Recent from talks
Nothing was collected or created yet.
Bikont
View on Wikipedia
| Bikonts Temporal range:
| |
|---|---|

| |
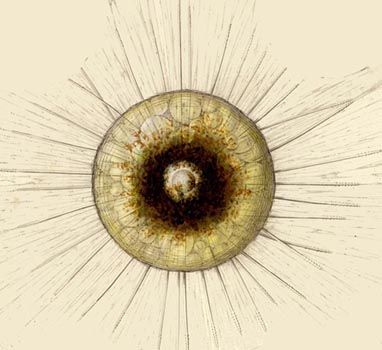
| A radiolarian | |
| Scientific classification | |
| Domain: | Eukaryota |
| (unranked): | Orthokaryotes |
| (unranked): | Neokaryotes |
| (unranked): | Bikonta Cavalier-Smith, 1993 |
| Subgroups | |
| Synonyms | |
| |
A bikont ("two flagella") is any of the eukaryotic organisms classified in the group Bikonta. Many single-celled and multi-celled organisms are members of the group, and these, as well as the presumed ancestor, have two flagella.[1]
Enzymes
[edit]Another shared trait of bikonts is the fusion of two genes into a single unit: the genes for thymidylate synthase (TS) and dihydrofolate reductase (DHFR) encode a single protein with two functions.[2]
The genes are separately translated in unikonts.
Relationships
[edit]Some research suggests that a unikont (a eukaryotic cell with a single flagellum) was the ancestor of opisthokonts (Animals, Fungi, and related forms) and Amoebozoa, and a bikont was the ancestor of Archaeplastida (Plants and relatives), Excavata, Rhizaria, and Chromalveolata. Cavalier-Smith has suggested that Apusozoa, which are typically considered incertae sedis, are in fact bikonts.[3]
Relationships within the bikonts are not yet clear. Cavalier-Smith has grouped the Excavata and Rhizaria into the Cabozoa and the Archaeplastida and Chromalveolata into the Corticata, but at least one other study has suggested that the Rhizaria and Chromalveolata form a clade.[4]
An alternative to the Unikont–Bikont division was suggested by Derelle et al. in 2015,[5] where they proposed the acronyms Opimoda–Diphoda respectively, as substitutes to the older terms. The name Diphoda is formed from the letters of DIscoba and diaPHOretickes (shown in capitals). [suggested singular forms are Opneme-Dipheme respectively][citation needed]
Cladogram
[edit]A "classical" cladogram (data from 2012, 2015) is:[6][7]
| Eukaryotes |
| |||
However, a cladogram (data from 2015, 2016) with the root in Excavata is[5][8][9]
| Eukaryotes |
| |||||||||
The corticates correspond roughly to the bikonts. While Haptophyta, Cryptophyta, Glaucophyta, Rhodophyta, the SAR supergroup and Viridiplantae are usually considered monophyletic, Archaeplastida may be paraphyletic, and the mutual relationships between these phyla are still to be fully resolved.
Recent reconstructions placed Archaeplastida and Hacrobia together in an "HA supergroup" or "AH supergroup", which was a sister clade to the SAR supergroup within the SAR/HA supergroup. However, this seems to have fallen out of favor as the monophyly of hacrobia has come under dispute.
See also
[edit]References
[edit]- ^ Burki F, Pawlowski J (October 2006). "Monophyly of Rhizaria and multigene phylogeny of unicellular bikonts". Mol. Biol. Evol. 23 (10): 1922–30. doi:10.1093/molbev/msl055. PMID 16829542.
- ^ Thomas Cavalier-Smith (2003). "Protist phylogeny and the high-level classification of Protozoa". European Journal of Protistology. 39 (4): 338–348. doi:10.1078/0932-4739-00002.
- ^ Cavalier-Smith, Thomas (1 January 2003). "Protist phylogeny and the high-level classification of Protozoa". European Journal of Protistology. 39 (4): 338–348. doi:10.1078/0932-4739-00002.
- ^ Burki F, Shalchian-Tabrizi K, Minge M, Skjæveland Å, Nikolaev SI, et al. (2007). Butler G (ed.). "Phylogenomics Reshuffles the Eukaryotic Supergroups". PLOS ONE. 2 (8: e790): e790. Bibcode:2007PLoSO...2..790B. doi:10.1371/journal.pone.0000790. PMC 1949142. PMID 17726520.
- ^ a b Derelle, Romain; Torruella, Guifré; Klimeš, Vladimír; Brinkmann, Henner; Kim, Eunsoo; Vlček, Čestmír; Lang, B. Franz; Eliáš, Marek (17 February 2015). "Bacterial proteins pinpoint a single eukaryotic root". Proceedings of the National Academy of Sciences. 112 (7): E693 – E699. Bibcode:2015PNAS..112E.693D. doi:10.1073/pnas.1420657112. PMC 4343179. PMID 25646484.
- ^ Jackson, Christopher; Clayden, Susan; Reyes-Prieto, Adrian (2015). "The Glaucophyta: the blue-green plants in a nutshell". Acta Societatis Botanicorum Poloniae. 84 (2): 149–165. Bibcode:2015AcSBP..84..149J. doi:10.5586/asbp.2015.020.
- ^ Burki, Fabien; Okamoto, Noriko; Pombert, Jean-François; Keeling, Patrick J. (7 June 2012). "The evolutionary history of haptophytes and cryptophytes: phylogenomic evidence for separate origins". Proceedings of the Royal Society of London B: Biological Sciences. 279 (1736): 2246–2254. doi:10.1098/rspb.2011.2301. PMC 3321700. PMID 22298847.
- ^ Cavalier-Smith, Thomas; Chao, Ema E.; Lewis, Rhodri (December 2015). "Multiple origins of Heliozoa from flagellate ancestors: New cryptist subphylum Corbihelia, superclass Corbistoma, and monophyly of Haptista, Cryptista, Hacrobia and Chromista". Molecular Phylogenetics and Evolution. 93: 331–362. Bibcode:2015MolPE..93..331C. doi:10.1016/j.ympev.2015.07.004. PMID 26234272.
- ^ Eliáš, Marek; Klimeš, Vladimír; Derelle, Romain; Petrželková, Romana; Tachezy, Jan (2016). "A paneukaryotic genomic analysis of the small GTPase RABL2 underscores the significance of recurrent gene loss in eukaryote evolution". Biology Direct. 11 (1): 5. doi:10.1186/s13062-016-0107-8. PMC 4736243. PMID 26832778.
Further reading
[edit] Data related to Bikonta at Wikispecies
Data related to Bikonta at Wikispecies Media related to Bikonta at Wikimedia Commons
Media related to Bikonta at Wikimedia Commons- Stechmann A, Cavalier-Smith T (September 2003). "The root of the eukaryote tree pinpointed". Curr. Biol. 13 (17): R665–6. Bibcode:2003CBio...13.R665S. doi:10.1016/S0960-9822(03)00602-X. PMID 12956967.
- Tree of Life Eukaryotes
Bikont
View on GrokipediaDefinition and Characteristics
Etymology and Core Concept
The term "bikont" derives from the Greek prefix "bi-" meaning "two" and "kontos" meaning "pole" or "flagellum," referring to the ancestral possession of two flagella in these organisms.[9] It was coined by Thomas Cavalier-Smith in his 2002 paper proposing a revised classification of eukaryotic protozoa based on morphological and ultrastructural traits. At its core, the bikont concept defines a major clade of eukaryotes whose last common ancestor possessed two heterodynamic flagella: an anterior tinsel flagellum for propulsion and a posterior smooth flagellum for steering, a configuration distinct from the single flagellum typical of unikonts. Although influential, the bikont clade has been challenged by subsequent phylogenomic studies.[6] This biflagellate kinetid is considered a key synapomorphy, reflecting an early evolutionary innovation in eukaryotic motility and cell organization that facilitated diverse modes of locomotion and feeding. Bikonts thus represent one of the two primary branches in the eukaryotic tree, encompassing a broad array of lineages that diverged after the acquisition of this flagellar apparatus. Bikonts include diverse eukaryotic groups such as various protists, plants, and certain algae, underscoring their role as a foundational supergroup in eukaryotic evolution. Supporting this morphological hypothesis, molecular evidence like the fused thymidylate synthase-dihydrofolate reductase (TS-DHFR) enzyme provides a genetic synapomorphy unique to bikonts, though detailed phylogenetic implications are explored elsewhere.[10]Key Morphological Traits
Bikonts are characterized by an ancestral condition of possessing two flagella, or cilia, arising from a pair of kinetosomes arranged in a characteristic two-pole configuration, which supports their motility and cellular organization.[11] In many lineages, such as stramenopiles within the SAR clade, the anterior flagellum is often equipped with hair-like structures known as mastigonemes or vanes that enhance propulsion through undulating motion, while the posterior flagellum is typically smooth; excavates may have variations, such as a dorsal vane on the posterior flagellum in some groups.[11] In rhizarians, the anterior flagellum may be smoother. This biflagellate apparatus contrasts with the single flagellum typical of unikonts, underscoring a fundamental divergence in eukaryotic locomotor evolution.[12] Cytoskeletal elements in bikonts further reflect this dual organization, featuring paired microtubular rootlets emanating from the kinetosomes that stabilize the cell and facilitate directed movement.[11] A prominent synapomorphy among many bikont groups is the presence of a ventral feeding groove or cytostome, often supported by these microtubular arrays, which enables phagotrophic nutrition by channeling prey toward an oral apparatus; this is evident in excavates like jakobids and bicosoecids, as well as alveolates such as ciliates.[12] In rhizarians, such as cercozoans, the cytoskeleton includes radiating microtubules or filose pseudopodia that extend the two-pole theme to foraging structures.[11] Trait variation occurs across bikont diversity, with the biflagellate condition retained in free-living protists like euglenozoans and cercozoans for active swimming, but frequently lost or modified in derived multicellular forms.[12] For instance, in the Plantae clade (archaeplastids), flagella are present in unicellular green algae such as Chlamydomonas but absent in vascular land plants, where sessile lifestyles have rendered them obsolete.[11] Similarly, in some rhizarians and alveolates, flagella may be reduced to gametes or replaced by amoeboid locomotion, highlighting adaptive shifts from the ancestral motile state.[11]Distinguishing Cellular Features
Bikonts exhibit distinct non-flagellar cellular features that contribute to their polarized organization and metabolic adaptations, setting them apart from unikonts. A key trait is the orientation of the Golgi apparatus, which is typically positioned anterior to the nucleus in many bikont lineages, facilitating directed secretion and vesicle trafficking aligned with cellular polarity. For instance, in bicosoecids such as Cafeteria roenbergensis, the single Golgi body is located anterior to the nucleus and near the flagellar bases.[13] In contrast, unikonts like choanoflagellates show the Golgi apparatus positioned posterior to the nucleus, reflecting differences in cytoskeletal and secretory dynamics.[14] Mitochondrial cristae in bikonts are predominantly tubular or discoidal, providing increased surface area for respiratory processes compared to the flat cristae characteristic of unikonts. This morphology is evident across diverse bikont groups, such as stramenopiles and rhizaria, where tubular cristae predominate, although secondary modifications like flat cristae appear in excavates. These structural variations support efficient energy production tailored to the ecological niches of bikonts, including free-living and parasitic lifestyles.[15] Plastid traits further highlight bikont diversity, with photosynthetic members featuring chlorophyll-based pigments acquired through endosymbiotic events. Archaeplastida, a core bikont lineage, possess primary plastids derived from a single cyanobacterial endosymbiosis, containing chlorophyll a and phycobilins in red algae or chlorophyll a and b in green algae. Chromalveolates, another bikont supergroup, generalize secondary endosymbiosis with a red alga, yielding four-membraned plastids rich in chlorophyll a and c, as seen in stramenopiles and haptophytes. These endosymbiotic innovations underscore the role of organelle acquisition in bikont evolutionary success.[16]Historical Development
Initial Proposal
The bikont hypothesis was initially proposed by Thomas Cavalier-Smith in his 1993 classification of eukaryotic kingdoms, where he introduced the branch Bikonta within the kingdom Protozoa to encompass a major clade of eukaryotes characterized by the possession of two cilia or flagella.[17] This grouping included 16 phyla of primarily unicellular or plasmodial organisms with mitochondria, distinguishing them from other protozoan lineages.[17] Cavalier-Smith's framework positioned Bikonta as a fundamental division within Protozoa.[17] The rationale for this proposal stemmed from ultrastructural analyses using electron microscopy, which revealed shared morphological features in the flagellar apparatus across diverse protist groups.[17] Specifically, bikonts were unified by the presence of a bikinetid, a paired ciliary or flagellar structure with distinct insertion points on the cell surface and associated rootlet systems that provide structural support and orientation.[17] These traits, including tubular or flat mitochondrial cristae (as opposed to discoid in plants), were interpreted as synapomorphies indicating a common ancestry.[17] This initial proposal emerged during ongoing debates in the early 1990s about the root of the eukaryotic tree of life, where molecular and morphological data were beginning to challenge traditional groupings.[17] By emphasizing ultrastructural evidence over emerging genetic sequences, the hypothesis aimed to resolve uncertainties in protist phylogeny and establish a stable higher-level taxonomy for eukaryotes.[17]Subsequent Revisions and Synonyms
Following the initial proposal of the bikont concept, Thomas Cavalier-Smith elevated Bikonta to infrakingdom status in 2002, supported by phylogenetic analyses of a derived dihydrofolate reductase-thymidylate synthase gene fusion that rooted the eukaryotic tree between Unikonts and Bikonts.[18] In 2004, Cavalier-Smith further refined the taxonomic scope by incorporating chromalveolate lineages—such as stramenopiles and alveolates—into a new supergroup called Chromalveolata within Bikonta, justified by evidence of a shared secondary endosymbiosis of a red alga and unified membrane topologies in their plastids.[19] Key revisions in the late 2000s reflected expanding genomic datasets; in 2009, Rhizaria was integrated into the Bikont lineage through its transfer from kingdom Protozoa to an expanded kingdom Chromista, as multigene phylogenies confirmed its close relationship with cercozoans and alveolates, forming the core of the SAR clade.[20] This integration contributed to the 2013 proposal of the Corticata supergroup by Cavalier-Smith, which united Chromista, Archaeplastida, and related Bikont-derived lineages based on shared cortical alveoli or microtubule bands and cytochrome c biogenesis pathways, highlighting the role of growing sequence data in delineating higher-level eukaryotic relationships.[21] In the 2010s, synonyms emerged to describe the broader Bikont assemblage; Adl et al. introduced Diaphoretickes in 2012 as a major clade encompassing SAR, Archaeplastida, Cryptista, and Haptophyta, defined node-based on phylogenomic trees and distinguished from Amorphea by unique combinations of ribosomal protein genes and rRNA expansion segments.[22] By 2015, the Opimoda–Diphoda rooting model repositioned the eukaryotic root between Opimoda (Amorphea) and Diphoda (encompassing Diaphoretickes and Excavata), with Diphoda serving as a synonym for the derived Bikont radiation, validated by phylogenies of bacterial-derived eukaryotic proteins that resolved long-standing ambiguities in tree rooting.[6]Taxonomic Scope
Major Included Lineages
The classical bikont hypothesis encompasses several major eukaryotic groups, unified by ancestral possession of two flagella and the derived fusion of dihydrofolate reductase and thymidylate synthase genes.[1] Archaeplastida, also known as Plantae, represents the primary lineage of eukaryotes with plastids derived directly from an ancient cyanobacterial endosymbiont; it includes three main divisions: the green plants and algae (Viridiplantae, encompassing Chlorophyta and land plants), red algae (Rhodophyta), and glaucophytes (Glaucophyta), which retain primitive plastid features like peptidoglycan walls.[23] Excavata comprises diverse anaerobic and aerobic protists characterized by a ventral feeding groove or cytostome, reflecting their name; key subgroups include diplomonads (e.g., Giardia), parabasalids (e.g., trichomonads like Trichomonas), and euglenozoans (e.g., euglenids and kinetoplastids such as Trypanosoma).[1] Rhizaria is a supergroup of mostly marine, often filose or reticulose amoeboid protists, many with intricate mineralized tests; prominent lineages are foraminiferans (shelled amoebae forming calcium carbonate tests), radiolarians (with silica skeletons and axopodia for prey capture), cercozoans, and Apusozoa (filopodial amoeboflagellates).[24][1] Chromalveolata unites chromists and alveolates, forming a diverse assemblage of photosynthetic, parasitic, and predatory forms; chromists (Chromista) include stramenopiles (heterokonts, such as diatoms with silica frustules and oomycetes like Phytophthora), haptophytes (e.g., coccolithophores with calcium carbonate scales), and cryptophytes (flagellated algae with nucleomorphs from red algal secondary endosymbiosis); alveolates feature cortical alveoli and encompass ciliates for motility and apicomplexans like Plasmodium as parasites.[1]Boundaries and Exclusions
The primary exclusion from the Bikont clade encompasses the Unikonts, which comprise Opisthokonta (including animals, fungi, and choanoflagellates) and Amoebozoa. These groups are distinguished by the presence of a single posterior flagellum in motile forms and separate, unfused genes for thymidylate synthase (TS) and dihydrofolate reductase (DHFR), contrasting with the ancestral biflagellate condition and fused TS-DHFR gene characteristic of Bikonts.[1]Molecular Evidence
Fused Enzymes as a Synapomorphy
The bifunctional thymidylate synthase-dihydrofolate reductase (TS-DHFR) enzyme represents a key genetic synapomorphy proposed for bikonts, where the genes encoding thymidylate synthase (TS) and dihydrofolate reductase (DHFR) are fused into a single bifunctional protein. TS catalyzes the methylation of deoxyuridine monophosphate to deoxythymidine monophosphate, a critical step in DNA synthesis, while DHFR reduces dihydrofolate to tetrahydrofolate, regenerating the cofactor necessary for this reaction and supporting overall folate metabolism.[25] This fusion enables coordinated expression and potential substrate channeling between the two enzymatic domains, enhancing efficiency in nucleotide biosynthesis across diverse environmental conditions.[26] In evolutionary terms, the TS-DHFR fusion is absent in unikonts, where TS and DHFR exist as separate genes, but is present in many major bikont lineages such as Plantae, Alveolata, Stramenopiles, and Rhizaria, though variable in Excavata (e.g., absent in Giardia).[27] This derived gene fusion provides evidence for the monophyly of a subset of bikonts, supporting a deep divergence between bikonts and unikonts early in eukaryote evolution, though its reliability is questioned due to losses in some subgroups.[25] The fusion's presence in diverse groups underscores a shared biochemical innovation that likely contributed to their adaptive success in varied niches. However, subsequent studies have noted its inconsistent distribution, complicating its use as a strict synapomorphy.[25] The TS-DHFR fusion was first identified through comparative genome analyses, revealing its presence in diverse bikont groups such as plants and alveolates. For instance, genome sequencing of Arabidopsis thaliana (a plant) confirmed the bifunctional TS-DHFR gene, integral to its folate-dependent pathways.[26] Similarly, in Plasmodium falciparum (an alveolate), the fusion is evident in the nuclear genome, where the bifunctional enzyme is essential for the parasite's replication.[28] Critically, the fusion's distribution in non-photosynthetic bikonts like Plasmodium demonstrates that it originated prior to the acquisition of plastids in the Archaeplastida lineage, reinforcing its status as an ancient synapomorphy predating major endosymbiotic events.[29]Additional Genetic Markers
Bikonts are supported by several additional genetic markers beyond fused enzymes, including distinctive patterns in ribosomal proteins, gene content, and multi-gene phylogenetic analyses. Distinctive ribosomal protein compositions have been noted in the Diaphoretickes clade, a major subgroup within the proposed bikonts, contributing to differences in ribosomal structure and function compared to unikonts. Shared gene content among non-archaeplastid bikonts includes cyanobacterial-derived genes acquired through secondary endosymbiosis, such as those involved in photosynthesis and carbon fixation pathways. These genes, transferred from engulfed red or green algal endosymbionts, are present in groups like chromalveolates and haptophytes, providing a genomic signature of ancient endosymbiotic events that unified diverse bikont lineages. Pre-2015 phylogenomic studies using concatenated datasets of 100 or more genes revealed strong phylogenetic signal for bikont clustering, placing major lineages like Rhizaria, Alveolata, and Stramenopiles together as a monophyletic group separate from unikonts. For instance, an analysis of 85 conserved proteins confirmed the inclusion of Rhizaria within unicellular bikonts, highlighting the robustness of multi-gene approaches in resolving deep eukaryotic relationships despite challenges from long-branch attraction.[30] Later analyses, however, have challenged the overall monophyly of bikonts using larger datasets.[6]Phylogenetic Position
Relation to Unikonts
The bikont-unikont dichotomy represents the primary bifurcation in eukaryotic evolution, as proposed by Cavalier-Smith, dividing eukaryotes into two major clades that diverged from the last eukaryotic common ancestor (LECA).[31] In classical phylogenetic models, the root of the eukaryotic tree lies precisely between these clades, supported by analyses of rare genomic events such as the fusion of dihydrofolate reductase and thymidylate synthase genes, which is absent in unikonts but present in bikonts.[32] This split posits bikonts as encompassing diverse lineages with ancestral biciliate (two-flagellum) organization, including Plantae, chromalveolates, and rhizarians, while unikonts include amoebozoans and opisthokonts (such as animals and fungi) with ancestral uniciliate (single-flagellum) structure.[31] Bikonts and unikonts share a common ancestry in LECA, which possessed a hydrogenosome-like mitochondrion derived from an alphaproteobacterial endosymbiont, enabling proto-aerobic metabolism, and a sophisticated cytoskeleton featuring actin, tubulins, and associated motors for cellular motility and division.[33][34] Fossil-calibrated molecular phylogenies estimate this divergence occurred approximately 1.8 billion years ago, marking the onset of major eukaryotic diversification during the Paleoproterozoic era.[24] However, this rooting has become controversial in recent phylogenomic studies (as of 2025), with alternative positions proposed.[35] Key contrasts between the clades arise from their ancestral flagellar apparatuses, with bikonts exhibiting two flagella arising from orthogonally arranged basal bodies and unikonts a single flagellum, influencing early evolutionary trajectories in locomotion and feeding mechanisms.[31] This distinction implies that amoeboid locomotion, prevalent in unikont lineages like Amoebozoa, evolved independently from the excavate-style ventral groove feeding and swimming seen in basal bikont groups such as Excavata, highlighting divergent adaptations from the shared ciliary heritage of LECA.[31]Internal Relationships and Supergroups
The internal relationships among bikonts, a proposed clade of eukaryotes characterized by ancestral possession of two flagella, have been hypothesized to involve several major supergroups based on shared ultrastructural, genetic, and biochemical synapomorphies. These include Cabozoa and Corticata, with additional debated groupings such as the HA supergroup. These proposals stem primarily from phylogenomic analyses and comparative morphology, though ongoing debates highlight the challenges in resolving deep eukaryotic divergences due to long-branch attraction and incomplete taxon sampling.[36] Cabozoa, proposed by Cavalier-Smith in 1999, unites Excavata and Rhizaria as a clade potentially retaining ancestral traits from early bikont evolution, such as reduced mitochondria (mitosomes in Excavata and hydrogenosomes or mitosomes in some Rhizaria) and a shared cytoskeletal organization involving ventral feeding grooves and extrusomes. This grouping suggests a common origin for these lineages as free-living, biciliate predators that diversified through organelle reduction and pseudopodial adaptations in Rhizaria, with molecular support from multigene phylogenies showing moderate bootstrap values for their sister relationship. However, the monophyly of Cabozoa has been contested in broader phylogenomic studies, which sometimes place Rhizaria closer to other chromalveolate-like groups rather than Excavata.[37][38][39] Corticata, elevated to superkingdom status by Cavalier-Smith and colleagues in 2015, encompasses Archaeplastida (including land plants and glaucophytes) and Chromista (comprising SAR—Stramenopiles, Alveolates, Rhizaria—and Hacrobia), linked by the synapomorphy of cortical alveoli (small membrane sacs beneath the plasma membrane) and associated subpellicular microtubules that support a structured cell cortex for feeding and motility. This clade is robustly supported in multiprotein tree analyses, reflecting an ancestral neokaryote excavate-like ancestor that acquired Golgi-derived alveoli and secondary red algal plastids, with shared periplastid reticulum structures facilitating protein targeting in chromist members. Plastid-related features, such as chlorophyll c and shared derlin paralogues for envelope biogenesis, further reinforce these connections, distinguishing Corticata from non-plastid-bearing bikont lineages.[36][40] The HA supergroup, an earlier proposal linking Haptophytes (within Hacrobia) and Alveolates based on shared red algal-derived plastids and ciliary transition zone features like short transition zones and axosomes, has faced significant scrutiny since 2010. Phylogenomic evidence from nuclear genes has repeatedly failed to recover their monophyly, instead supporting separate acquisitions of secondary plastids in these lineages and placing Haptophytes closer to Cryptophytes or Stramenopiles in revised trees. This challenges the original chromalveolate framework that included HA as a core component, highlighting horizontal gene transfers and differential plastid retention as complicating factors in bikont diversification.[36][41]Alternative Hypotheses and Cladograms
Classical Bikont Cladogram
The classical bikont cladogram posits the root of the eukaryotic tree between the sister groups Unikonts and Bikonts, with Bikonts subsequently branching into three major lineages: Cabozoa, Rhizaria, and Corticata.[42] This structure reflects the traditional view of bikont monophyly during the 2010–2015 period, emphasizing shared synapomorphies such as the presence of two cilia in ancestral forms and fused gene pairs like dihydrofolate reductase-thymidylate synthase.[42] Supporting evidence derives from multigene phylogenetic analyses that aimed to recover the Unikont-Bikont bipartition. Major nodes, such as the bikont crown and its primary branches, exhibit varying support in maximum likelihood reconstructions. These analyses underscore the evolutionary coherence of Bikonts as a clade encompassing diverse protozoan and multicellular lineages. A text-based outline of the cladogram illustrates the hierarchical relationships as follows:Eukaryota
├── Unikonts (e.g., [Opisthokonts](/page/Opisthokont), [Amoebozoa](/page/Amoebozoa))
└── Bikonts
├── Cabozoa (e.g., [Excavata](/page/Excavata), including diplomonads and parabasalids)
├── [Rhizaria](/page/Rhizaria) (e.g., cercozoans, foraminiferans, radiolarians)
└── Corticata
├── [Archaeplastida](/page/Archaeplastida) (e.g., red, green, and glaucophyte algae)
└── Chromalveolata (e.g., Stramenopiles, [Alveolates](/page/Alveolate); note: later variants group [Rhizaria](/page/Rhizaria) with Stramenopiles and [Alveolates](/page/Alveolate) as SAR, positioning it within or sister to Corticata)
Eukaryota
├── Unikonts (e.g., [Opisthokonts](/page/Opisthokont), [Amoebozoa](/page/Amoebozoa))
└── Bikonts
├── Cabozoa (e.g., [Excavata](/page/Excavata), including diplomonads and parabasalids)
├── [Rhizaria](/page/Rhizaria) (e.g., cercozoans, foraminiferans, radiolarians)
└── Corticata
├── [Archaeplastida](/page/Archaeplastida) (e.g., red, green, and glaucophyte algae)
└── Chromalveolata (e.g., Stramenopiles, [Alveolates](/page/Alveolate); note: later variants group [Rhizaria](/page/Rhizaria) with Stramenopiles and [Alveolates](/page/Alveolate) as SAR, positioning it within or sister to Corticata)
Contemporary Phylogenetic Models
Contemporary phylogenetic models have increasingly challenged the classical bikont hypothesis by incorporating advanced phylogenomic techniques and broader taxon sampling, often reconfiguring the eukaryotic root and rendering traditional bikont groupings paraphyletic or polyphyletic. These models emphasize the integration of bacterial gene transfers and sophisticated reconciliation methods to resolve deep divergences, diverging from the fixed structure of earlier cladograms that posited a monophyletic Bikonta clade.[6] The Opimoda–Diphoda model, proposed in 2012, represents a seminal reconfiguration of eukaryotic phylogeny based on the analysis of bacterial proteins transferred to the eukaryotic ancestor. This model places the root of the eukaryotic tree between two primary clades: Opimoda, which encompasses Amorphea (including Opisthokonta) and a reconfigured Diaphoretickes, and Diphoda, which aligns closely with the traditional Bikonta supergroup. Under this framework, Bikonts are identified as the Diphoda clade, characterized by shared innovations such as diphthamide synthesis pathways, but the rooting implies that certain lineages previously considered basal to Bikonta are now positioned within Opimoda, thus altering interpretations of early eukaryotic evolution. The model's robustness was supported by concatenated and coalescent-based analyses of up to 108 universal eukaryotic genes across 23 diverse taxa, highlighting a single root not confounded by phylogenetic artifacts.[6][6] More recent excavations of the eukaryotic root, as explored in 2024 preprints, suggest an excavate-anchored phylogeny that further undermines bikont monophyly. Utilizing state-of-the-art phylogenetic models on extensive datasets, these studies position Excavata (including groups like jakobids and diplonemids) as the earliest-branching eukaryotic lineage, with the root lying between Excavata and a clade comprising Amorphea, Diaphoretickes, and other supergroups. This excavate-rooted tree renders traditional Bikonta paraphyletic, as excavates—often central to bikont proposals—are separated from other purported bikont lineages like Rhizaria and Alveolata, which cluster within later branches. The analyses employed comprehensive phylogenomic pipelines on hundreds of genes from over 200 taxa, incorporating mixture models to account for site-heterogeneous evolution and long-branch attraction, providing a framework that prioritizes excavate ancestry for key eukaryotic innovations.[7][7] Gene tree parsimony (GTP) approaches have also contributed to contemporary debates, particularly through a 2022 study analyzing 2,786 gene families across 158 eukaryotic lineages. This method reconciles individual gene trees with species trees to minimize duplication and loss events, placing the eukaryotic root between Opisthokonta and all other lineages, thereby supporting the unity of Bikonta (all non-Opisthokonta eukaryotes) as monophyletic while challenging the traditional Unikonta by aligning Amoebozoa with Bikonta. The GTP framework outperformed traditional concatenation methods in handling incomplete lineage sorting and horizontal gene transfer, with results validated across multiple reconciliation algorithms and taxon subsets.[43][43] As of 2025, further advances in phylogenomics, such as the EukPhylo v1.0 toolkit, have refined these estimates, recovering established clades and reinforcing support for excavate-rooted or Opisthokonta-basal models that continue to question bikont monophyly.[44]Criticisms and Current Consensus
Challenges to the Hypothesis
One major challenge to the monophyly of Bikonts arises from evidence suggesting paraphyly, particularly involving Excavata, where long-branch attraction (LBA) artifacts in phylogenetic reconstructions often pull these taxa toward the base of the eukaryotic tree. Analyses of rare genomic changes (RGCs), such as specific amino acid insertions or fusions, indicate that Excavata may affiliate more closely with Unikonts than with other proposed Bikont groups like Plantae, contradicting the expected monophyletic clustering. This 2008 study, which examined RGCs across eukaryotic supergroups, found no support for Bikont monophyly and instead highlighted mixed affinities, attributing the basal positioning of Excavata to LBA rather than true evolutionary relationships.[45] Further inconsistencies in proposed synapomorphies undermine the Bikont hypothesis, notably the dihydrofolate reductase-thymidylate synthase (DHFR-TS) gene fusion, originally posited as a derived marker uniting Bikonts. While present in many Bikont lineages such as Excavata and Rhizaria, this fusion is absent or variably distributed in some groups sometimes included in expanded Bikont proposals, like certain Apusozoa, where genomic data show separate DHFR and TS genes rather than the expected fusion. A 2006 phylogenomic analysis of Apusomonads revealed a lack of robust independent support for the DHFR-TS fusion defining a cohesive Bikont clade, suggesting it may have arisen independently or been lost in peripheral members. Additionally, morphological synapomorphies, such as the anteriorly directed pair of flagella, are complicated by secondary losses in numerous lineages; for instance, centrohelid heliozoans, classified as Bikonts, evolved from flagellate ancestors through ciliary loss, resulting in amoeboid forms that obscure ancestral traits.[46][47] Rate heterogeneity across Bikont subgroups also poses significant obstacles to phylogenetic resolution, as accelerated evolutionary rates in certain lineages distort tree topologies. Rhizarians and alveolates, key components of Bikonts, exhibit notably fast substitution rates in multigene datasets, leading to LBA artifacts that artificially cluster them with distant groups or pull them away from closer relatives. A 2006 multigene study of unicellular Bikonts demonstrated that this rate acceleration in rhizarian and alveolate phyla likely contributes to inconsistent signals in eukaryotic phylogenies, complicating efforts to confirm monophyly and suggesting rapid early diversification masked true relationships.[30]Recent Advances in Eukaryotic Phylogeny
Recent phylogenomic studies from 2022 to 2025 have significantly advanced the understanding of eukaryotic deep phylogeny, particularly through large-scale analyses that support a root between Opisthokonta (including animals, fungi, and their relatives) and all other eukaryotes. A key 2022 study utilizing gene tree parsimony on 2,786 gene families across 158 diverse eukaryotic lineages provided robust evidence for this bipartition, resolving long-standing ambiguities in the tree's base and emphasizing the need for reconciliation methods to account for gene duplication and loss events.[43] These analyses highlight the diversity within Excavata, a major bikont subgroup, with a 2024 preprint demonstrating excavate-like features in the last eukaryotic common ancestor through comprehensive rooting strategies involving thousands of orthologs, suggesting early divergence and ancestral traits like ventral feeding grooves.[7] This rooting has implications for the bikont concept, shifting interpretations toward greater resolution of ancient polytomies while portraying Bikonta more as an evolutionary grade than a strict clade in some models. For instance, the integration of excavate ancestry challenges monophyly by positioning Excavata near the base, with subsequent radiations leading to paraphyletic arrangements among archaeplastids and other protist groups.[7] Additionally, molecular clock estimates place the primary cyanobacterial endosymbiosis, foundational to photosynthetic bikont lineages like Archaeplastida, around 1.5 billion years ago, providing temporal context for the diversification of oxygen-producing eukaryotes.[48] Ongoing debates center on incorporating uncultured protists through environmental metagenomics and single-cell sequencing, which have revealed hidden diversity and refined supergroup boundaries. A 2025 global metagenomic survey uncovered uneven protist distributions across ecosystems, enhancing sampling of rare lineages and aiding placement of protists with uncertain phylogenetic affiliations (PUPAs) via hundreds of marker genes.[49] This approach has specifically refined the SAR (Stramenopiles, Alveolates, Rhizaria) supergroup by clarifying positions like Telonemia's debated affinity and strengthened Diaphoretickes through bridges like the CAM clade linking Cryptista, Alveolates, and Microheliella to Archaeplastida.[50] Such integrations continue to test alternative models, like those emphasizing Amorphea expansions, but underscore a consensus on Bikonta's utility as a descriptive framework despite internal complexities.[50]References
- https://en.wiktionary.org/wiki/unikont