis
[edit]Mitochondrial diseases are usually detected by analysing muscle samples, where the presence of these organelles is higher. The most common tests for the detection of these diseases are:
- Southern blot to detect large deletions or duplications
- Polymerase chain reaction and specific mutation testing[29]
- Sequencing
Treatments
[edit]Although research is ongoing, treatment options are currently limited; vitamins are frequently prescribed, though the evidence for their effectiveness is limited.[30] Pyruvate has been proposed in 2007 as a treatment option.[31] N-acetyl cysteine reverses many models of mitochondrial dysfunction.[32]
Mood disorders
[edit]In the case of mood disorders, specifically bipolar disorder, it is hypothesized that N-acetyl-cysteine (NAC), acetyl-L-carnitine (ALCAR), S-adenosylmethionine (SAMe), coenzyme Q10 (CoQ10), alpha-lipoic acid (ALA), creatine monohydrate (CM), and melatonin could be potential treatment options.[12]
Gene therapy prior to conception
[edit]Mitochondrial replacement therapy (MRT), where the nuclear DNA is transferred to another healthy egg cell leaving the defective mitochondrial DNA behind, is an IVF treatment procedure.[33] Using a similar pronuclear transfer technique, researchers at Newcastle University led by Douglass Turnbull successfully transplanted healthy DNA in human eggs from women with mitochondrial disease into the eggs of women donors who were unaffected.[34][35] In such cases, ethical questions have been raised regarding biological motherhood, since the child receives genes and gene regulatory molecules from two different women. Using genetic engineering in attempts to produce babies free of mitochondrial disease is controversial in some circles and raises important ethical issues.[36][37] A male baby was born in Mexico in 2016 from a mother with Leigh syndrome using MRT.[38]
In September 2012 a public consultation was launched in the UK to explore the ethical issues involved.[39] Human genetic engineering was used on a small scale to allow infertile women with genetic defects in their mitochondria to have children.[40] In June 2013, the United Kingdom government agreed to develop legislation that would legalize the 'three-person IVF' procedure as a treatment to fix or eliminate mitochondrial diseases that are passed on from mother to child. The procedure could be offered from 29 October 2015 once regulations had been established.[41][42][43] Embryonic mitochondrial transplant and protofection have been proposed as a possible treatment for inherited mitochondrial disease, and allotopic expression of mitochondrial proteins as a radical treatment for mtDNA mutation load.
In June 2018 Australian Senate's Senate Community Affairs References Committee recommended a move towards legalising MRT. Research and clinical applications of MRT were overseen by laws made by federal and state governments. State laws were, for the most part, consistent with federal law. In all states, legislation prohibited the use of MRT techniques in the clinic, and except for Western Australia, research on a limited range of MRT was permissible up to day 14 of embryo development, subject to a license being granted. In 2010, the Hon. Mark Butler MP, then Federal Minister for Mental Health and Ageing, had appointed an independent committee to review the two relevant acts: the Prohibition of Human Cloning for Reproduction Act 2002 and the Research Involving Human Embryos Act 2002. The committee's report, released in July 2011, recommended the existing legislation remain unchanged
Currently, human clinical trials are underway at GenSight Biologics (ClinicalTrials.gov # NCT02064569) and the University of Miami (ClinicalTrials.gov # NCT02161380) to examine the safety and efficacy of mitochondrial gene therapy in Leber's hereditary optic neuropathy.
Epidemiology
[edit]About 1 in 4,000 children in the United States will develop mitochondrial disease by the age of 10 years. Up to 4,000 children per year in the US are born with a type of mitochondrial disease.[44] Because mitochondrial disorders contain many variations and subsets, some particular mitochondrial disorders are very rare.
The average number of births per year among women at risk for transmitting mtDNA disease is estimated to approximately 150 in the United Kingdom and 800 in the United States.[45]
History
[edit]The first pathogenic mutation in mitochondrial DNA was identified in 1988; from that time to 2016, around 275 other disease-causing mutations were identified.[46]
Notable cases
[edit]Notable people with mitochondrial disease include:
- Mattie Stepanek, a poet, peace advocate, and motivational speaker who had dysautonomic mitochondrial myopathy, and who died at age 13.[47]
- Rocco Baldelli, a coach and former center fielder in Major League Baseball who had to retire from active play at age 29 due to mitochondrial channelopathy.
- Charlie Gard, a British boy who had mitochondrial DNA depletion syndrome; decisions about his care were taken to various law courts.
- Charles Darwin, a nineteenth century naturalist who suffered from a disabling illness, is speculated to have MELAS syndrome.[48]
References
[edit]- ^ "Mitochondrial Diseases". medlineplus.gov. Retrieved 2023-03-15.
- ^ a b c d Rahman S (2020). "Mitochondrial disease in children". Journal of Internal Medicine. 287 (6): 609–633. doi:10.1111/joim.13054. PMID 32176382.
- ^ a b c La Morgia C, Maresca A, Caporali L, Valentino ML, Carelli V (2020). "Mitochondrial diseases in adults". Journal of Internal Medicine. 287 (6): 592–608. doi:10.1111/joim.13064. PMID 32463135.
- ^ Tsang SH, Aycinena AR, Sharma T (2018). "Mitochondrial disorder: maternally inherited diabetes and deafness". Atlas of Inherited Retinal Diseases. Advances in Experimental Medicine and Biology. Vol. 1085. pp. 163–5. doi:10.1007/978-3-319-95046-4_31. ISBN 978-3-319-95045-7. PMID 30578504.
- ^ Shamsnajafabadi H, MacLaren RE, Cehajic-Kapetanovic J (2023). "Current and future landscape in genetic therapies for Leber hereditary optic neuropathy". Cells. 12 (15): 2013. doi:10.3390/cells12152013. PMC 10416882. PMID 37566092.
- ^ Rahman S (2023). "Leigh syndrome". Mitochondrial Diseases. Handbook of Clinical Neurology. Vol. 194. pp. 43–63. doi:10.1016/B978-0-12-821751-1.00015-4. ISBN 9780128217511. PMID 36813320.
- ^ Singh, Tashvinder; Sharma, Kangan; Jena, Laxmipriya; Kaur, Prabhsimran; Singh, Sandeep; Munshi, Anjana (2024-11-01). "Mitochondrial bioenergetics of breast cancer". Mitochondrion. 79 101951. doi:10.1016/j.mito.2024.101951. ISSN 1567-7249. PMID 39218051.
- ^ Muyderman, H; Chen, T (April 2014). "Mitochondrial dysfunction in amyotrophic lateral sclerosis – a valid pharmacological target?". British Journal of Pharmacology. 171 (8): 2191–2205. doi:10.1111/bph.12476. PMC 3976630. PMID 24148000.
- ^ Abyadeh, Morteza; Gupta, Vivek; Chitranshi, Nitin; Gupta, Veer; Wu, Yunqi; Saks, Danit; WanderWall, Roshana; Fitzhenry, Matthew J; Basavarajappa, Devaraj; You, Yuyi; H Hosseini, Ghasem; A Haynes, Paul; L Graham, Stuart; Mirzaei, Mehdi (2021). "Mitochondrial dysfunction in Alzheimer's disease - a proteomics perspective". Expert Review of Proteomics. 18 (4): 295–304. doi:10.1080/14789450.2021.1918550. PMID 33874826. S2CID 233310698.
- ^ Stork, C; Renshaw, P F (2005). "Mitochondrial dysfunction in bipolar disorder: Evidence from magnetic resonance spectroscopy research". Molecular Psychiatry. 10 (10): 900–19. doi:10.1038/sj.mp.4001711. PMID 16027739.
- ^ a b Pieczenik, Steve R; Neustadt, John (2007). "Mitochondrial dysfunction and molecular pathways of disease". Experimental and Molecular Pathology. 83 (1): 84–92. doi:10.1016/j.yexmp.2006.09.008. PMID 17239370.
- ^ a b Nierenberg, Andrew A; Kansky, Christine; Brennan, Brian P; Shelton, Richard C; Perlis, Roy; Iosifescu, Dan V (2012). "Mitochondrial modulators for bipolar disorder: A pathophysiologically informed paradigm for new drug development". Australian & New Zealand Journal of Psychiatry. 47 (1): 26–42. doi:10.1177/0004867412449303. PMID 22711881. S2CID 22983555.
- ^ Valiente-Pallejà, A; Tortajada, J; Bulduk, BK (2022). "Comprehensive summary of mitochondrial DNA alterations in the postmortem human brain: A systematic review". eBioMedicine. 76 (103815) 103815. doi:10.1016/j.ebiom.2022.103815. PMC 8790490. PMID 35085849.
- ^ Misiewicz, Zuzanna; Iurato, Stella; Kulesskaya, Natalia; Salminen, Laura; Rodrigues, Luis; Maccarrone, Giuseppina; Martins, Jade; Czamara, Darina; Laine, Mikaela A.; Sokolowska, Ewa; Trontti, Kalevi; Rewerts, Christiane; Novak, Bozidar; Volk, Naama; Park, Dong Ik; Jokitalo, Eija; Paulin, Lars; Auvinen, Petri; Voikar, Vootele; Chen, Alon; Erhardt, Angelika; Turck, Christoph W.; Hovatta, Iiris (26 September 2019). "Multi-omics analysis identifies mitochondrial pathways associated with anxiety-related behavior". PLOS Genetics. 15 (9) e1008358. doi:10.1371/journal.pgen.1008358. PMC 6762065. PMID 31557158.
- ^ Nunnari J, Suomalainen A (2012). "Mitochondria: in sickness and in health". Cell. 148 (6): 1145–59. doi:10.1016/j.cell.2012.02.035. PMC 5381524. PMID 22424226.
- ^ Goldman A, Khiste S, Freinkman E, Dhawan A, Majumder B, Mondal J, et al. (August 2019). "Targeting tumor phenotypic plasticity and metabolic remodeling in adaptive cross-drug tolerance". Science Signaling. 12 (595). doi:10.1126/scisignal.aas8779. PMC 7261372. PMID 31431543.
- ^ Saha T, Dash C, Jayabalan R, et al. (2021). "Intercellular nanotubes mediate mitochondrial trafficking between cancer and immune cells". Nat. Nanotechnol. 17 (1): 98–106. doi:10.1038/s41565-021-01000-4. PMC 10071558. PMID 34795441. S2CID 244349825.
- ^ Finsterer, Josef (2007). "Hematological Manifestations of Primary Mitochondrial Disorders". Acta Haematologica. 118 (2): 88–98. doi:10.1159/000105676. PMID 17637511. S2CID 24222021.
- ^ Lax, Nichola Zoe; Hepplewhite, Philippa Denis; Reeve, Amy Katherine; Nesbitt, Victoria; McFarland, Robert; Jaros, Evelyn; Taylor, Robert William; Turnbull, Douglass Matthew (2012). "Cerebellar Ataxia in Patients with Mitochondrial DNA Disease". Journal of Neuropathology & Experimental Neurology. 71 (2): 148–61. doi:10.1097/NEN.0b013e318244477d. PMC 3272439. PMID 22249460.
- ^ "Mitochondrial diseases". MeSH. Retrieved 2 August 2019.
- ^ Alexeyev M, Shokolenko I, Wilson G, LeDoux S (May 2013). "The maintenance of mitochondrial DNA integrity--critical analysis and update". Cold Spring Harbor Perspectives in Biology. 5 (5) a012641. doi:10.1101/cshperspect.a012641. PMC 3632056. PMID 23637283.
- ^ Scharfe C, Lu HH, Neuenburg JK, Allen EA, Li GC, Klopstock T, Cowan TM, Enns GM, Davis RW (2009). Rzhetsky A (ed.). "Mapping gene associations in human mitochondria using clinical disease phenotypes". PLOS Comput Biol. 5 (4) e1000374. Bibcode:2009PLSCB...5E0374S. doi:10.1371/journal.pcbi.1000374. PMC 2668170. PMID 19390613.
- ^ Petersen, Kitt Falk; Dufour, Sylvie; Befroy, Douglas; Garcia, Rina; Shulman, Gerald I. (12 February 2004). "Impaired Mitochondrial Activity in the Insulin-Resistant Offspring of Patients with Type 2 Diabetes". New England Journal of Medicine. 350 (7): 664–671. doi:10.1056/NEJMoa031314. PMC 2995502. PMID 14960743.
- ^ Sparks, Lauren M.; Xie, Hui; Koza, Robert A.; Mynatt, Randall; Hulver, Matthew W.; Bray, George A.; Smith, Steven R. (July 2005). "A High-Fat Diet Coordinately Downregulates Genes Required for Mitochondrial Oxidative Phosphorylation in Skeletal Muscle". Diabetes. 54 (7): 1926–33. doi:10.2337/diabetes.54.7.1926. PMID 15983191. Gale A134380159 ProQuest 216493144.
- ^ a b Mitchell, Peter. "David Keilin's respiratory chain concept and its chemiosmotic consequences" (PDF). Nobel institute.
- ^ a b Michelakis, Evangelos (January 2007). "A Mitochondria-K+ Channel Axis Is Suppressed in Cancer and Its Normalization Promotes Apoptosis and Inhibits Cancer Growth". University of Alberta. 11 (1). University of Alberta, 2007: 37–51. doi:10.1016/j.ccr.2006.10.020. PMID 17222789.
- ^ Lorini, M; Ciman, M (1962). "Hypoglycaemic action of Diisopropylammonium salts in experimental diabetes". Institute of Biochemistry, University of Padua, September 1962. 11 (9). Biochemical Pharmacology: 823–7. doi:10.1016/0006-2952(62)90177-6. PMID 14466716.
- ^ Stacpoole PW, Henderson GN, Yan Z, James MO (1998). "Clinical pharmacology and toxicology of dichloroacetate". Environ. Health Perspect. 106 (Suppl 4): 989–94. Bibcode:1998EnvHP.106S.989S. doi:10.1289/ehp.98106s4989. PMC 1533324. PMID 9703483.
- ^ Bulduk, Bengisu Kevser; Kiliç, Hasan Basri; Bekircan-Kurt, Can Ebru; Haliloğlu, Göknur; Erdem Özdamar, Sevim; Topaloğlu, Haluk; Kocaefe, Y. Çetin (March 2020). "A Novel Amplification-Refractory Mutation System-PCR Strategy to Screen MT-TL1 Pathogenic Variants in Patient Repositories". Genetic Testing and Molecular Biomarkers. 24 (3): 165–170. doi:10.1089/gtmb.2019.0079. PMID 32167396. S2CID 212693790.
- ^ Marriage B, Clandinin MT, Glerum DM (2003). "Nutritional cofactor treatment in mitochondrial disorders". J Am Diet Assoc. 103 (8): 1029–38. doi:10.1016/S0002-8223(03)00476-0. PMID 12891154.
- ^ Tanaka M, Nishigaki Y, Fuku N, Ibi T, Sahashi K, Koga Y (2007). "Therapeutic potential of pyruvate therapy for mitochondrial diseases". Mitochondrion. 7 (6): 399–401. doi:10.1016/j.mito.2007.07.002. PMID 17881297.
- ^ Frantz MC, Wipf P (Jun 2010). "Mitochondria as a target in treatment". Environ Mol Mutagen. 51 (5): 462–75. Bibcode:2010EnvMM..51..462F. doi:10.1002/em.20554. PMC 2920596. PMID 20175113.
- ^ Tachibana M, Sparman M, Sritanaudomchai H, Ma H, Clepper L, Woodward J, Li Y, Ramsey C, Kolotushkina O, Mitalipov S (September 2009). "Mitochondrial gene replacement in primate offspring and embryonic stem cells". Nature. 461 (7262): 367–372. Bibcode:2009Natur.461..367T. doi:10.1038/nature08368. PMC 2774772. PMID 19710649.
- ^ Boseley, Sarah (2010-04-14). "Scientists reveal gene-swapping technique to thwart inherited diseases". Guardian. London.
- ^ Craven, Lyndsey; Tuppen, Helen A.; Greggains, Gareth D.; Harbottle, Stephen J.; Murphy, Julie L.; Cree, Lynsey M.; Murdoch, Alison P.; Chinnery, Patrick F.; Taylor, Robert W.; Lightowlers, Robert N.; Herbert, Mary; Turnbull, Douglass M. (2010). "Pronuclear transfer in human embryos to prevent transmission of mitochondrial DNA disease". Nature. 465 (7294): 82–85. Bibcode:2010Natur.465...82C. doi:10.1038/nature08958. PMC 2875160. PMID 20393463.

- ^ "UK urged to permit IVF procedure to prevent fatal genetic diseases". The Guardian. London. 2015-04-30.
- ^ "Three parent baby law is 'irresponsible' says Church of England ahead of vote". The Telegraph. London. 2015-04-30.
- ^ Hamzelou, Jessica (2016-09-27). "Exclusive: World's first baby born with new "3 parent" technique". New Scientist. Retrieved 2016-11-26.
- ^ Sample, Ian (2012-09-17). "Regulator to consult public over plans for new fertility treatments". The Guardian. London. Retrieved 8 October 2012.
- ^ "Genetically altered babies born". BBC News. 2001-05-04. Retrieved 2008-04-26.
- ^ The Human Fertilisation and Embryology (Mitochondrial Donation) Regulations 2015 No. 572
- ^ "UK government backs three-person IVF". BBC News. 27 June 2013.
- ^ Knapton, Sarah (1 March 2014) 'Three-parent babies' could be born in Britain next year The Daily Telegraph Science News, Retrieved 1 March 2014
- ^ The Mitochondrial and Metabolic Disease Center
- ^ Gorman, Gráinne S.; Grady, John P.; Ng, Yi; Schaefer, Andrew M.; McNally, Richard J.; Chinnery, Patrick F.; Yu-Wai-Man, Patrick; Herbert, Mary; Taylor, Robert W.; McFarland, Robert; Turnbull, Doug M. (26 February 2015). "Mitochondrial Donation — How Many Women Could Benefit?". New England Journal of Medicine. 372 (9): 885–7. doi:10.1056/NEJMc1500960. PMC 4481295. PMID 25629662.
- ^ Claiborne, A.; English, R.; Kahn, J. (2016). "Etiology, Clinical Manifestation, and Diagnosis". In Claiborne, Anne; English, Rebecca; Kahn, Jeffrey (eds.). Mitochondrial Replacement Techniques. p. 37. doi:10.17226/21871. ISBN 978-0-309-38870-2. PMID 27054230.
- ^ "Young poet, peace advocate Mattie dies | the Spokesman-Review". 23 June 2004.
- ^ Hayman, John (May 2013). "Charles Darwin's Mitochondria". Genetics. 194 (1): 21–25. doi:10.1534/genetics.113.151241. PMC 3632469. PMID 23633139.
External links
[edit]Mitochondrial disease
View on GrokipediaDefinition and Overview
Core Pathophysiological Features
Mitochondrial diseases primarily result from dysfunction of the mitochondrial respiratory chain (MRC), which constitutes the core of oxidative phosphorylation (OXPHOS) and ATP production. The MRC comprises five enzyme complexes (I-V), with 13 essential subunits encoded by mitochondrial DNA (mtDNA) and over 70 additional polypeptides, including assembly factors, encoded by nuclear DNA (nDNA). Mutations in either genome disrupt electron transport, proton gradient formation, and ATP synthase activity, leading to cellular energy deficits particularly in tissues reliant on aerobic metabolism, such as muscle, brain, and heart.[1][4] A hallmark of mtDNA-related disorders is heteroplasmy, the coexistence of mutant and wild-type mtDNA within cells, with proportions varying across tissues and individuals. The threshold effect dictates that clinical and biochemical abnormalities emerge only when the mutant mtDNA fraction surpasses a critical level, typically 60-90%, beyond which compensatory mechanisms fail and OXPHOS capacity drops precipitously. This tissue-specific threshold explains variable phenotypic expression, as high-energy-demand cells tolerate lower mutation loads less effectively.[5][1] Impaired OXPHOS also elevates reactive oxygen species (ROS) production, particularly from complexes I and III, causing oxidative damage to mtDNA, lipids, and proteins, which perpetuates a vicious cycle of mitochondrial deterioration. Additional mechanisms include disrupted mitochondrial dynamics—fusion, fission, and mitophagy—and altered calcium homeostasis, promoting apoptosis in affected cells. Chronic energy failure often triggers compensatory mitochondrial proliferation, manifesting histologically as ragged red fibers in skeletal muscle biopsies, reflecting subsarcolemmal accumulation of dysfunctional organelles.[5][4]
Primary vs. Secondary Forms
Primary mitochondrial diseases result from germline mutations in mitochondrial DNA (mtDNA) or nuclear DNA (nDNA) encoding proteins critical for oxidative phosphorylation (OXPHOS) or mitochondrial biogenesis, directly disrupting cellular energy production.[6] These mutations, affecting over 350 identified genes, lead to inherent defects in mitochondrial function, often manifesting as progressive, multisystem disorders with variable heteroplasmy in mtDNA cases—where mutant mtDNA load must exceed a tissue-specific threshold (typically 60-90%) to cause dysfunction.[7] [8] Inheritance patterns include maternal transmission for mtDNA mutations and Mendelian patterns (autosomal recessive, dominant, or X-linked) for nDNA variants, with de novo mutations accounting for some sporadic cases.[1] Secondary mitochondrial dysfunction, by contrast, arises indirectly from non-mitochondrial genetic defects, environmental insults, or acquired conditions that impair mitochondrial integrity without primary OXPHOS gene mutations.[9] Common causes include drug toxicities (e.g., nucleoside analogs like zidovudine), ischemia-reperfusion injury, infections, or comorbidities such as lysosomal storage disorders, where lysosomal pathology secondarily depletes mtDNA or disrupts fusion/fission dynamics.[10] [11] It frequently accompanies neurodegenerative diseases like Alzheimer's or Parkinson's, muscular dystrophies, or even cancer, where inflammation or metabolic stress elevates reactive oxygen species (ROS) and inhibits electron transport chain complexes.[12] Unlike primary forms, secondary dysfunction may be reversible upon addressing the root cause, such as withdrawing offending agents or managing inflammation.[9] The distinction between primary and secondary forms hinges on comprehensive evaluation, including genetic sequencing, muscle biopsy for ragged red fibers or enzyme assays, and exclusion of mimics via biochemical markers like elevated lactate.[1] Misclassification can delay targeted therapies—e.g., avoiding mitochondrial toxins in primary cases—or overlook treatable secondaries, underscoring the need for mutation-specific confirmation in primary disease via next-generation sequencing panels.[9] Peer-reviewed consensus emphasizes that while both share bioenergetic deficits, primary disorders exhibit higher mutation burdens in mitochondrial-targeted genes, whereas secondaries often involve broader proteomic disruptions.[6]Classification
By Genetic Origin
Mitochondrial diseases are primarily classified by the genetic origin of the causative mutations, which occur either in mitochondrial DNA (mtDNA) or nuclear DNA (nDNA). Mutations in mtDNA account for approximately 15-25% of primary mitochondrial disorders, while nDNA mutations predominate in the remainder, reflecting the nuclear genome's role in encoding the majority of mitochondrial proteins.[5][13] This distinction influences inheritance patterns, disease penetrance, and clinical heterogeneity due to phenomena like heteroplasmy in mtDNA disorders.[14] mtDNA mutations arise in the small, circular mitochondrial genome, which spans 16,569 base pairs and encodes 37 genes: 13 for oxidative phosphorylation (OXPHOS) complex subunits, 22 transfer RNAs (tRNAs), and 2 ribosomal RNAs (rRNAs). These mutations are maternally inherited because sperm contribute negligible mitochondria to the zygote, resulting in uniparental transmission from the oocyte; affected mothers transmit mutant mtDNA to all offspring, but clinical expression varies due to random segregation and heteroplasmy—the coexistence of mutant and wild-type mtDNA within cells.[15][16] Common mtDNA defects include point mutations (e.g., in tRNA genes causing syndromes like MERRF) and large-scale deletions (e.g., associated with Pearson syndrome or Kearns-Sayre syndrome), often arising de novo or somatically but propagating maternally when germline.[17] Heteroplasmy levels above a tissue-specific threshold (typically 60-90%) trigger dysfunction, explaining variable expressivity even within families.[5] In contrast, nDNA mutations affect over 1,700 genes that encode mitochondrial proteins imported into mitochondria, including OXPHOS assembly factors, metabolic enzymes, and mtDNA maintenance machinery (e.g., POLG for replication). These follow Mendelian inheritance: most (~75%) are autosomal recessive, requiring biallelic variants; fewer are autosomal dominant or X-linked, with the latter impacting hemizygous males more severely.[1][18] nDNA defects often disrupt multiple mitochondrial pathways, leading to syndromic presentations like Leigh syndrome when involving OXPHOS genes (e.g., SURF1 mutations) or mtDNA depletion syndromes from replication failures (e.g., TK2 variants).[19] Unlike mtDNA, nDNA mutations lack heteroplasmy but can exhibit incomplete penetrance due to modifier genes or environmental factors. Diagnostic genetic testing distinguishes these origins via targeted panels or whole-exome sequencing, with mtDNA analysis requiring quantitative assessment of heteroplasmy.[20][21]Syndromic and Non-Syndromic Types
Mitochondrial diseases are clinically classified into syndromic and non-syndromic forms based on the presence of characteristic, recognizable multi-system phenotypes versus more isolated or nonspecific presentations.[22] [23] Syndromic mitochondrial diseases feature well-defined constellations of symptoms affecting multiple organs, often with hallmark neurological, muscular, or metabolic signs that facilitate pattern recognition for diagnosis.[24] In contrast, non-syndromic forms lack these distinct syndromes, typically manifesting as isolated organ dysfunction or vague, progressive symptoms without fitting established clinical criteria, making them more prevalent yet diagnostically challenging.[23] [25] Prominent syndromic examples include mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS), characterized by recurrent stroke-like episodes, seizures, and lactic acidosis often due to mitochondrial DNA (mtDNA) mutations like m.3243A>G; myoclonic epilepsy with ragged-red fibers (MERRF), marked by myoclonus, epilepsy, and muscle biopsy findings of ragged-red fibers from mtDNA point mutations such as m.8344A>G; and Leigh syndrome, a subacute necrotizing encephalomyelopathy with bilateral basal ganglia lesions, psychomotor regression, and respiratory failure, linked to defects in over 80 genes affecting respiratory chain complexes.[1] [12] Kearns-Sayre syndrome represents another, involving progressive external ophthalmoplegia, pigmentary retinopathy, and cardiac conduction blocks before age 20, typically from large mtDNA deletions.[2] These syndromes often exhibit maternal inheritance due to mtDNA involvement, though nuclear gene defects can also contribute.[3] Non-syndromic mitochondrial diseases, comprising the majority of cases, present without syndromic specificity, such as isolated mitochondrial myopathies with exercise intolerance and proximal weakness but no encephalopathy or epilepsy, or pure optic neuropathies like Leber hereditary optic neuropathy (LHON) confined to acute vision loss from mtDNA mutations (e.g., m.11778G>A) without broader systemic features.[23] [26] Other examples include nonsyndromic sensorineural hearing loss or cardiomyopathy attributable to mitochondrial dysfunction, where symptoms progress insidiously and require biochemical confirmation like elevated lactate or muscle enzyme defects rather than clinical pattern matching.[27] [25] This category's heterogeneity stems from diverse nuclear or mtDNA variants disrupting oxidative phosphorylation in a tissue-restricted manner, often necessitating genetic testing for confirmation since phenotypes overlap with non-mitochondrial conditions.[1]Specific Disorders
Leigh syndrome, also known as subacute necrotizing encephalomyelopathy, is a severe, progressive neurodegenerative mitochondrial disorder typically presenting in infancy or early childhood with symptoms including hypotonia, developmental regression, seizures, lactic acidosis, and characteristic bilateral symmetric lesions in the basal ganglia and brainstem on neuroimaging.[28] It arises from defects in mitochondrial oxidative phosphorylation, often due to mutations in nuclear genes encoding respiratory chain subunits or assembly factors, or in about 20% of cases, maternally inherited mitochondrial DNA (mtDNA) variants affecting complex I, IV, or V; prevalence is estimated at 1 in 40,000 live births.[29] Prognosis is poor, with most affected individuals succumbing before age five due to respiratory or cardiac failure, though supportive treatments like ketogenic diet may mitigate symptoms in select cases.[30] MELAS syndrome (mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes) manifests primarily in childhood or young adulthood with recurrent stroke-like episodes causing hemiparesis, cortical blindness, or aphasia, alongside chronic progressive external ophthalmoplegia, proximal myopathy, diabetes mellitus, and elevated serum lactate levels.[31] Over 80% of cases stem from the mtDNA point mutation m.3243A>G in the MT-TL1 gene, impairing tRNA function and mitochondrial protein synthesis, with maternal inheritance; lactic acidosis results from impaired ATP production and pyruvate metabolism.[32] Additional features include short stature, hearing loss, and cardiomyopathy, with episodes often triggered by illness or metabolic stress; while no curative therapy exists, arginine supplementation has shown efficacy in reducing stroke-like episode frequency in clinical studies.[33] MERRF syndrome (myoclonic epilepsy with ragged-red fibers) is characterized by myoclonic jerks, generalized epilepsy, cerebellar ataxia, and muscle weakness, with muscle biopsy revealing subsarcolemmal accumulations of abnormal mitochondria (ragged-red fibers) due to mtDNA mutations disrupting oxidative phosphorylation.[34] The predominant mutation, m.8344A>G in the MT-TK gene (accounting for ~80% of cases), impairs mitochondrial tRNA lysine and leads to maternal transmission; other symptoms encompass lipomas, optic atrophy, and dementia, with onset typically in late childhood or adolescence.[35] Disease severity varies heteroplasmically, correlating with mutation load in tissues; anticonvulsants like levetiracetam provide symptomatic epilepsy control, but progressive neurologic decline often results in wheelchair dependence by adulthood.[36] Kearns-Sayre syndrome (KSS) features onset before age 20 with progressive external ophthalmoplegia, ptosis, and pigmentary retinopathy, accompanied by cardiac conduction defects (e.g., complete heart block requiring pacemaker implantation), cerebellar ataxia, and elevated cerebrospinal fluid protein (>100 mg/dL).[37] It results from large-scale sporadic deletions in mtDNA (typically 2-8 kb, affecting multiple genes), disrupting replication and transcription without maternal inheritance; ragged-red fibers may appear on biopsy, reflecting compensatory mitochondrial proliferation.[38] Multisystem involvement includes endocrinopathies like hypoparathyroidism and sensorineural hearing loss; while coenzyme Q10 and idebenone supplementation support mitochondrial function, life expectancy is reduced primarily by cardiac arrhythmias.[39] Other notable syndromes include NARP (neuropathy, ataxia, retinitis pigmentosa), caused by mtDNA m.11778G>A or related variants impairing complex V, presenting with sensory neuropathy and visual loss, and Alpers-Huttenlocher syndrome, a nuclear POLG-related disorder with refractory epilepsy, liver failure, and mtDNA depletion.[1] These disorders underscore the heterogeneity of mitochondrial pathology, where tissue-specific energy demands dictate phenotypic expression.[26]Clinical Manifestations
Multi-System Symptoms
Mitochondrial diseases exhibit multi-system involvement because mitochondrial dysfunction impairs ATP production in energy-demanding tissues, leading to heterogeneous clinical presentations that can onset in infancy, childhood, or adulthood and progress variably. Common features include lactic acidosis, exercise intolerance, and episodic decompensation triggered by illness or stress, reflecting the reliance of organs like the brain, skeletal muscle, and heart on oxidative phosphorylation.[1][40] Neurological manifestations predominate due to the brain's high metabolic rate, encompassing fluctuating encephalopathy, seizures, migraines, stroke-like episodes, ataxia, spasticity, developmental delay or regression, dementia, and peripheral neuropathy; for instance, in MELAS syndrome, stroke-like episodes typically occur before age 40 and involve seizures and cognitive decline.[1][40][41] Neuromuscular symptoms include proximal muscle weakness, myalgia, cramps, chronic muscle spasms, fatigue, and exercise intolerance, often with elevated creatine kinase and ragged red fibers on biopsy; chronic progressive external ophthalmoplegia (CPEO) features ptosis and restricted eye movements, while myoclonus and ataxia occur in MERRF syndrome.[1][40][41][42] Sensory deficits frequently involve sensorineural hearing loss, optic atrophy, pigmentary retinopathy, and visual impairment, with deafness common in syndromes like KSS and MELAS, and retinopathy in NARP.[1][40] Cardiovascular involvement, reported in 18-21% of cases, manifests as hypertrophic or dilated cardiomyopathy, conduction blocks, arrhythmias, Wolff-Parkinson-White syndrome, or sudden cardiac death, particularly in MELAS and KSS where initial hypertrophy may progress to dilation.[43][44][40] Gastrointestinal symptoms arise from dysmotility and malabsorption, including delayed gastric emptying with nausea and vomiting, constipation, diarrhea, pseudo-obstruction, and fat intolerance; hepatic manifestations range from elevated transaminases to fulminant failure or cirrhosis, as in Alpers syndrome.[40][45] Endocrine disturbances feature diabetes mellitus resembling type 2 with impaired insulin secretion, short stature from growth hormone deficiency, and less commonly adrenal insufficiency or hypoparathyroidism, prevalent in KSS and Wolfram syndrome variants.[40][1] Renal complications include tubular acidosis, Fanconi-like syndrome, nephrotic syndrome, cysts, or progressive insufficiency, often with increased echogenicity on imaging and focal segmental glomerulosclerosis on histology, as seen in MELAS-associated cases.[46][41]Associated Conditions and Complications
Mitochondrial diseases frequently lead to multi-organ complications due to impaired cellular energy production, with neurological, cardiac, and endocrine systems being particularly vulnerable. Stroke-like episodes, distinct from vascular strokes, occur in syndromes like mitochondrial encephalopathy with lactic acidosis and stroke-like episodes (MELAS), resulting from metabolic crises rather than ischemia, and can cause focal neurological deficits. Seizures and encephalopathy are common, often exacerbated by infections or metabolic stress, contributing to developmental delays and cognitive impairment. Hearing loss affects up to 20-30% of patients, stemming from cochlear dysfunction, while optic atrophy leads to vision loss in conditions like Leber hereditary optic neuropathy variants.[47][1] Cardiac complications, including hypertrophic or dilated cardiomyopathy, arise in approximately 20-30% of primary mitochondrial disease cases, predisposing to heart failure, arrhythmias, and sudden death, particularly during acute decompensation. Respiratory muscle weakness can progress to ventilatory failure, a leading cause of mortality, often requiring mechanical support. Gastrointestinal dysmotility manifests as dysphagia, gastroparesis, pseudo-obstruction, or chronic diarrhea, complicating nutrition and increasing aspiration risk.[48][49][50] Endocrine dysfunction is prevalent, with diabetes mellitus occurring in 10-20% of patients due to pancreatic beta-cell failure, alongside growth hormone deficiency, adrenal insufficiency, hypogonadism, and parathyroid disorders leading to hypocalcemia. Renal involvement includes proximal tubulopathy or glomerulosclerosis, progressing to chronic kidney disease in severe cases. Hematologic abnormalities, such as sideroblastic anemia and cytopenias, affect 10-30% of individuals, linked to ineffective erythropoiesis from mitochondrial defects in bone marrow precursors. Hepatic failure and increased susceptibility to infections further compound morbidity, with multi-system crises often triggered by fever or fasting.[51][52][2]Pathogenesis
Genetic Causes
Mitochondrial diseases primarily arise from pathogenic variants in either mitochondrial DNA (mtDNA) or nuclear DNA (nDNA), disrupting oxidative phosphorylation (OXPHOS) and energy production. mtDNA, a 16.6 kb circular genome encoding 13 OXPHOS protein subunits, 22 tRNAs, and 2 rRNAs, is maternally inherited and prone to mutations due to its proximity to reactive oxygen species (ROS) and limited repair mechanisms. nDNA encodes approximately 1,500 proteins imported into mitochondria, including most OXPHOS components, assembly factors, and mtDNA maintenance genes, with mutations following Mendelian patterns. Over 500 genetic defects across these genomes have been linked to primary mitochondrial disorders (PMDs), with nuclear variants predominant in pediatric cases and mtDNA variants more common in adults.[19][1][53] mtDNA mutations include point mutations (e.g., m.3243A>G in MT-TL1 causing MELAS syndrome) and large-scale deletions/duplications (e.g., common in Pearson syndrome or Kearns-Sayre syndrome), often exhibiting heteroplasmy where mutant mtDNA coexists with wild-type in varying proportions. Heteroplasmy levels determine phenotypic severity via a threshold effect, with clinical manifestation typically requiring >70-90% mutant load in affected tissues. These mutations arise sporadically or inherit maternally, as sperm contribute negligible mtDNA; de novo events occur in ~1 in 200 healthy individuals, but pathogenic shifts amplify in offspring due to replicative segregation. mtDNA defects account for ~15-20% of PMDs, with prevalence estimates of 1 in 5,000 for mtDNA-related disease.[5][8][5] nDNA mutations, responsible for the majority of childhood-onset PMDs, affect genes for OXPHOS subunits (e.g., MT-ATP6 homologs), supercomplex assembly (e.g., TACO1), or mtDNA dynamics (e.g., POLG for polymerase gamma, causing Alpers-Huttenlocher syndrome). Inheritance is typically autosomal recessive (e.g., ~80% of nuclear PMDs), with autosomal dominant or X-linked rarer; examples include SURF1 variants in Leigh syndrome. Over 300 nuclear genes are implicated, often disrupting mitochondrial biogenesis or quality control. Unlike mtDNA, nDNA variants are biallelic in recessives, leading to complete loss-of-function in homozygotes or compounds.[18][14][3] Overall PMD prevalence exceeds 1 in 4,000-5,000 live births, with genetic confirmation via next-generation sequencing revealing nuclear defects in ~30% of suspected cases. Somatic mosaicism complicates diagnosis, as tissue-specific heteroplasmy or haploinsufficiency varies.[19][3][54]Biochemical and Cellular Mechanisms
Mitochondrial diseases primarily arise from defects in oxidative phosphorylation (OXPHOS), the process by which mitochondria generate adenosine triphosphate (ATP) through the electron transport chain (ETC) complexes I–IV and ATP synthase (complex V). These complexes transfer electrons from NADH and FADH₂ to oxygen, establishing a proton gradient that drives ATP synthesis. Mutations in mitochondrial DNA (mtDNA), encoding 13 essential OXPHOS subunits, or nuclear DNA (nDNA), encoding the majority of structural and assembly proteins, impair complex assembly, stability, or activity, leading to reduced ATP production.[5][55] Bioenergetic failure manifests as insufficient ATP in high-demand tissues such as muscle, brain, and heart, causing cellular dysfunction and lactic acidosis from pyruvate accumulation due to impaired NADH oxidation. Defective ETC function also promotes electron leakage, elevating mitochondrial reactive oxygen species (ROS) production, particularly from complexes I and III. Excessive ROS damages mtDNA, lipids, and proteins, perpetuating a cycle of mitochondrial deterioration and triggering oxidative stress.[5][56] At the cellular level, energy deficits and ROS induce mitochondrial permeability transition pore (mPTP) opening, cytochrome c release, and activation of apoptosis pathways. Impaired mitophagy fails to clear dysfunctional mitochondria, exacerbating accumulation. In skeletal muscle, compensatory proliferation of mitochondria accumulates subsarcolemmally, appearing as ragged red fibers on Gomori trichrome staining due to elevated succinate dehydrogenase activity, a hallmark of mitochondrial myopathies. This proliferation reflects an adaptive response to OXPHOS impairment but often fails to restore function.[5][55]Diagnosis
Clinical Assessment
Clinical assessment of mitochondrial disease requires a high index of suspicion for multi-system involvement, particularly in organs with high energy demands such as muscle, brain, heart, and sensory systems, as symptoms often manifest as unexplained progressive dysfunction across these domains.[41] Initial evaluation focuses on eliciting patterns of exercise intolerance, chronic fatigue, and episodic crises like stroke-like events or lactic acidosis, which differentiate mitochondrial disorders from isolated organ pathologies.[41] A detailed family history is essential, probing for maternal inheritance patterns, such as maternally transmitted diabetes, epilepsy, or myopathy, given the role of mitochondrial DNA in up to 20% of cases.[41] Medical history should systematically document onset (often childhood but variable), progression, and specific symptoms including proximal muscle weakness, myalgia, ptosis, external ophthalmoplegia (as in chronic progressive external ophthalmoplegia, affecting about 20% of adult-onset cases), ataxia, seizures, sensorineural hearing loss, retinopathy, cardiomyopathy, gastrointestinal dysmotility, and endocrine disturbances like diabetes.[41] In pediatric patients, delayed developmental milestones or regression may predominate, while adults more commonly present with milder, later-onset phenotypes.[41] Psychiatric symptoms, movement disorders, and sleep disturbances should also be screened, as they occur frequently and contribute to diagnostic delays.[57] Physical examination emphasizes neuromuscular evaluation, including assessment of muscle strength and endurance (e.g., via repeated fist-making or exercise tests), tone, reflexes, coordination, and gait for ataxia.[12] Ocular examination for ptosis, ophthalmoparesis, visual acuity, and fundoscopy is critical, alongside auscultation for cardiomyopathy or respiratory weakness.[57] A comprehensive neurological examination identifies peripheral neuropathy, central deficits, or multi-system signs like short stature or lipomas.[12] Standardized tools quantify disease burden and track progression during assessment. The Newcastle Mitochondrial Disease Adult Scale (NMDAS), validated in 2007 for patients over 16 years, evaluates current function, system-specific involvement (e.g., neurology, cardiology, endocrinology), and clinical status via a semiquantitative score, aiding in objective monitoring.[58] For children, the Newcastle Paediatric Mitochondrial Disease Scale assesses similar domains tailored to developmental stages.[59] These scales, encompassing domains like communication, mobility, and gastrointestinal function, support clinical decision-making by correlating with overall severity.[58]Laboratory and Imaging Techniques
Laboratory diagnosis of mitochondrial disease often begins with biochemical screening of blood and cerebrospinal fluid (CSF). Elevated lactate levels in plasma or CSF, typically measured via enzymatic assays, serve as a key initial marker, with concentrations exceeding 2.5 mmol/L at rest suggesting impaired oxidative phosphorylation, though normal levels do not exclude the diagnosis.[60] [61] Accompanying elevations in pyruvate, alanine, or a high lactate-to-pyruvate ratio further support suspicion, as these reflect NADH/NAD+ redox imbalances in affected tissues.[62] Urine organic acids and plasma amino acids are also assessed to detect patterns like increased fumarate or alanine, guiding toward mitochondrial dysfunction.[60] Tissue-based analyses provide more definitive evidence. Muscle biopsy remains a cornerstone, revealing characteristic histopathological features such as ragged red fibers (RRFs) on modified Gomori trichrome staining, which appear due to subsarcolemmal accumulation of dysfunctional mitochondria.[34] These RRFs, often quantified as greater than 2% of fibers, correlate with mitochondrial proliferation and are pathognomonic in syndromes like MERRF.[35] Electron microscopy may show paracrystalline inclusions, while histochemical stains identify cytochrome c oxidase (COX)-negative fibers, indicating respiratory chain defects.[41] Biochemical assays on biopsy homogenates measure activities of electron transport chain complexes I-V, with deficiencies confirming primary mitochondrial disorders when normalized to citrate synthase.[63] Imaging techniques complement laboratory findings, particularly for neurological involvement. Brain magnetic resonance imaging (MRI) frequently demonstrates symmetric T2 hyperintensities in the basal ganglia, brainstem, or white matter, with stroke-like lesions in conditions like MELAS lacking diffusion restriction or vascular distribution.[64] MR spectroscopy (MRS) detects elevated lactate peaks in affected regions, mirroring systemic biochemical abnormalities.[65] Computed tomography (CT) can identify basal ganglia calcifications, as seen in Leigh syndrome, while muscle MRI reveals fatty replacement or edema in proximal muscles, aiding in biopsy site selection.[66] These modalities enhance diagnostic specificity when integrated with clinical and genetic data.[67]Genetic Testing and Confirmation
Genetic testing is pivotal for confirming suspected mitochondrial diseases by identifying pathogenic variants in mitochondrial DNA (mtDNA) or nuclear DNA (nDNA) genes, typically following clinical evaluation and biochemical indicators such as elevated lactate levels.[1] Multigene panels or comprehensive genomic approaches like whole exome sequencing (WES) and whole genome sequencing (WGS) are standard, enabling analysis of both mtDNA and nDNA simultaneously.[1] These methods have diagnostic yields of approximately 20-25% in pediatric cohorts with oxidative phosphorylation deficiencies, surpassing traditional Sanger sequencing.[68] For mtDNA variants, which follow maternal inheritance and often manifest as heteroplasmy—the mixture of mutant and wild-type molecules—next-generation sequencing (NGS) of the full mitochondrial genome from blood is first-line, with tissue-specific sampling (e.g., muscle biopsy or urine sediment) recommended when blood heteroplasmy is low or undetectable due to the threshold effect.[20] [1] NGS detects heteroplasmic variants down to about 7.5% mutant load at 500x coverage depth, though challenges include interference from nuclear mitochondrial DNA segments (NUMTs) and variable tissue distribution.[20] [68] Confirmation involves orthogonal techniques like Sanger sequencing, digital PCR, or real-time PCR for precise quantification, particularly for common point mutations (e.g., m.3243A>G in MELAS syndrome).[20] nDNA testing targets over 300 genes linked to mitochondrial function, using NGS-based panels focused on oxidative phosphorylation, Krebs cycle, or phenotype-specific loci (e.g., POLG for polymerase gamma-related disorders), progressing to WES or WGS for broader coverage in unresolved cases.[20] [68] Blood is the preferred sample, as it is non-invasive and sufficient for germline variants.[20] Pathogenic autosomal recessive, dominant, or X-linked variants are interpreted per American College of Medical Genetics guidelines, with segregation analysis in families aiding confirmation.[1] Variants of uncertain significance (VUS) require additional scrutiny, potentially including functional assays (e.g., mitochondrial enzyme activities from muscle biopsy) or modeling in cybrids to assess pathogenicity, as genetic findings must align with clinical and biochemical evidence for definitive diagnosis.[20] [1] Multidisciplinary review, incorporating inheritance patterns and population databases like MitoMap, ensures accurate classification and guides counseling.[20]Management and Treatment
Supportive and Symptomatic Therapies
Supportive and symptomatic therapies represent the primary approach to managing mitochondrial diseases, emphasizing symptom alleviation, complication prevention, and quality-of-life improvement in the absence of curative options. These interventions rely on a multidisciplinary team, including neurologists, cardiologists, gastroenterologists, physiotherapists, occupational therapists, speech and language therapists, dietitians, and palliative care specialists, to address the multi-systemic nature of the disorders. Regular screening for comorbidities, such as cardiac arrhythmias or renal dysfunction, guides tailored care plans, with emergency protocols recommended to handle acute decompensations.[41][57] Rehabilitation therapies target neurological and musculoskeletal symptoms prominent in many mitochondrial diseases. Physical therapy focuses on optimizing muscle strength, preventing contractures, and enhancing mobility in cases of hypotonia, myopathy, or exercise intolerance, with programs individualized to patient tolerance to avoid excessive fatigue. Occupational therapy supports activities of daily living, while speech therapy addresses dysphagia and communication impairments, reducing aspiration risk through swallowing assessments and techniques. Evidence from expert consensus supports these interventions, though randomized trials remain limited; aerobic and resistance exercise training has shown benefits in improving skeletal muscle oxidative capacity and reducing fatigue in metabolic myopathies, prescribed cautiously based on cardiac evaluation.[41][57] Nutritional management is critical to counteract energy deficits and prevent catabolism, with dietitians recommending high-calorie, nutrient-dense diets tailored to gastrointestinal tolerance. Malnutrition screening every six months is advised, with gastrostomy tube placement considered for persistent dysphagia or failure to thrive to ensure adequate intake and hydration. Avoidance of fasting during illness is emphasized to mitigate metabolic crises.[41][57] Symptom-specific supportive measures include device-based interventions for organ involvement: cardiac pacemakers or implantable defibrillators for conduction defects, non-invasive ventilation for respiratory weakness, and hearing aids or cochlear implants for sensorineural loss. Patients should avoid mitochondrial toxins, such as valproate, statins, metformin, aminoglycosides, and certain anesthetics like propofol, which impair respiratory chain function; comprehensive lists guide perioperative and chronic care. Palliative care integration is recommended for advanced disease, focusing on symptom control and end-of-life planning, particularly in progressive encephalomyopathies.[41][57]Pharmacological Interventions
Certain pharmacological agents are contraindicated in mitochondrial diseases due to their potential to worsen mitochondrial dysfunction. Metformin, for instance, is avoided because it inhibits mitochondrial complex I, thereby exacerbating impaired oxidative phosphorylation and precipitating severe lactic acidosis even at therapeutic doses without classic triggers; multiple case reports document the unmasking or rapid worsening of maternally inherited diabetes and deafness (MIDD) or mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS) with profound metformin-associated lactic acidosis (MALA).[69][70][71] Pharmacological interventions for mitochondrial diseases primarily seek to bolster electron transport chain function, mitigate oxidative stress, or address specific enzymatic deficiencies, yet comprehensive evidence from large-scale randomized controlled trials remains scarce, with most therapies showing modest or subtype-specific benefits. Coenzyme Q10 (CoQ10), a key component of the mitochondrial respiratory chain, is frequently administered at doses ranging from 100 to 1200 mg daily to support ATP production and reduce reactive oxygen species; however, a phase 3 double-blind trial involving 30 patients with confirmed mitochondrial disorders (including MELAS syndrome) demonstrated only minor improvements in aerobic capacity and post-exercise lactate levels, without significant gains in primary endpoints such as gross motor function or quality of life compared to placebo.[72] Similarly, supplementation with vitamins like riboflavin (for complex I defects) or thiamine (for pyruvate dehydrogenase deficiency) is employed empirically in cases of identified cofactor responsiveness, but systematic reviews indicate benefits are largely confined to genetically confirmed responsive subtypes, with no broad efficacy across heterogeneous mitochondrial pathologies.[73] For specific mitochondrial disorders, targeted agents have yielded more promising results. Idebenone, a short-chain ubiquinone analog that bypasses complex I defects to facilitate electron transfer, received European Medicines Agency approval in 2015 for Leber's hereditary optic neuropathy (LHON); the RHODOS phase 3 trial (196 patients) reported a non-significant trend toward improved visual acuity overall but significant benefits in subgroups with discordant eye involvement or early treatment initiation, with real-world data confirming vision stabilization or recovery in up to 42% of treated eyes versus 21% in controls when administered within one year of onset.[74] In September 2025, the U.S. Food and Drug Administration granted accelerated approval to elamipretide (Forzinity), a mitochondria-targeted peptide that stabilizes cardiolipin to enhance inner membrane integrity and bioenergetics, as the first therapy for Barth syndrome—a rare X-linked disorder involving cardioskeletal myopathy and neutropenia—based on surrogate endpoints of improved left ventricular function and exercise tolerance in phase 2/3 trials, though confirmatory studies are required.[75] Emerging pharmacological options include vatiquinone (EPI-743), a redox-modulating naphthoquinone that showed delayed disease progression and reduced hospitalizations (11.8% versus 42.8% on placebo) in a phase 2b trial for Leigh syndrome, though subsequent studies were terminated due to insufficient overall efficacy. L-arginine, used acutely for stroke-like episodes in MELAS syndrome, has demonstrated reduced episode severity and frequency in multiple open-label studies by improving cerebral vasodilation and nitric oxide bioavailability.[73] Despite these advances, causal mechanisms linking interventions to clinical outcomes often rely on preclinical models, and meta-analyses underscore the need for precision approaches tailored to mutation-specific defects, as "one-size-fits-all" antioxidants frequently fail to alter natural history in non-responsive cases.[5]Emerging and Experimental Approaches
Experimental approaches to mitochondrial disease treatment target the underlying genetic and biochemical defects, including mitochondrial DNA (mtDNA) heteroplasmy, nuclear DNA mutations affecting mitochondrial function, and impaired energy production. These strategies encompass gene editing, mitochondrial replacement, allotopic expression, and novel small molecules, often evaluated in preclinical models or early-phase clinical trials. As of 2025, no curative therapies exist for most primary mitochondrial disorders, but advances in delivery systems like AAV vectors and base editors show promise in reducing mutant mtDNA load or restoring protein function.[76][77] Gene therapy efforts focus on allotopic expression, relocating mtDNA genes to the nucleus for nuclear-encoded production of corrected proteins imported into mitochondria, and direct mtDNA editing via CRISPR-like tools or restriction enzymes. In preclinical rodent models, base editors have corrected pathogenic mtDNA mutations with efficiencies up to 50%, modeling disease reversal without off-target nuclear effects.[77] A 2024 review highlights phase I/II trials for nuclear-encoded defects, such as those in Leigh syndrome, using AAV9 vectors to deliver functional genes, achieving transient metabolic improvements in animal studies but facing challenges like immune responses and incomplete penetrance in heteroplasmic tissues.[78] Japanese researchers reported in May 2025 a novel enzyme technology altering mutant mtDNA levels in patient-derived stem cells, potentially scalable to in vivo editing.[79] Mitochondrial replacement therapy (MRT), including pronuclear transfer and spindle transfer, aims to prevent maternal transmission by replacing defective maternal mitochondria with donor cytoplasm during IVF. A July 2025 UK study demonstrated MRT compatibility with embryo viability, resulting in eight live births from licensed procedures in Newcastle, with no evidence of carryover mtDNA mutations above 2%.[80][81] Ongoing trials in the UK and Australia report high fertilization and euploid blastocyst rates, though long-term safety data remain limited to post-2016 cases.[82] Ethical and regulatory hurdles persist outside select jurisdictions, with preclinical data confirming reduced heteroplasmy but variable drift in offspring tissues.[83] Pharmacological interventions in trials include elamipretide (also known as SS-31), a mitochondria-targeted antioxidant tetrapeptide that interacts with cardiolipin to stabilize the inner mitochondrial membrane, reduce reactive oxygen species, improve mitochondrial morphology and function, and ameliorate symptoms in mitochondrial diseases. It received FDA accelerated approval in September 2025 for Barth syndrome based on phase III trials demonstrating improved left ventricular function and exercise capacity in 12 patients.[75] Elamipretide has also shown promise in preclinical and clinical studies for primary mitochondrial myopathy (PMM), with the MMPOWER-3 phase III trial (NCT03323749) testing its efficacy, though it did not meet primary endpoints for 6-minute walk test distance or fatigue at 24 weeks compared to placebo; however, it has been tested in hundreds of mitochondrial disease patients and is under further investigation.[84][85] Vatiquinone (EPI-743) and KH176, redox-modulating agents, are in phase II/III for primary mitochondrial myopathies, with 2023-2025 data indicating modest reductions in fatigue and biomarkers like lactate in open-label extensions, though placebo-controlled efficacy varies.[82] Mitochondrial transplantation, involving exogenous healthy mitochondria delivery, has preclinical success in restoring ATP in ischemic models but lacks human trials due to delivery and immune rejection issues.[56] Active clinical trials as of 2025 include SPIMD-301 evaluating subcutaneous vatiquinone analogs for safety over 48 weeks in adults with mitochondrial disease, and CoQ10 extensions for pediatric energy defects, emphasizing endpoints like 6-minute walk tests.[86][72] These approaches underscore the shift toward personalized, mutation-specific interventions, yet heteroplasmy thresholds, tissue specificity, and scalability challenges limit broad applicability.[82]Epidemiology
Prevalence and Incidence
Mitochondrial diseases encompass a heterogeneous group of disorders caused by dysfunction in mitochondrial oxidative phosphorylation, with prevalence estimates ranging from 5 to 20 per 100,000 individuals based on clinical diagnoses, though these figures likely underestimate the true burden due to diagnostic challenges and underrecognition.[53] In adults, the minimum prevalence exceeds 1 in 5,000, positioning mitochondrial disease among the more common inherited metabolic disorders.[3] Specific subtypes show narrower ranges; for instance, mitochondrial DNA (mtDNA)-related diseases in adults have a minimum prevalence of 9.2 per 100,000 (95% CI: 6.5–12.7), derived from population-based studies in regions like the UK.[87] Nuclear DNA mutations contributing to overt adult mitochondrial disease occur at approximately 2.9 per 100,000.[88] In the United States, overall prevalence is estimated at 1 in 4,000, while global figures approximate 1 in 5,000, reflecting data from registries and cohort studies.[89] Pediatric prevalence may differ, with some studies indicating higher rates in children due to early-onset forms, though comprehensive population-level data remain limited. In Japan, a national survey from April 2018 to March 2019 identified 3,629 patients, yielding a prevalence of 2.9 per 100,000 (95% CI: 2.8–3.0), encompassing both primary and secondary mitochondrial dysfunctions.[90] Variations arise from methodological differences, such as reliance on clinical manifestations versus genetic confirmation, and geographic factors; for example, mtDNA point mutations alone have a population prevalence of about 1 in 200 in certain cohorts.[8] Underestimation persists because many cases present with nonspecific symptoms like fatigue or myopathy, evading specialized diagnosis until advanced stages.[91] Incidence data are sparser and primarily derived from birth cohorts or regional surveillance. Historical studies of preschool children born between 1884 and 1992 reported an incidence of mitochondrial encephalomyopathies at 8.9 per 100,000 (95% CI: 5.3–14.0), or roughly 1 in 11,000.[92] More recent figures from Hong Kong (2016–2020) indicate an incidence of 1.40 cases per million people per year, with similar trends observed in Spain, suggesting low but steady annual occurrence tied to de novo mutations or inherited defects.[93] These rates underscore the predominantly genetic etiology, with maternal inheritance for mtDNA disorders and autosomal patterns for nuclear variants contributing to sporadic and familial cases, though precise incidence remains elusive without universal newborn screening.[94]Demographic and Geographic Patterns
Mitochondrial diseases manifest across all age groups, with onset ranging from neonatal period to adulthood, though pediatric presentations predominate in many cohorts; for instance, in a retrospective study of 183 Chinese patients, 124 were children under 18 years while 59 were adults, with a mean age of 16 years.[95] Adult-onset forms, often involving encephalomyopathies like MELAS syndrome, account for a substantial minority of cases, reflecting the progressive accumulation of heteroplasmy or nuclear gene defects over time.[95] Sex distribution is generally balanced, with no consistent predominance; the same Chinese cohort showed 49.72% males among diagnosed patients, consistent with broader genetic inheritance patterns where mtDNA mutations transmit maternally but affect both sexes equally, while nuclear mutations follow Mendelian inheritance without sex linkage.[95] Certain subtypes, such as Leber hereditary optic neuropathy, exhibit male bias due to incomplete penetrance influenced by X-linked modifiers or hormones, but overall demographic data do not indicate systemic sex disparities.[92] Ethnic and population variations arise primarily from haplogroup-specific mtDNA polymorphisms and founder effects, rather than uniform racial predispositions; for example, the MT-RNR1 m.1555A>G mutation, linked to aminoglycoside-induced ototoxicity and some mitochondrial syndromes, shows higher frequencies in Asian populations (mean 4.13% in China, 5.36% in Japan) compared to Europeans or Africans.[96] Consanguinity in regions like the Arab Middle East elevates recessive nuclear-encoded mitochondrial disorders through increased homozygosity, while isolated populations exhibit bottlenecks amplifying rare mtDNA haplotypes.[92][97] Geographic prevalence estimates differ, potentially due to diagnostic access and genetic drift rather than environmental factors; reported adult rates include 23 per 100,000 in northeast England versus 5.7 per 100,000 in Spain, with global figures converging around 1 in 5,000 overall but lower documented incidence in under-resourced areas like parts of Asia.[90] Temporal increases in diagnoses, as seen in Finland from 2009–2022, likely stem from improved genetic testing rather than rising true incidence.[98] Specific syndromes show regional clustering via founder mutations, underscoring the role of historical migrations in shaping local burdens.[92]History
Early Discoveries
The first clinically and biochemically characterized mitochondrial disease was reported in 1962 by Rolf Luft and colleagues, describing a 37-year-old woman with severe non-thyroidal hypermetabolism, profuse perspiration, heat intolerance, and generalized muscle weakness. Biochemical analysis of isolated muscle mitochondria revealed defective maintenance of respiratory control, with loose coupling between oxidation and phosphorylation leading to inefficient ATP production despite high oxygen consumption.[99] Electron microscopy of the patient's skeletal muscle showed enlarged, densely packed mitochondria with abnormal cristae, marking the initial histopathological evidence of mitochondrial dysfunction as a cause of human disease. This case, now known as Luft's disease, established the paradigm of primary mitochondrial disorders characterized by energy failure in high-demand tissues like muscle.00227-6) In the mid-1960s, advances in muscle biopsy techniques further illuminated structural abnormalities in mitochondrial myopathies. In 1964, G. Milton Shy and Nicholas K. Gonatas identified giant abnormal mitochondria in a case of human myopathy, using electron microscopy to demonstrate subsarcolemmal aggregations and disrupted cristae architecture.[100] This was followed in 1966 by their description of two childhood myopathies—megaconial (enlarged mitochondria) and pleoconial (increased number of mitochondria)—highlighting proliferation of dysfunctional organelles as a compensatory response to bioenergetic deficits. Concurrently, William K. Engel and George Cunningham introduced the modified Gomori trichrome stain in 1963, which revealed "ragged red fibers" (RRFs) in affected muscle fibers, a hallmark histological feature resulting from subsarcolemmal accumulation of abnormal mitochondria staining red due to excess RNA and mitochondrial mass.[99] ![Ragged red fibers in Gomori trichrome stain][center] These early observations shifted focus from endocrine mimics to intrinsic mitochondrial pathology, with RRFs becoming a diagnostic clue for disorders like chronic progressive external ophthalmoplegia, first detailed clinically in the 1950s but linked to mitochondrial proliferation by the 1970s.[99] By the late 1970s, biochemical assays confirmed respiratory chain deficiencies in isolated mitochondria from RRF-containing biopsies, underscoring oxidative phosphorylation impairment as the causal mechanism in these myopathies.[101] Such findings laid the groundwork for recognizing mitochondrial diseases as a spectrum of multisystem disorders driven by cellular energy failure, prior to the molecular identification of genetic defects.[102]Key Milestones and Advances
The recognition of mitochondrial diseases began in the premolecular era with clinical and histopathological observations. In 1962, Rolf Luft and colleagues described the first confirmed case of a mitochondrial disorder in a 35-year-old woman exhibiting severe hypermetabolism, heat intolerance, and profuse perspiration due to loose coupling of oxidative phosphorylation, confirmed by electron microscopy revealing structurally abnormal mitochondria in skeletal muscle.[99] This marked the initial linkage of clinical symptoms to mitochondrial dysfunction. In 1963, William K. Engel and George Cunningham developed the modified Gomori trichrome staining technique for muscle biopsies, which revealed ragged-red fibers—accumulations of abnormal mitochondria—as a characteristic pathological feature in affected tissues.[99] Subsequent advances in the 1970s expanded clinical classification. Salvatore DiMauro reported cases of mitochondrial myopathy associated with ophthalmoplegia in 1973, emphasizing biochemical defects in cytochrome c oxidase.[99] By 1977, Y. Shapira and colleagues introduced the term "mitochondrial encephalomyopathies" to describe multisystem disorders involving defects in oxidative metabolism, integrating neurological and muscular manifestations.[99] In 1984, S.G. Pavlakis et al. delineated mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS) as a distinct syndrome, differentiating it from myoclonic epilepsy with ragged-red fibers (MERRF) and Kearns-Sayre syndrome (KSS) based on phenotypic criteria.[99] The molecular era commenced in 1988 with the identification of mitochondrial DNA (mtDNA) mutations, transforming diagnosis from morphological and biochemical assays to genetic analysis. Ian J. Holt et al. detected large-scale mtDNA deletions in patients with chronic progressive external ophthalmoplegia and Kearns-Sayre syndrome.[102] Concurrently, Douglas C. Wallace identified a point mutation in the MT-ND4 gene causing Leber's hereditary optic neuropathy (LHON), establishing maternally inherited mtDNA defects as a cause of neurodegeneration.[102] Massimo Zeviani's group confirmed mtDNA deletions in KSS.[102] These findings elucidated heteroplasmy and threshold effects in mtDNA disorders. In 1995, Thomas Bourgeron et al. reported the first nuclear DNA mutation (in the SURF1 gene) causing respiratory chain deficiency in Leigh syndrome, highlighting the role of nuclear-encoded proteins in mitochondrial function.[99]| Year | Milestone | Key Contributors and Details |
|---|---|---|
| 1962 | First clinical description of mitochondrial disease | Luft et al.: Hypermetabolic state with abnormal mitochondria in muscle.[99] |
| 1963 | Discovery of ragged-red fibers | Engel and Cunningham: Modified Gomori trichrome stain identifies mitochondrial proliferations.[99] |
| 1973 | Mitochondrial myopathy with ophthalmoplegia | DiMauro: Biochemical defects in electron transport chain.[99] |
| 1984 | Definition of MELAS syndrome | Pavlakis et al.: Stroke-like episodes linked to mitochondrial dysfunction.[99] |
| 1988 | First mtDNA mutations identified | Holt (deletions), Wallace (LHON point mutation), Zeviani (KSS); onset of molecular diagnostics.[102] [99] |
| 1995 | First nuclear DNA mutation in mitochondrial disease | Bourgeron et al.: SURF1 mutations in Leigh syndrome.[99] |

