Recent from talks
Nothing was collected or created yet.
Mediastinum
View on WikipediaThis article needs additional citations for verification. (September 2014) |
| Mediastinum | |
|---|---|
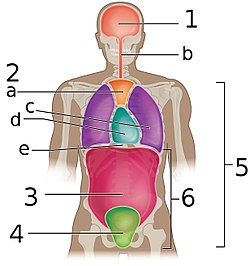
 Frontal view of the body cavities: superior mediastinum labeled a, and the pericardial cavity, which is part of the inferior mediastinum, labeled d | |
 Mediastinum, lateral view. The division between superior and inferior is at the sternal angle. | |
| Details | |
| Identifiers | |
| Latin | mediastinum[1] |
| MeSH | D008482 |
| TA98 | A07.1.02.101 |
| TA2 | 3333 |
| FMA | 9826 |
| Anatomical terminology | |
The mediastinum (from Medieval Latin: mediastinus, lit. 'midway';[2]pl.: mediastina) is the central compartment of the thoracic cavity. Surrounded by loose connective tissue, it is a region that contains vital organs and structures within the thorax, mainly the heart and its vessels, the esophagus, the trachea, the vagus, phrenic and cardiac nerves, the thoracic duct, the thymus and the lymph nodes of the central chest.
Anatomy
[edit]The mediastinum lies within the thorax and is enclosed on the right and left by pleurae. It is surrounded by the chest wall in front, the lungs to the sides and the spine at the back. It extends from the sternum in front to the vertebral column behind.[3][4] It contains all the organs of the thorax except the lungs.[4][5] It is continuous with the loose connective tissue of the neck.
The mediastinum can be divided into an upper (or superior) and lower (or inferior) part:
- The superior mediastinum starts at the superior thoracic aperture and ends at the thoracic plane.
- The inferior mediastinum from this level to the diaphragm. This lower part is subdivided into three regions, all relative to the pericardium – the anterior mediastinum being in front of the pericardium, the middle mediastinum contains the pericardium and its contents, and the posterior mediastinum being behind the pericardium.[6]
Anatomists, surgeons, and clinical radiologists compartmentalize the mediastinum differently. For instance, in the radiological scheme of Felson, there are only three compartments (anterior, middle, and posterior), and the heart is part of the middle (inferior) mediastinum.[7][page needed]
Thoracic plane
[edit]The transverse thoracic plane, thoracic plane, plane of Louis or plane of Ludwig is an important anatomical plane at the level of the sternal angle and the T4/T5 intervertebral disc.[8][9][10] It serves as an imaginary boundary that separates the superior and inferior mediastinum.[8][9][10]
A number of important anatomical structures and transitions occur at the level of the thoracic plane, including:
- The carinal bifurcation of the trachea into the left and right main bronchi.
- The left recurrent laryngeal nerve branching off the left vagus nerve and hooking under the ligamentum arteriosum between the aortic arch above and the pulmonary trunk below.
- The starting of the cardiac plexus.
- The azygos vein arching over the right main bronchus and joining into the superior vena cava.
- The thoracic duct crossing the midline from right to left behind the esophagus
- The end of the pretracheal and prevertebral fasciae.
Superior mediastinum
[edit]The superior mediastinum is bounded:
- superiorly by the thoracic inlet, the upper opening of the thorax;
- inferiorly by the transverse thoracic plane. which is an imaginary plane passing from the sternal angle anteriorly to the lower border of the body of the 4th thoracic vertebra posteriorly;
- laterally by the pleurae;
- anteriorly by the manubrium of the sternum;
- posteriorly by the first four thoracic vertebral bodies.
Contents
|
|---|
 
|
Inferior mediastinum
[edit]Anterior inferior mediastinum
[edit]Is bounded:
- laterally by the pleurae;
- posteriorly by the pericardium;[4]
- anteriorly by the sternum,[4] the left transversus thoracis and the fifth, sixth, and seventh left costal cartilages.
Contents
|
|---|
|
Middle inferior mediastinum
[edit]Bounded: pericardial sac – It contains the vital organs and is classified into the serous and fibrous pericardium.[4]
Contents
|
|---|
|
Posterior inferior mediastinum
[edit]Is bounded:
- Anteriorly by (from above downwards): bifurcation of trachea; pulmonary vessels; fibrous pericardium and posterior sloping surface of diaphragm
- Inferiorly by the thoracic surface of the diaphragm (below);
- Superiorly by the transverse thoracic plane;
- Posteriorly by the bodies of the vertebral column from the lower border of the fifth to the twelfth thoracic vertebra (behind);[4]
- Laterally by the mediastinal pleura (on either side).
Contents
|
|---|
|
-
A transverse section of the thorax, showing the contents of the middle and the posterior mediastinum.
Clinical significance
[edit]
The mediastinum is frequently the site of involvement of various tumors:
- Anterior mediastinum: substernal thyroid goiters, lymphoma, thymoma, and teratoma.
- Middle mediastinum: lymphadenopathy, metastatic disease such as from small cell carcinoma from the lung.
- Posterior mediastinum: Neurogenic tumors, either from the nerve sheath (mostly benign) or elsewhere (mostly malignant).
Mediastinitis is inflammation of the tissues in the mediastinum, usually bacterial and due to rupture of organs in the mediastinum. As the infection can progress very quickly, this is a serious condition.
Pneumomediastinum is the presence of air in the mediastinum, which in some cases can lead to pneumothorax, pneumoperitoneum, and pneumopericardium if left untreated. However, that does not always occur and sometimes those conditions are actually the cause, not the result, of pneumomediastinum. These conditions frequently accompany Boerhaave syndrome, or spontaneous esophageal rupture.
Widening
[edit]| Widened mediastinum | |
|---|---|
| Other names | Mediastinal widening |
 | |
| Widened mediastinum in a patient with achalasia | |
Widened mediastinum/mediastinal widening is where the mediastinum has a width greater than 6 cm on an upright PA chest X-ray or 8 cm on supine AP chest film.[11]
A widened mediastinum can be indicative of several pathologies:[12][13]
- aortic aneurysm[14]
- aortic dissection[15]
- aortic unfolding
- aortic rupture
- hilar lymphadenopathy
- anthrax inhalation - a widened mediastinum was found in 7 of the first 10 victims infected by anthrax (Bacillus anthracis) in 2001.[16]
- esophageal rupture - presents usually with pneumomediastinum and pleural effusion. It is diagnosed with water-soluble swallowed contrast.
- mediastinal mass
- mediastinitis
- cardiac tamponade[17]
- pericardial effusion
- thoracic vertebrae fractures in trauma patients.
See also
[edit]- Mediastinum testis (unrelated structure in the scrotum)
- Mediastinal germ cell tumor
- Mediastinitis
- Mediastinal tumor
- List of anatomy mnemonics#Mediastinum
References
[edit]![]() This article incorporates text in the public domain from page 1090 of the 20th edition of Gray's Anatomy (1918)
This article incorporates text in the public domain from page 1090 of the 20th edition of Gray's Anatomy (1918)
- ^ "A index W LA".
- ^ "Mediastinum dictionary definition - mediastinum defined". www.yourdictionary.com.
- ^ Fong, K.M.; Windsor, M.; Bowman, R.V.; Duhig, E. (2006). "TUMORS, MALIGNANT | Carcinoma, Lymph Node Involvement". Encyclopedia of Respiratory Medicine. pp. 366–373. doi:10.1016/b0-12-370879-6/00407-5. ISBN 978-0-12-370879-3.
- ^ a b c d e f Ng, Wai-Kuen (2008). "Mediastinum". Comprehensive Cytopathology. pp. 773–809. doi:10.1016/B978-141604208-2.10026-0. ISBN 978-1-4160-4208-2.
- ^ Cheng, Guang-Shing; Varghese, Thomas K.; Park, David R. (2016). "Mediastinal Tumors and Cysts". Murray and Nadel's Textbook of Respiratory Medicine. pp. 1478–1495.e38. doi:10.1016/B978-1-4557-3383-5.00083-X. ISBN 978-1-4557-3383-5.
- ^ Breul, Rainer (2012). "The deeper fasciae of the neck and ventral torso". Fascia: The Tensional Network of the Human Body. pp. 45–52. doi:10.1016/b978-0-7020-3425-1.00041-6. ISBN 978-0-7020-3425-1.
- ^ Goodman, Lawrence. Felson's Principles of Chest Roentgenology.
- ^ a b "Thoracic Wall, Pleura, and Pericardium – Dissector Answers". Archived from the original on September 1, 2012.
- ^ a b "Cell Biology and Anatomy - School of Medicine - University of South Carolina". dba.med.sc.edu. Archived from the original on 2006-09-05. Retrieved 2014-09-22.
- ^ a b "UAMS Department of Neurobiology and Developmental Sciences - Topographical Anatomy of the Thorax". anatomy.uams.edu. Archived from the original on 2004-08-17. Retrieved 2014-09-22.
- ^ D'Souza, Donna; Le, Lam; Hacking, Craig (2 May 2008). "Thoracic aortic injury". Radiopaedia.org. doi:10.53347/rID-2171.
- ^ Geusens; Pans, S.; Prinsloo, J.; Fourneau, I. (2005). "The widened mediastinum in trauma patients". European Journal of Emergency Medicine. 12 (4): 179–184. doi:10.1097/00063110-200508000-00006. PMID 16034263.
- ^ Richardson; Wilson, M. E.; Miller, F. B. (1990). "The widened mediastinum. Diagnostic and therapeutic priorities". Annals of Surgery. 211 (6): 731–736, discussion 736–7. doi:10.1097/00000658-199006000-00012. PMC 1358125. PMID 2357135.
- ^ Chandra, Sumesh; Laor, Yehuda G. (April 1975). "Lung scan and wide mediastinum". Journal of Nuclear Medicine: Official Publication, Society of Nuclear Medicine. 16 (4): 324–325. PMID 1113190.
- ^ von Kodolitsch Y, Nienaber C, Dieckmann C, Schwartz A, Hofmann T, Brekenfeld C, Nicolas V, Berger J, Meinertz T (2004). "Chest radiography for the diagnosis of acute aortic syndrome". Am J Med. 116 (2): 73–7. doi:10.1016/j.amjmed.2003.08.030. PMID 14715319.
- ^ Jernigan JA, Stephens DS, Ashford DA, et al. (2001). "Bioterrorism-related inhalational anthrax: the first 10 cases reported in the United States". Emerging Infect. Dis. 7 (6): 933–44. doi:10.3201/eid0706.010604. PMC 2631903. PMID 11747719.
- ^ Naudé, Gideon P.; Bongard, Fred S.; Demetriades, Demetrios (2003). Trauma Secrets. Hanley & Belfus. pp. 95–. ISBN 978-1-56053-506-5.
External links
[edit]- Anatomy figure: 21:01-03 at Human Anatomy Online, SUNY Downstate Medical Center – "Divisions of the mediastinum."
- Anatomy figure: 21:02-03 at Human Anatomy Online, SUNY Downstate Medical Center – "The anatomical divisions of the inferior mediastinum."
- thoraxlesson3 at The Anatomy Lesson by Wesley Norman (Georgetown University) – "Subdivisions of the Thoracic Cavity"
Mediastinum
View on GrokipediaOverview
Definition and Location
The mediastinum is defined as the central compartment of the thoracic cavity, situated between the pleural sacs of the lungs and extending from the thoracic inlet superiorly to the diaphragm inferiorly.[1] This midline space occupies the thoracic region, separating the left and right lungs while housing essential structures such as the heart, major blood vessels, trachea, esophagus, thymus, and lymph nodes.[3][4] The term and its anatomical conceptualization were described by Henry Gray in his 1858 publication Anatomy, Descriptive and Surgical, where it was portrayed as the interpleural space containing thoracic viscera excluding the lungs, positioned between the right and left pleurae in the median sagittal plane.[5] Subsequent refinements to this definition have incorporated advancements in imaging modalities like computed tomography (CT), enabling more precise delineation of its compartments for clinical applications such as mass diagnosis and surgical planning.[6] Functionally, the mediastinum serves to protect and support the thoracic viscera by providing a stable, connective tissue-enclosed passageway amid the dynamic respiratory movements of the surrounding lungs.[2] It is conventionally divided into superior and inferior portions, with the latter further subdivided into anterior, middle, and posterior compartments, though modern imaging-based systems emphasize prevascular, visceral, and paravertebral zones for enhanced diagnostic utility.[6]Boundaries and Divisions
The mediastinum occupies the central compartment of the thoracic cavity, bounded anteriorly by the sternum and the costal cartilages from the second to the fourth ribs, which provide a rigid osseocartilaginous framework. Posteriorly, it is delimited by the vertebral column, encompassing the bodies of the thoracic vertebrae and the intervertebral discs from T1 to T12, forming a stable bony posterior wall. Laterally, the boundaries are defined by the parietal pleura of both lungs, which separate the mediastinum from the pleural cavities and allow for independent lung expansion.[1][7] Superiorly, the mediastinum is demarcated by the plane of Louis, also known as the sternal angle or angle of Louis, which corresponds to the junction of the manubrium and body of the sternum at the level of the T4/T5 intervertebral disc; this transverse plane separates the superior mediastinum from the inferior mediastinum below. Inferiorly, the boundary is formed by the thoracic surface of the diaphragm, which domes upward to approximately the T12 vertebral level at its central tendon, marking the transition to the abdominal cavity. These boundaries enclose a dynamic space that accommodates vital thoracic structures while maintaining separation from adjacent compartments.[1][7] The standard anatomical divisions of the mediastinum organize this space into the superior mediastinum, extending from the thoracic inlet down to the plane of Louis, and the inferior mediastinum, which spans from that plane to the diaphragm. The inferior mediastinum is further partitioned into three compartments: the anterior mediastinum, located between the sternum and the pericardium; the middle mediastinum, centered on the pericardial sac containing the heart; and the posterior mediastinum, situated between the pericardium and the vertebral column. These subdivisions are primarily conceptual, with the pericardium serving as the key physical landmark separating the anterior and middle from the posterior regions, though no complete septa exist across the entire space.[1][7] This compartmental framework aids in clinical and surgical navigation by providing a standardized reference for localizing structures and pathologies within the thorax. Originating from 19th-century cadaveric dissections that emphasized gross anatomical landmarks, as initially outlined in seminal works like Henry Gray's Anatomy (1858), the divisions have evolved with advancements in imaging; modern CT-based classifications, such as the International Thymic Malignancy Interest Group (ITMIG) model introduced in 2014, refine these into three compartments (prevascular, visceral, and paravertebral) to better align with cross-sectional radiology and improve diagnostic precision.[8]Anatomy
Superior Mediastinum
The superior mediastinum is the uppermost compartment of the mediastinal space, serving as a critical passageway connecting the neck and thoracic cavity. It extends superiorly from the thoracic inlet to the transverse plane at the level of the sternal angle (angle of Louis) and the T4/T5 intervertebral disc, with lateral boundaries formed by the mediastinal pleurae, an anterior limit at the manubrium of the sternum, and a posterior boundary along the anterior surfaces of the upper four thoracic vertebrae.[1][9] This region facilitates the transit of major neurovascular structures from the neck into the thorax, accommodating dynamic arrangements essential for systemic circulation and innervation. Key contents of the superior mediastinum include portions of several vital structures. The thymus gland occupies a central position, particularly in younger individuals, though it may involute with age. The upper segments of the trachea and esophagus traverse this space posteriorly, maintaining patency for air and food passage. The thoracic duct, the primary lymphatic vessel, ascends along the posterior aspect, draining lymph from the lower body into the venous system.[1][10] Vascular elements dominate the superior mediastinum, with intricate arrangements supporting venous return and arterial distribution. The right and left brachiocephalic veins form by the union of the internal jugular and subclavian veins posterior to the sternoclavicular joints; the left brachiocephalic vein, longer at approximately 6-7 cm, crosses midline anterior to the aortic arch to join the right, forming the superior vena cava, whose uppermost portion resides here. The aortic arch originates from the ascending aorta and gives rise to three major branches: the brachiocephalic trunk (supplying the right subclavian and common carotid arteries), the left common carotid artery, and the left subclavian artery, all branching within this compartment to deliver oxygenated blood to the head, neck, and upper limbs. The arch of the azygos vein also enters the superior vena cava at the right tracheobronchial angle.[1][10][9] Neural components include the bilateral phrenic nerves (arising from C3-C5 roots), which descend anterior to the subclavian arteries and root of the lung to innervate the diaphragm and pericardium, and the vagus nerves (cranial nerve X), which course posteriorly to the brachiocephalic veins, giving off recurrent laryngeal branches and contributing to the cardiac, pulmonary, and esophageal plexuses. The sympathetic trunks lie posterolaterally, providing autonomic innervation to thoracic viscera.[1][10][9] Lymphatic drainage in the superior mediastinum is mediated by paratracheal and tracheobronchial lymph nodes, which receive afferents from the lungs, trachea, and esophagus before channeling into the thoracic duct or right lymphatic duct.[10] In clinical practice, magnetic resonance imaging (MRI) is particularly valuable for evaluating vascular anomalies in the superior mediastinum, such as malformations causing mass effect on adjacent structures; T2-weighted sequences reveal hyperintense lesions with flow voids, aiding precise diagnosis and surgical planning.[11]Anterior Mediastinum
The anterior mediastinum constitutes the smallest compartment of the inferior mediastinum, situated between the body of the sternum anteriorly and the pericardium posteriorly. It extends from the plane of the sternal angle (angle of Louis) superiorly to the diaphragm inferiorly, with lateral boundaries formed by the mediastinal pleurae and anterior limits including the transversus thoracis muscles and the fifth, sixth, and seventh left costal cartilages.[12][1] This compartment primarily contains loose areolar connective tissue, which includes the sternopericardial ligaments, small fat deposits, and lymphatic structures such as anterior mediastinal lymph nodes that drain to the parasternal nodes along the internal thoracic vessels. The internal thoracic (mammary) arteries and veins course through this space, providing vascular supply to the anterior thoracic wall. Key among its contents is the thymus gland, whose main body resides here in children and adolescents but involutes with age, undergoing fatty replacement in adults; thymic remnants or vestigial tissue may persist.[12][1][1] Embryologically, the thymus originates from endodermal thickenings in the third pharyngeal pouch around the sixth week of gestation, forming bilateral thymopharyngeal ducts that descend caudally and medially to fuse in the anterior mediastinum by the eighth week, with the ducts subsequently involuting. The gland reaches peak size at puberty before atrophying, often leaving only fatty or cystic remnants derived from embryonic structures like the ultimobranchial body. Variations include ectopic thymic tissue, which may occur along the descent path due to incomplete migration, such as in the neck or intrathyroidal locations, with a prevalence of up to 0.99% for intrathyroidal cases in pediatric populations; such anomalies are typically asymptomatic but can mimic masses on imaging.[13][14]Middle Mediastinum
The middle mediastinum constitutes the central division of the inferior mediastinum, encompassing the heart and its enveloping pericardium as primary structures. It is bounded anteriorly and posteriorly by the pericardium, laterally by the mediastinal pleura of the lungs, superiorly by the transverse plane from the sternal angle to the T4 vertebral level, and inferiorly by the superior surface of the diaphragm. This compartment extends from the plane of Louis (sternal angle) to the diaphragm base, forming a protective enclosure for vital cardiovascular elements.[15][1] Central to the middle mediastinum is the heart, including all four chambers (right and left atria, right and left ventricles), positioned within the pericardium. The pericardium comprises an outer fibrous layer—a tough, inelastic connective tissue sac continuous with the diaphragm's central tendon and great vessels—and an inner serous layer, divided into parietal (lining the fibrous layer) and visceral (epicardium adhering to the heart) components. The narrow pericardial cavity between these serous layers contains 15–50 mL of lubricating fluid, enabling frictionless cardiac movement. The phrenic nerves traverse the pericardial surfaces laterally, providing sensory innervation to the pericardial layers.[16][17][1] Pericardial recesses include the transverse and oblique sinuses, formed by serous pericardium reflections. The transverse sinus lies superior to the left atrium, posterior to the ascending aorta and pulmonary trunk, and anterior to the superior vena cava and atria, creating a passageway that separates great vessel outflows from inflows. The oblique sinus forms a J-shaped cul-de-sac posterior to the left atrium, bounded by the pulmonary veins anteriorly and the inferior vena cava inferiorly, extending between the visceral and parietal layers. These sinuses and the pericardial cavity represent potential spaces where fluid can accumulate, as in pericardial effusions, leading to increased intrapericardial pressure.[17][16][18] Key vascular elements encompass the ascending aorta emerging from the aortic root at the left ventricular base, the pulmonary trunk arising from the right ventricular outflow tract and dividing into the pulmonary arteries, the lower segment of the superior vena cava, the superior portion of the inferior vena cava, and the azygos vein's arch crossing the right tracheobronchial region. The aortic root and pulmonary valve are situated centrally within the pericardial sac, optimizing hemodynamic efficiency. These vessels link to continuations from the superior mediastinum.[19][15][1] Innervation of the middle mediastinum is biphasic, with parasympathetic input from vagus nerve branches forming the cardiac plexus to influence heart rate and conduction, and sympathetic supply from T1–T4 chain ganglia also via the cardiac plexus for acceleratory effects. The phrenic nerves (C3–C5 origins) provide additional somatic sensory fibers to the pericardium and motor supply to the diaphragm. Echocardiography offers detailed visualization of these structures, using views like parasternal long-axis (for aortic root and effusions), parasternal short-axis (for chambers and valves), apical four-chamber (for atrial and ventricular assessment), and subcostal (for inferior pericardium), where effusions manifest as echo-free spaces exceeding 15–35 mL.[1][15][20]Posterior Mediastinum
The posterior mediastinum is the compartment of the inferior mediastinum situated posterior to the pericardium and anterior to the vertebral column, extending superiorly from the plane of Louis (sternal angle at the T4/T5 intervertebral disc level) to the diaphragm inferiorly.[21] Laterally, it is bounded by the mediastinal pleura, enclosing structures involved in vascular descent, esophageal transit, and lymphatic drainage.[22] This region facilitates the passage of key conduits from the thorax to the abdomen, distinct from the cardiac focus of the adjacent middle mediastinum.[23] Key contents of the posterior mediastinum include the thoracic portion of the esophagus, the descending thoracic aorta, the azygos system of veins (comprising the azygos vein, hemiazygos vein, and accessory hemiazygos vein), the thoracic duct, and neural elements such as the vagus nerves, sympathetic trunks, and splanchnic nerves.[24] The descending thoracic aorta originates from the aortic arch at the T4/T5 level, courses posteriorly to the left of the vertebral column, and gives off branches including esophageal, bronchial, and posterior intercostal arteries before passing through the aortic hiatus at T12.[25] The azygos system drains venous blood from the posterior thoracic and abdominal walls, with the azygos vein ascending on the right side to join the superior vena cava at T4, while the hemiazygos and accessory hemiazygos veins on the left cross midline to anastomose with it.[21] The thoracic esophagus, approximately 20-25 cm in length, traverses the posterior mediastinum from the T1 level to the T10/T11 esophageal hiatus, featuring two physiological constrictions: at the broncho-aortic crossing (T4/T5 level), and at the diaphragmatic hiatus (T10).[23] Its muscularis externa transitions from striated muscle in the upper third (innervated by somatic fibers via the vagus nerve) to smooth muscle in the lower two-thirds (controlled by autonomic fibers), enabling peristaltic propulsion.[26] Arterial supply derives primarily from esophageal branches of the descending thoracic aorta in the middle third, supplemented by the inferior thyroid artery superiorly and left gastric artery inferiorly; venous drainage follows similar segmental patterns into the azygos system and portal vein.[23] The vagus nerves (CN X) descend bilaterally, forming the esophageal plexus around the mid-thoracic esophagus to provide parasympathetic innervation, while the sympathetic trunks lie posterolaterally with greater and lesser splanchnic nerves (T5-T9 and T10-T12 origins, respectively) piercing the crura to reach abdominal viscera.[27] The thoracic duct, the primary lymphatic vessel, originates from the cisterna chyli at the L1/L2 vertebral level in the abdomen, ascends through the aortic hiatus into the posterior mediastinum between the descending aorta and azygos vein, and courses posterior to the esophagus before arching laterally at T5 to drain into the left subclavian vein junction.[28] It conveys lymph and chyle from approximately 75% of the body, including lipids absorbed from the intestines.[29] Clinically, the posterior mediastinum's structures are prone to specific pathologies; for instance, portal hypertension can lead to esophageal varices in the lower thoracic esophagus due to portosystemic shunts via submucosal veins, risking rupture and hemorrhage.[23] Injuries to the thoracic duct, often from trauma, surgery (e.g., esophageal or aortic procedures), or malignancy, may result in chylothorax, a chyle accumulation in the pleural space causing respiratory compromise.[28]Clinical Significance
Mediastinal Masses and Tumors
Mediastinal masses and tumors encompass a diverse group of neoplastic lesions arising within the mediastinal compartments, classified primarily as anterior (prevascular), middle (visceral), and posterior (paravertebral) based on the International Thymic Malignancy Interest Group (ITMIG) system. These lesions vary in etiology, with benign tumors often congenital or developmental in origin, such as teratomas, while malignant ones include thymic carcinomas and lymphomas, frequently linked to genetic alterations or lymphoid proliferation. Incidence is low overall, representing less than 1% of all tumors, but anterior compartment involvement predominates, accounting for approximately 60% of cases in adults.[30][31][32] In the anterior mediastinum, common masses include thymomas, which comprise 20-50% of lesions and typically affect adults aged 40-60, often presenting as benign or low-grade malignancies associated with paraneoplastic syndromes like myasthenia gravis; teratomas, mature and benign in 70-80% of cases, arising from germ cell migration errors in young adults; thyroid goiters extending retrosternally; and lymphomas, particularly Hodgkin and non-Hodgkin types, which are malignant in nearly all instances and peak in younger patients. The "terrible T's" mnemonic highlights these thymic, teratoma, thyroid, and T-cell lymphoma entities as representative anterior pathologies. Middle mediastinal tumors are less common and often benign, including bronchogenic and pericardial cysts (50-60% of compartment masses), which are congenital foregut malformations, though malignant esophageal tumors like adenocarcinomas can occur. Posterior masses are dominated by neurogenic tumors, such as schwannomas and neurofibromas, which account for over 60% of lesions and are benign in 70% of cases, originating from neural crest derivatives; esophageal tumors, including gastrointestinal stromal tumors (GISTs), represent malignant subsets.[30][31][32] Etiologically, benign masses like teratomas and neurogenic tumors often stem from embryonic remnants, while malignant ones, such as germ cell tumors in young adults (15-35 years) and thymic carcinomas, involve oncogenic drivers including TP53 mutations (prevalent in approximately 25-30% of thymic carcinoma cases, correlating with poor prognosis) and KIT mutations (found in 10-20% of thymic carcinomas and esophageal GISTs, enabling targeted therapies like imatinib). Many masses are asymptomatic, discovered incidentally on imaging, but symptomatic cases arise from compression of adjacent structures, leading to dyspnea (in 30-50% of large tumors), superior vena cava (SVC) syndrome (particularly with anterior lymphomas or thymomas), cough, or chest pain.[30][31][33] Diagnosis relies on multimodal imaging and biopsy for confirmation and staging. Computed tomography (CT) is the initial modality for localization and characterization, revealing density patterns (e.g., fat in teratomas), while positron emission tomography-CT (PET-CT) assesses malignancy via FDG uptake, aiding lymphoma staging and distinguishing benign from malignant thymic lesions. CT-guided fine-needle aspiration biopsy provides tissue diagnosis in 80-90% of cases, though video-assisted thoracoscopic surgery (VATS) is preferred for anterior or equivocal lesions to avoid complications like pneumothorax. Staging follows Masaoka-Koga or TNM systems, with PET-CT crucial for detecting metastases in thymic carcinomas.[30][31][32] Recent advances emphasize molecular profiling and targeted therapies. In thymic carcinomas, KIT mutations drive responsiveness to tyrosine kinase inhibitors, with response rates up to 50% in mutated subsets, while CYLD mutations may predict immunotherapy efficacy. Immunotherapy, particularly PD-1 inhibitors like pembrolizumab, has emerged in the 2020s for advanced thymic carcinoma post-platinum chemotherapy, showing objective response rates of 20-25% and progression-free survival of 4-6 months in phase II trials, though contraindicated in thymomas due to high (40-70%) immune-related adverse events like myositis. Combination approaches, such as avelumab with axitinib, yield higher response rates (up to 34%), highlighting ongoing shifts toward personalized treatment.[33][34][35]Widening and Shift
Widening of the mediastinum refers to an abnormal increase in the transverse diameter of the mediastinal silhouette on chest radiography, typically defined as greater than 8 cm measured at the level of the aortic knob on a posteroanterior (PA) view.[36][37][38] The normal mediastinal width ranges from 6 to 8 cm in adults on a properly positioned PA chest X-ray (CXR), though measurements can vary slightly based on patient body habitus and inspiratory effort.[39][40] This radiographic sign often prompts further evaluation, as it may indicate underlying pathology, but technical factors such as supine anteroposterior (AP) projections or poor inspiration can artifactually magnify the appearance.[41][42] Common causes of mediastinal widening include vascular emergencies such as aortic dissection and thoracic aortic aneurysm, which expand the silhouette due to intramural hematoma or vessel dilatation.[43][40][44] Traumatic injuries, particularly blunt aortic injury from deceleration mechanisms like motor vehicle collisions, lead to mediastinal hematoma and acute widening.[43][45] Lymphadenopathy, often from reactive or neoplastic processes, can also contribute by enlarging mediastinal nodes, though this is distinguished from vascular causes via cross-sectional imaging.[46][37] Mediastinal shift describes the deviation of central structures, such as the trachea or cardiac silhouette, from the midline on imaging, resulting from unequal intrathoracic pressures.[47] This shift occurs away from the affected side in conditions like tension pneumothorax, where accumulating air increases ipsilateral pressure and compresses the contralateral lung.[48][49] Large pleural effusions cause contralateral shift by mass effect, displacing the trachea and heart shadow.[50] Lung collapse, such as from atelectasis or endobronchial obstruction, produces ipsilateral shift as volume loss pulls mediastinal structures toward the affected hemithorax.[47][51] Diagnostic criteria for mediastinal widening on CXR involve measuring the horizontal distance from the right heart border to the left mediastinal margin at the aortic knob, with values exceeding 8 cm or more than one-third of the thoracic diameter considered abnormal, particularly on supine films.[36][52] On computed tomography (CT), which provides superior anatomic detail, widening is assessed by evaluating mediastinal contours, vessel caliber, and tissue density; for instance, mediastinal fat typically measures -50 to -100 Hounsfield units (HU), while soft-tissue components range from 40 to 60 HU, aiding differentiation of hematoma (30-60 HU) from physiologic fat.[39][53][30] Clinically, acute mediastinal widening correlates with life-threatening trauma, such as blunt aortic injury, where up to 80-90% of cases are fatal without prompt intervention, often detected within hours of deceleration trauma.[54][55] Chronic widening is frequently linked to progressive aortic aneurysms, which may remain asymptomatic until rupture risk escalates, with diameters exceeding 5.5 cm indicating higher intervention thresholds per guidelines.[56][44] By 2025, artificial intelligence (AI)-assisted tools have emerged to enhance detection of mediastinal widening on CXR, with algorithms like Lunit INSIGHT CXR achieving high sensitivity for abnormalities including widening, pneumothorax, and effusions, improving radiologist workflow in emergency settings.[57][58] These deep learning models, trained on large datasets, localize widening with accuracy comparable to specialists, facilitating faster triage.[59][60]Infections and Inflammation
Infections and inflammation of the mediastinum encompass a range of conditions that can compromise vital thoracic structures, including acute and chronic forms of mediastinitis as well as granulomatous diseases like sarcoidosis. These processes often arise from direct spread of pathogens or systemic inflammatory responses, leading to significant morbidity if not promptly addressed. Acute mediastinitis, in particular, is a life-threatening emergency characterized by rapid progression, while chronic variants involve progressive fibrosis.[61] Mediastinitis refers to inflammation or infection of the mediastinal tissues, divided into acute and chronic subtypes based on etiology and tempo. Acute mediastinitis most commonly occurs postoperatively following sternotomy for cardiac surgery, with an incidence of 0.5% to 2.5%, or as descending necrotizing mediastinitis originating from odontogenic infections in the oropharynx that spread via fascial planes. Chronic mediastinitis, often fibrosing, is typically triggered by granulomatous infections such as histoplasmosis, leading to scarring and compression of mediastinal structures over months to years.[61][62][63] Pathophysiologically, acute forms are predominantly bacterial, with Staphylococcus aureus (including methicillin-resistant strains) accounting for 60% to 80% of postoperative cases due to surgical contamination, while polymicrobial infections involving anaerobes predominate in descending types. Fungal pathogens like Aspergillus are rarer but occur in immunocompromised patients, contributing to necrotizing tissue destruction and potential lymphatic spread through posterior mediastinal channels. In chronic fibrosing mediastinitis, Histoplasma capsulatum induces a granulomatous response that evolves into dense fibrosis, narrowing vascular and airway lumens.[61][64] Symptoms of mediastinitis include high fever, retrosternal chest pain, dysphagia, and odynophagia, often accompanied by neck swelling in descending cases; respiratory distress and tachycardia may signal progression. Complications such as sepsis, mediastinal abscesses, and multiorgan failure are common, with mortality rates ranging from 20% to 40% in acute descending necrotizing mediastinitis despite intervention, and lower (1% to 14%) in postoperative variants.[61][65][66] Treatment of acute mediastinitis emphasizes broad-spectrum intravenous antibiotics (e.g., vancomycin plus piperacillin-tazobactam) initiated empirically, followed by culture-guided adjustment, alongside urgent surgical drainage and debridement to remove necrotic tissue. For chronic fibrosing mediastinitis, management is supportive, with antifungal therapy (e.g., itraconazole for histoplasmosis) and potential stenting for symptomatic compression; corticosteroids may be used adjunctively in inflammatory flares. Early diagnosis via contrast-enhanced CT, which reveals mediastinal air-fluid collections or widening, is critical to improving outcomes.[61][62][67] Beyond mediastinitis, sarcoidosis frequently manifests as mediastinal inflammation through noncaseating granulomatous lymphadenopathy, affecting over 90% of patients with pulmonary involvement and often presenting as bilateral hilar and right paratracheal enlargement on imaging. This granulomatous process, driven by macrophage and T-cell clusters, can cause cough, dyspnea, or chest discomfort in symptomatic cases (about 50%), though many remain asymptomatic; treatment involves glucocorticoids like prednisone (20-40 mg/day initially) for progressive disease to suppress inflammation.[68][68] Purulent pericarditis represents another inflammatory condition with mediastinal extension, where bacterial infection (e.g., from Streptococcus or gram-negatives) spreads contiguously from mediastinal or pulmonary sources, leading to pus accumulation and widespread thoracic involvement. This rare entity carries high mortality due to its association with extensive mediastinal sepsis, managed with antibiotics and pericardial drainage, though outcomes remain poor without addressing the underlying source.[69][69]References
- https://wikem.org/wiki/Widened_mediastinum