Recent from talks
Nothing was collected or created yet.
Peritonsillar abscess
View on Wikipedia| Peritonsillar abscess | |
|---|---|
| Other names | Quinsy, quinsey |
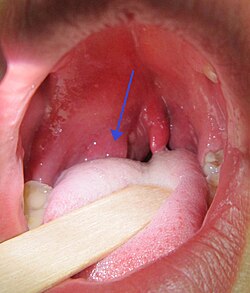 | |
| Right-sided peritonsillar abscess | |
| Specialty | Otorhinolaryngology |
| Symptoms | Fever, throat pain, trouble opening the mouth, change to the voice[1] |
| Complications | Blockage of the airway, aspiration pneumonitis[1] |
| Causes | Multiple types of bacteria[1] |
| Risk factors | Streptococcal pharyngitis[1] |
| Diagnostic method | Based on the symptoms[1] |
| Differential diagnosis | Retropharyngeal abscess, infectious mononucleosis, epiglottitis, cancer[1] |
| Treatment | Remove pus, antibiotics, fluids, pain medication, steroids[1] |
| Frequency | ~3 per 10,000 per year (USA)[1] |
A peritonsillar abscess (PTA), also known as a quinsy, is an accumulation of pus due to an infection behind the tonsil.[2] Symptoms include fever, throat pain, trouble opening the mouth, and a change to the voice.[1] Pain is usually worse on one side.[1] Complications may include blockage of the airway or aspiration pneumonitis.[1]
PTA is typically due to infection by several types of bacteria.[1] Often, it follows streptococcal pharyngitis.[1] They do not typically occur in those who have had a tonsillectomy.[1] Diagnosis is usually based on the symptoms.[1] Medical imaging may be done to rule out complications.[1]
Treatment is by removing the pus, antibiotics, sufficient fluids, and pain medication.[1] Steroids may also be useful.[1] Hospital admission is generally not needed.[1] In the United States, about 3 per 10,000 people per year are affected.[1] Young adults are most commonly affected.[1]
Signs and symptoms
[edit]Physical signs of a peritonsillar abscess include redness and swelling in the tonsillar area of the affected side and swelling of the jugulodigastric lymph nodes. The uvula may be displaced towards the unaffected side.[3]
Unlike tonsillitis, which is more common in children, PTA has a more even age spread, from children to adults. Symptoms start appearing two to eight days before the formation of an abscess. A progressively severe sore throat on one side and pain during swallowing (odynophagia) are usually the earliest symptoms. As the abscess develops, persistent pain in the peritonsillar area, fever, a general sense of feeling unwell, headache, and a distortion of vowels informally known as "hot potato voice" may appear. Neck pain associated with tender, swollen lymph nodes, referred ear pain, and foul breath are also common. While these signs may be present in tonsillitis itself, a PTA should be specifically considered if there is limited ability to open the mouth (trismus).[3]
Complications
[edit]While most people recover uneventfully, a wide range of possible complications may occur.[4] These may include:[1]
- Retropharyngeal abscess
- Extension of the abscess in other deep neck spaces leading to airway compromise (see Ludwig's angina)
- Airway obstruction
- Aspiration pneumonitis
- Lung abscess (following rupture)
- Sepsis
- Life-threatening hemorrhage (following erosion or septic necrosis into the carotid sheath of the neck)
- Glomerulonephritis and rheumatic fever (chronic complications of strep throat)
Difficulty swallowing can lead to decreased oral intake and dehydration.
Causes
[edit]PTA is usually a complication of an untreated or partially treated episode of acute tonsillitis. The infection, in these cases, spreads to the peritonsillar area (peritonsillitis). This region comprises loose connective tissue and is hence susceptible to the formation of an abscess. PTA can also occur de novo. Both aerobic and anaerobic bacteria can be causative. Commonly involved aerobic pathogens include Streptococcus, Staphylococcus and Haemophilus. The most common anaerobic species include Fusobacterium necrophorum, Peptostreptococcus, Prevotella species, and Bacteroides.[5][6][7][8][9][10]
Diagnosis
[edit]
Diagnosis is usually based on the symptoms.[1] Medical imaging may be done to rule out complications.[1] Medical imaging may include CT scan, MRI, or ultrasound is also useful in diagnosis.[1]
Treatment
[edit]Medical treatment with antibiotics, volume repletion with fluids, and pain medication is usually adequate, although in cases where airway obstruction or systemic sepsis occurs, surgical drainage may be necessary.[1][11] Corticosteroids may also be useful.[1] Hospital admission is generally not needed.[1]
Medication
[edit]The infection is frequently penicillin resistant.[1] There are several antibiotics options including amoxicillin/clavulanate, ampicillin/sulbactam, clindamycin, or metronidazole in combination with benzylpenicillin (penicillin G) or penicillin V.[1][12] Piperacillin/tazobactam may also be used.[1]
Surgery
[edit]The pus can be removed by several methods, including needle aspiration, incision and drainage, and tonsillectomy.[1] Incision and drainage may be associated with a lower chance of recurrence than needle aspiration, but the evidence is very uncertain. Needle aspiration may be less painful, but again, the evidence is very uncertain.[13]
Treatment can also be given while a patient is under anesthesia, but this is usually reserved for children or anxious patients. Tonsillectomy can be indicated if a patient has recurring peritonsillar abscesses or a history of tonsillitis. For patients with their first peritonsillar abscess, most ENT surgeons prefer to "wait and observe" before recommending tonsillectomy.[14]
Epidemiology
[edit]It is a commonly encountered otorhinolaryngological (ENT) emergency.[14]
The number of new cases per year of peritonsillar abscess in the United States has been estimated at approximately 30 cases per 100,000 people.[15] In a study in Northern Ireland, the number of new cases was 10 cases per 100,000 people per year.[16] In Denmark, the number of new cases is higher and reaches 41 cases per 100,000 people per year.[17] Younger children who develop a peritonsillar abscess are often immunocompromised and in them, the infection can cause airway obstruction.[18]
Etymology
[edit]The condition is often referred to as "quincy", "quinsy",[19] or "quinsey", anglicised versions of the French word esquinancie which was originally rendered as squinsey and subsequently quinsy.[20]
Notable cases
[edit]- Sultan Tekish of Kwarezm[21]
- Osceola[22]
- Michel de Montaigne[23]
- Dan Minogue, the captain/coach of the Australian Rules football team Richmond was rumoured to be dead a week before the 1920 VFL Grand Final, but in fact, was in his hometown of Bendigo recovering from quinsy.
- Eiichiro Oda, author of the best-selling One Piece manga, was hospitalized due to complications.[24]
- Ian Maclaren died of complications from quinsy while on a lecture tour of the United States.[25]
The ancient Roman goddess Angerona was claimed to cure quinsy (Latin angina) in humans and sheep.[26][27]
References
[edit]- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af Galioto NJ (April 2017). "Peritonsillar Abscess". American Family Physician. 95 (8): 501–506. PMID 28409615.
- ^ "Tonsillar Cellulitis and Tonsillar Abscess - Ear, Nose, and Throat Disorders - Merck Manuals Consumer Version". Merck Manuals Consumer Version. Archived from the original on 25 October 2017. Retrieved 24 October 2017.
- ^ a b "Peritonsillar Abscess". The Lecturio Medical Concept Library. Retrieved 4 October 2021.
- ^ Klug TE, Greve T, Hentze M (2020). "Complications of peritonsillar abscess". Annals of Clinical Microbiology and Antimicrobials. 19 (1): 32. doi:10.1186/s12941-020-00375-x. PMC 7391705. PMID 32731900.
- ^ Brook I, Frazier EH, Thompson DH (March 1991). "Aerobic and anaerobic microbiology of peritonsillar abscess". The Laryngoscope. 101 (3): 289–92. doi:10.1288/00005537-199103000-00012. PMID 2000017. S2CID 23505346.
- ^ Sakae FA, Imamura R, Sennes LU, Araújo Filho BC, Tsuji DH (2006). "Microbiology of peritonsillar abscesses". Brazilian Journal of Otorhinolaryngology. 72 (2): 247–51. doi:10.1016/S1808-8694(15)30063-X. PMC 9445683. PMID 16951860.
- ^ Gavriel H, Lazarovitch T, Pomortsev A, Eviatar E (January 2009). "Variations in the microbiology of peritonsillar abscess". European Journal of Clinical Microbiology & Infectious Diseases. 28 (1): 27–31. doi:10.1007/s10096-008-0583-6. PMID 18612664. S2CID 26365493.
- ^ Sunnergren O, Swanberg J, Mölstad S (2008). "Incidence, microbiology and clinical history of peritonsillar abscesses". Scandinavian Journal of Infectious Diseases. 40 (9): 752–5. doi:10.1080/00365540802040562. PMID 19086341. S2CID 40973972.
- ^ Klug TE, Henriksen JJ, Fuursted K, Ovesen T (May 2011). "Significant pathogens in peritonsillar abscesses". European Journal of Clinical Microbiology & Infectious Diseases. 30 (5): 619–27. doi:10.1007/s10096-010-1130-9. PMID 21181222. S2CID 6451474.
- ^ Powell EL, Powell J, Samuel JR, Wilson JA (September 2013). "A review of the pathogenesis of adult peritonsillar abscess: time for a re-evaluation". The Journal of Antimicrobial Chemotherapy. 68 (9): 1941–50. CiteSeerX 10.1.1.1001.2391. doi:10.1093/jac/dkt128. PMID 23612569.
- ^ Johnson RF (October 2017). "Emergency department visits, hospitalizations, and readmissions of patients with a peritonsillar abscess". Laryngoscope. 127 (Suppl 5): S1 – S9. doi:10.1002/lary.26777. PMID 28782104. S2CID 206205296.
- ^ Visvanathan V, Nix P (April 2010). "National UK survey of antibiotics prescribed for acute tonsillitis and peritonsillar abscess". The Journal of Laryngology and Otology. 124 (4): 420–3. doi:10.1017/S0022215109991939. PMID 19930783. S2CID 27721802.
- ^ Chang BA, Thamboo A, Burton MJ, Diamond C, Nunez DA, et al. (Cochrane ENT Group) (December 2016). "Needle aspiration versus incision and drainage for the treatment of peritonsillar abscess". The Cochrane Database of Systematic Reviews. 2016 (12) CD006287. doi:10.1002/14651858.CD006287.pub4. PMC 6463807. PMID 28009937.
- ^ a b Raut VV (2000). "Management of peritonsillitis/peritonsillar". Revue de Laryngologie - Otologie - Rhinologie. 121 (2): 107–10. PMID 10997070.
- ^ Johnson RF, Stewart MG (June 2005). "The contemporary approach to diagnosis and management of peritonsillar abscess". Current Opinion in Otolaryngology & Head and Neck Surgery. 13 (3): 157–60. doi:10.1097/01.moo.0000162259.42115.38. PMID 15908813. S2CID 38122236.
- ^ Hanna BC, McMullan R, Gallagher G, Hedderwick S (April 2006). "The epidemiology of peritonsillar abscess disease in Northern Ireland". The Journal of Infection. 52 (4): 247–53. doi:10.1016/j.jinf.2005.07.002. PMID 16125782.
- ^ Ehlers Klug T, Rusan M, Fuursted K, Ovesen T (November 2009). "Fusobacterium necrophorum: most prevalent pathogen in peritonsillar abscess in Denmark". Clinical Infectious Diseases. 49 (10): 1467–72. doi:10.1086/644616. PMID 19842975.
- ^ Hardingham M (May 1987). "Peritonsillar infections". Otolaryngologic Clinics of North America. 20 (2): 273–8. doi:10.1016/S0030-6665(20)31644-3. PMID 3474580.
- ^ "Peritonsillitis (Peritonsillar Cellulitis and Peritonsillar Abscess)". Marx: Rosen's Emergency Medicine (7th ed.). Mosby, An Imprint of Elsevier. 2009. Retrieved 8 July 2013.
- ^ Greene RG (1890). The International cyclopedia: a compendium of human knowledge, Volume 12. Mead Dodd. pp. 355–6. Archived from the original on 2016-09-25.
- ^ Juvaini Aa (1997). History of the World Conqueror. Manchester U.K.: Manchester University Press. p. 314.
- ^ Wickman PR (2006). Osceola's Legacy. University of Alabama Press. p. 144.
- ^ de Montaigne M (1877). "Essays of Michel de Montaigne". In William Carew Hazlitt (ed.). The Life of Montaigne. Vol. 1. Translated by Charles Cotton (Kindle ed.).
- ^ "NEWS: One Piece on Break". Viz Media. Archived from the original on 2013-06-07. Retrieved 2013-05-21.
- ^ Nicoll WR (1908). 'Ian Maclaren': Life of the Rev. John Watson, D.D. London: Hodder and Stoughton. p. 379.
- ^ "Classical World". Classical Association of the Atlantic States. February 13, 1918 – via Google Books.
- ^ "Angerona - Encyclopedia". theodora.com.
External links
[edit]Peritonsillar abscess
View on GrokipediaDefinition and pathophysiology
Definition
A peritonsillar abscess (PTA) is defined as a localized collection of pus within the peritonsillar space, situated between the tonsillar capsule and the superior constrictor muscle of the pharynx.[8] Also known historically as quinsy, this condition represents a suppurative complication typically arising from untreated or inadequately managed acute tonsillitis.[9] The peritonsillar space is a potential anatomical compartment bounded medially by the palatine tonsil, laterally by the superior pharyngeal constrictor muscle, and posteriorly by the palatopharyngeus muscle.[10] Abscess formation most commonly occurs at the superior pole of the tonsil, accounting for 41% to 70% of cases, with less frequent involvement at the inferior pole or mid-tonsillar region.[11] PTA must be differentiated from peritonsillar cellulitis, which involves inflammatory edema in the same space but lacks a discrete pus collection and is considered a precursor stage.[12]Pathophysiology
A peritonsillar abscess typically arises as a complication of acute bacterial tonsillitis, where the initial infection within the palatine tonsils leads to suppuration and pus accumulation in the peritonsillar space—a potential space between the tonsillar capsule and the superior constrictor muscle of the pharynx.[8] The process begins with bacterial invasion of the tonsillar parenchyma or crypts, progressing to localized infection when the pathogens breach the tonsillar barrier and enter the adjacent peritonsillar tissue.[1] Several mechanisms explain the spread of infection to the peritonsillar space. Direct extension from the tonsillar crypts is a primary pathway, allowing bacteria to invade the loose connective tissue laterally and superiorly. Additionally, involvement of the Weber glands—minor salivary glands located in the supratonsillar fossa—may serve as a nidus, where ductal obstruction leads to bacterial overgrowth and abscess formation.[8][1] The inflammatory response in peritonsillar abscess involves intense local edema, hyperemia, and tissue necrosis driven by the host immune reaction to bacterial toxins and enzymes. Mixed aerobic and anaerobic flora, often originating from the oral cavity, synergistically promote abscess maturation by producing proteolytic enzymes that liquefy tissue and facilitate pus formation. A fibrous capsule eventually forms around the necrotic debris and pus, walling off the infection but potentially delaying resolution due to reduced vascularity and poor antibiotic penetration in the avascular core.[8][1][13] The condition evolves through stages, starting as peritonsillar cellulitis—a diffuse, non-localized phlegmonous infection characterized by widespread inflammation without discrete pus collection. If untreated, this progresses to a mature abscess, marked by a fluctuant, encapsulated pus pocket, often influenced by factors such as impaired host immunity or persistent bacterial virulence that hinders clearance.[1][13]Clinical presentation
Signs and symptoms
Patients with peritonsillar abscess typically present with severe unilateral sore throat, often described as the most intense pain in the affected area.[14] This is accompanied by odynophagia, making swallowing extremely painful, and dysphagia, which contributes to reduced oral intake.[2] Fever is a common systemic symptom, frequently exceeding 38.5°C, along with malaise, fatigue, and headaches.[14] Voice changes are characteristic, including a muffled or "hot potato" voice due to peritonsillar swelling affecting resonance.[8] On physical examination, trismus is a hallmark sign, resulting from irritation of the internal pterygoid muscle and limiting mouth opening to less than 2 cm in many cases.[4] Tonsillar asymmetry is evident, with enlargement and bulging of the affected tonsil and soft palate toward the contralateral side, often accompanied by uvular deviation away from the abscess.[15] Unilateral exudate may be visible on the tonsil, and the soft palate appears erythematous and swollen.[8] Cervical lymphadenopathy, particularly tender anterior cervical nodes, is frequently observed ipsilateral to the abscess.[8] The combination of intense sore throat and trismus (inability to open the mouth) in individuals wearing orthodontic braces is a concerning symptom combination that may indicate a serious issue such as peritonsillar abscess (as a complication of tonsillitis), temporomandibular joint (TMJ) disorder, or other oral/dental infection. Orthodontic braces can contribute to oral irritation or hygiene issues but rarely cause this severity alone. Prompt medical or dental attention is recommended, as the Mayo Clinic advises seeing a doctor for sore throat with difficulty opening the mouth; self-diagnosis or delaying care should be avoided.[7] Associated features include drooling due to painful swallowing, halitosis from anaerobic infection, and referred otalgia to the ipsilateral ear.[14] The condition often progresses from initial symptoms of acute tonsillitis that fail to resolve, with worsening unilateral symptoms over 2-5 days.[8] Bilateral peritonsillar abscess is rare, occurring in less than 5% of cases, and presents with symmetric swelling, bilateral trismus, and muffled voice without uvular deviation.[16]Complications
Untreated or inadequately managed peritonsillar abscess can lead to serious local complications, primarily due to progressive swelling and potential rupture of the abscess. Airway obstruction is a rare but life-threatening outcome, resulting from extensive edema that compromises breathing, particularly when severe trismus limits mouth opening and exacerbates the risk. Rupture of the abscess into adjacent deep neck spaces may cause rapid spread of infection, potentially leading to Ludwig's angina, a bilateral cellulitis of the submandibular space that causes floor-of-mouth elevation and further airway compromise.[13][17][18] Systemic complications arise from dissemination of the infection beyond the peritonsillar region. Bacteremia and sepsis occur in a significant proportion of cases, with approximately half of patients meeting sepsis diagnostic criteria, often linked to factors like smoking or delayed antibiotic therapy. Extension into the parapharyngeal space can produce neck stiffness and further propagate infection, while deeper spread may result in descending mediastinitis, a severe necrotizing infection of the mediastinum reported in multiple cases.[19][20][21] Recurrent peritonsillar abscesses represent a common sequela, with an overall recurrence rate of approximately 10 to 15 percent in the absence of tonsillectomy, potentially evolving into chronic tonsillitis with repeated episodes of inflammation.[22] Rare complications include internal jugular vein thrombosis, a variant of Lemierre's syndrome involving septic thrombophlebitis, and intracranial extension such as brain abscess, both of which carry high morbidity. Immunocompromised patients face increased risk of severe morbidity from these infections due to impaired host defenses, often requiring more aggressive intervention.[21][23][24]Etiology and risk factors
Causes
Peritonsillar abscess (PTA) is primarily caused by bacterial infections originating from the oral flora, often progressing from an initial episode of acute tonsillitis. The condition typically arises when bacteria invade the peritonsillar space, leading to localized pus accumulation.[25] The predominant pathogen is Group A Streptococcus (Streptococcus pyogenes), which is identified in 20-30% of cases and is considered the most common aerobic isolate. Other frequent streptococcal species include Group C and G streptococci, as well as viridans group streptococci such as Streptococcus anginosus (formerly milleri group). Anaerobic bacteria play a significant role, with Fusobacterium necrophorum (prevalent in adolescents and young adults) and Prevotella species commonly isolated.[26][6][27] Most PTAs exhibit a polymicrobial nature, involving mixed aerobic and anaerobic infections derived from the normal tonsillar and oral microbiota. This combination facilitates abscess formation through synergistic bacterial interactions, where aerobes like streptococci may create conditions favorable for anaerobe proliferation in the tonsillar crypts.[6][4] PTA most commonly develops as a suppurative complication of bacterial pharyngitis or tonsillitis, allowing infection to spread from the tonsillar parenchyma to the peritonsillar space.[13][28] Less common causes include superinfections following viral upper respiratory infections, such as those associated with infectious mononucleosis, and rare non-infectious mimics like foreign bodies in the tonsillar area or neoplastic processes presenting similarly. However, these are exceptional, with bacterial etiology confirmed in the vast majority of instances.[4][8]Risk factors
Peritonsillar abscess (PTA) predominantly affects adolescents and young adults, with the highest incidence occurring between the ages of 15 and 30 years.[29] This age group experiences peak rates, potentially due to increased exposure to infectious agents in social settings and anatomical factors related to tonsillar development. The condition is slightly more common in males, particularly after adolescence, although the overall male-to-female ratio approximates 1:1, with females tending to present at younger ages.[30][29] A history of recurrent tonsillitis (such as five or more prior episodes) significantly predisposes individuals to PTA, as repeated episodes of tonsillar infection can lead to local tissue damage and bacterial superinfection. Additionally, a prior PTA episode markedly increases the likelihood of recurrence, with rates reported between 10% and 15%.[31][22] Behavioral and environmental factors play a key role in susceptibility. Smoking is a well-established risk factor, as it impairs mucosal defenses and promotes bacterial colonization in the oropharynx, with studies indicating a dose-dependent association. Poor oral hygiene, including significant periodontal disease, further heightens risk by facilitating polymicrobial infections. Seasonal viral infections, such as those caused by Epstein-Barr virus (EBV) or influenza, can act as triggers by inducing local immunosuppression and paving the way for secondary bacterial invasion.[26][13][26] Comorbidities and anatomical variations also contribute to PTA development. Immunosuppressive conditions, including HIV and diabetes mellitus, compromise host defenses, leading to higher susceptibility and more severe presentations; for instance, type 2 diabetes independently elevates PTA risk. Anatomical abnormalities, such as enlarged tonsils or deep tonsillar crypts, may obstruct drainage and promote abscess formation. Furthermore, individuals in low socioeconomic settings are at greater risk due to barriers to timely medical care, resulting in progression from uncomplicated tonsillitis to abscess.[13][32][33]Diagnosis
Clinical assessment
The clinical assessment of peritonsillar abscess begins with a detailed history taking to identify key features suggestive of the condition. Patients typically report a severe sore throat lasting several days, often preceded by tonsillitis, accompanied by fever, dysphagia, and the recent onset of trismus that limits mouth opening.[14] Systemic symptoms such as malaise and dehydration may also be elicited, helping to gauge severity and duration.[8] Physical examination focuses on intraoral inspection and palpation to detect characteristic abnormalities. Asymmetry in the oropharynx is common, with unilateral tonsillar enlargement, peritonsillar edema, and medial displacement of the soft palate or uvula toward the contralateral side; bimanual palpation along the superior tonsillar pole may reveal fluctuance indicating pus collection.[13] Trismus is assessed by measuring interincisor distance, where opening less than 2 cm is highly suggestive due to irritation of the internal pterygoid muscle.[34] A muffled "hot potato" voice and drooling may be noted during the exam.[8] The presumptive diagnosis relies on the constellation of these clinical signs, which collectively offer high diagnostic accuracy. For instance, the presence of uvula deviation, hot potato voice, and pharyngeal edema demonstrates a sensitivity of approximately 90% for peritonsillar abscess in recent prospective studies.[35] Trismus combined with peritonsillar swelling further supports suspicion.[14] Differential diagnosis during assessment includes distinguishing peritonsillar abscess from epiglottitis or retropharyngeal abscess, which may present with similar trismus but lack unilateral tonsillar asymmetry and show more diffuse airway involvement on exam.[13] Careful evaluation of voice quality and neck mobility aids in ruling out these alternatives.[8]Diagnostic tests
Diagnosis of peritonsillar abscess (PTA) often involves ancillary investigations to confirm the presence of a fluid collection and identify potential complications, particularly when clinical assessment is equivocal.[8] Imaging modalities play a key role in visualization, with intraoral ultrasound serving as a first-line, non-invasive option due to its portability and lack of radiation exposure. Ultrasound demonstrates high sensitivity, approximately 89-95%, for detecting fluid collections indicative of PTA, allowing for differentiation from peritonsillar cellulitis by identifying hypoechoic areas adjacent to the tonsil.[36][37] Its specificity ranges from 78-100%, making it effective for ruling out abscesses in emergency settings and guiding needle aspiration to avoid vascular structures like the carotid artery.[38] Computed tomography (CT) with contrast enhancement is reserved for cases with suspected complications, such as deep neck extension or airway involvement, revealing a rim-enhancing fluid collection adjacent to the enlarged tonsil with 100% sensitivity and 75% specificity.[21][39] Laboratory tests provide supportive evidence of infection but are not diagnostic on their own. A complete blood count (CBC) typically shows leukocytosis with a left shift, reflecting neutrophilia in response to bacterial infection.[40] Elevated C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) levels indicate systemic inflammation, often correlating with disease severity and hospitalization duration.[5] Throat swabs for culture or rapid antigen detection testing for group A Streptococcus have low yield in confirming PTA etiology, as surface cultures do not reliably sample the abscess cavity, with positivity rates as low as 8% in impending cases.[41][42] Needle aspiration of the suspected abscess serves as the gold standard for microbiologic confirmation, providing pus for Gram stain, culture, and sensitivity testing to guide antibiotic therapy.[8] This invasive procedure not only confirms the diagnosis by yielding purulent material but also offers therapeutic drainage, though it carries risks such as vascular injury if not ultrasound-guided.[40] Emerging research in 2025 has explored serological markers to aid differentiation between PTA and peritonsillar cellulitis without invasive procedures. Elevated serum levels of high-mobility group box 1 (HMGB1) and reduced kallistatin show promise as biomarkers, with receiver operating characteristic analysis indicating potential diagnostic utility in research settings, though they are not yet part of routine clinical practice.[43]Treatment
Medical management
Medical management of peritonsillar abscess primarily involves empiric antibiotic therapy targeted at the polymicrobial etiology, including group A Streptococcus and oral anaerobes, alongside supportive measures to alleviate symptoms and maintain hydration. Initial treatment typically consists of intravenous penicillin G (2-4 million units every 4-6 hours) or clindamycin (600-900 mg every 8 hours) for hospitalized patients, as these agents provide broad coverage against the predominant pathogens.[13][8] This regimen demonstrates high efficacy, with penicillin-based therapy achieving resolution rates of 95-99% when combined with drainage in most cases, though antibiotics alone can be effective in select scenarios.[4] Therapy duration is generally 10-14 days, transitioning to oral amoxicillin-clavulanate (875 mg twice daily) once the patient tolerates oral intake and clinical improvement occurs.[44] For patients with penicillin allergy, clindamycin remains the preferred alternative due to its anaerobic coverage.[13] Supportive care focuses on symptom relief and prevention of complications from odynophagia and dehydration. Analgesics such as ibuprofen (400-600 mg every 6-8 hours) or acetaminophen (500-1000 mg every 4-6 hours) are used to control pain and fever, often supplemented with nonsteroidal anti-inflammatory drugs for their anti-inflammatory effects. Corticosteroids, such as dexamethasone (8-10 mg IV) or methylprednisolone (2-3 mg/kg IV), may be administered to reduce swelling and improve symptoms.[22][45] Hydration is maintained through intravenous fluids if oral intake is impaired, and antiemetics like ondansetron (4-8 mg as needed) may be administered to manage nausea associated with severe swallowing pain.[13] These measures help stabilize patients while antibiotics take effect, typically within 24-48 hours. In cases of small abscesses, particularly those less than 2 cm in diameter with minimal symptoms, conservative management with antibiotics alone is a viable option, avoiding immediate drainage. Recent pediatric studies support this approach, reporting success rates of approximately 94% in avoiding surgical intervention within two weeks, with primary failure rates around 6% and no reported complications in selected children under 13 years.[46] This strategy is most appropriate for younger patients or those without airway compromise, emphasizing close follow-up to monitor resolution. Monitoring is essential, especially in severe cases warranting hospitalization for intravenous therapy, fluid support, and airway observation to detect potential obstruction from swelling.[13] Regimens should be adjusted for local resistance patterns; if methicillin-resistant Staphylococcus aureus (MRSA) is suspected based on risk factors or culture results, vancomycin (15-20 mg/kg every 8-12 hours) is added for coverage.[13][47] Drainage may serve as an adjunct in non-responders, but medical optimization remains the cornerstone.[44]Surgical interventions
Surgical interventions for peritonsillar abscess (PTA) primarily focus on draining the pus collection to alleviate symptoms and prevent complications, with techniques ranging from minimally invasive procedures to more definitive surgical options. Needle aspiration serves as the first-line drainage method, often feasible in an outpatient setting under local anesthesia. This procedure involves using an 18- to 20-gauge needle to puncture the most fluctuant area of the abscess, typically at the superior pole of the tonsil, allowing for both diagnosis via pus confirmation and therapeutic evacuation of contents. Studies indicate recurrence rates of 10-15% following needle aspiration, though it remains cost-effective and less invasive than alternatives.[48][44][49] Incision and drainage (I&D) offers a more thorough approach for larger or multiloculated abscesses, performed under local anesthesia by making a small incision over the fluctuant area followed by blunt dissection and evacuation. This method is considered more definitive than needle aspiration, with lower reported recurrence rates in comparative studies, and can be completed in an office or emergency setting. I&D may require repeat procedures in up to 10% of cases but provides superior pus yield and visualization of the cavity.[10][48][50] For patients with recurrent PTA or failure of initial drainage, advanced surgical options include quinsy tonsillectomy, which entails immediate bilateral tonsillectomy during the acute infection phase. This procedure is particularly indicated for recurrent cases and has demonstrated high efficacy with low complication rates in experienced hands, addressing the underlying tonsillar pathology directly. Alternatively, interval tonsillectomy is performed electively 6-8 weeks after abscess resolution, allowing inflammation to subside and reducing perioperative risks while preventing future episodes in those with a history of recurrent tonsillitis.[51][50][52] In pediatric patients, where cooperation may be challenging, conscious sedation is increasingly utilized during drainage procedures to enhance tolerability. Recent 2024 prospective studies report that conscious sedation facilitates effective PTA drainage with greater pus volume evacuated (mean 4.9 mL versus 3.2 mL with local anesthesia alone) and excellent safety profiles, including no major airway events. For smaller abscesses in children, less invasive alternatives such as ultrasound-guided aspiration under sedation may be preferred to minimize trauma. Post-procedure care involves administering culture-directed antibiotics based on aspirate analysis to target pathogens like Streptococcus pyogenes, alongside close follow-up to monitor for resolution of trismus, dysphagia, and fever. Potential complications include bleeding, occurring in approximately 1-2% of drainage cases, though rates rise to 11-13% following tonsillectomy; most are managed conservatively or with hemostasis.[44][53][54]Epidemiology and prognosis
Epidemiology
Peritonsillar abscess (PTA) has an annual incidence of approximately 30 cases per 100,000 persons in developed countries, primarily affecting individuals aged 5 to 49 years.[55] In the United States, this translates to an estimated 45,000 new cases each year.[1] The condition is most prevalent among adolescents and young adults, with rates reaching up to 40 cases per 100,000 person-years in those aged 15 to 19, compared to 30 to 40 cases per 100,000 in adults overall.[56] Males show a slight predominance.[1] In pediatric populations, incidence remains stable at 20 to 30 cases per 100,000 annually, though it is rarer in children under 5 years.[57] Historical trends indicate a relatively stable incidence in developed countries around 30 per 100,000.[1] However, post-COVID-19 data from 2024 and 2025 reveal shifts, including a temporary decrease during nonpharmaceutical interventions (14.9 cases per 100,000) followed by a rebound to 26.9 cases per 100,000 after restrictions lifted, potentially due to delayed care and accumulated infections.[58] Incidence exhibits seasonal peaks in winter and spring, correlating with higher rates of respiratory infections.[59] Geographic variations show higher PTA rates in rural and low-resource settings, where access to timely antibiotic treatment for tonsillitis is limited.[60] Studies indicate that over 50% of non-streptococcal pathogens in PTA cultures show penicillin resistance, complicating management.[13]Prognosis
With appropriate treatment, peritonsillar abscess (PTA) typically resolves rapidly, with 90-95% of cases showing significant symptom improvement within 24-48 hours.[22] Hospitalization duration typically ranges from 2 to 4 days, though it can extend to 5-7 days in complicated cases.[13] The risk of recurrence following a single episode is estimated at 5-15%, but rises to 20-30% in patients with prior recurrent tonsillitis or multiple abscess episodes.[61][62] Recent 2025 data indicate that conservative antibiotic management in pediatric cases achieves success in approximately 87% of patients, thereby reducing the immediate need for surgical intervention.[63] Prognosis is influenced by several factors, including timely diagnosis; delays can increase the risk of complications such as airway obstruction.[8] Outcomes are generally more favorable in immunocompetent individuals without underlying comorbidities.[8] Long-term sequelae are rare, with tonsillectomy reducing the risk of future PTA by 80-90% in high-risk patients.[64] Overall mortality is less than 0.1% with modern care, though it can rise significantly if treatment is delayed leading to severe complications.[8][25]History and etymology
Historical background
The peritonsillar abscess, historically known as quinsy, was first recognized in ancient medical texts as a severe suppurative infection of the throat. Hippocrates, around 400 BCE, described quinsy as a painful swelling of the tonsils accompanied by fever and difficulty swallowing, recommending incision and drainage as a therapeutic approach when suppuration occurred.[65] In the Roman era, Aulus Cornelius Celsus, in the first century CE, further detailed the condition in his work De Medicina, advocating for surgical incision to relieve the abscess and prevent complications such as airway obstruction, marking an early milestone in its surgical management.[66] By the 18th century, anatomical understanding advanced, with Italian physician Giovanni Battista Morgagni contributing to the pathological description of deep neck infections, including those akin to peritonsillar abscesses, through his systematic postmortem studies in De Sedibus et Causis Morborum per Anatomem Indagatis.[67] In the 19th century, management relied primarily on incision and drainage, with less invasive needle aspiration techniques developed in the mid-20th century and gaining widespread adoption in the 1980s for outpatient treatment.[8][68] The 20th century brought transformative changes with the advent of antibiotics in the 1940s, particularly penicillin, which dramatically reduced the incidence and mortality of streptococcal infections, including peritonsillar abscesses, by targeting group A Streptococcus pyogenes, the primary pathogen.[69] Pre-antibiotic era mortality rates for complicated peritonsillar and deep neck infections could reach 25%, dropping to less than 1% post-World War II due to penicillin and improved care, leading to a significant decline in severe suppurative complications.[70][71] From the 1980s onward, a shift toward conservative management emerged, emphasizing outpatient needle aspiration combined with oral antibiotics over immediate surgical intervention, supported by studies demonstrating high success rates and reduced hospitalization needs. In the mid-20th century, initial studies explored outpatient approaches; by the 1980s, Herzon's work demonstrated high success with needle aspiration and antibiotics, further promoting this shift.[68][72] In the modern era, imaging-guided techniques have gained prominence, with recent studies from 2024 and 2025 highlighting the use of ultrasound for diagnosis and, in select cases, guided drainage to aid precise localization and aspiration while minimizing risks like vascular injury in emergency settings.[73][74] These advancements reflect ongoing refinements in imaging and procedural safety, building on the foundational surgical principles established centuries earlier.Etymology
The term "peritonsillar abscess" combines the Greek prefix peri-, meaning "around" or "about," with tonsillar, pertaining to the tonsils, and abscess, denoting a localized collection of pus. The word tonsil derives from Latin tonsillae (plural), a diminutive form possibly related to tōlēs, referring to a swollen gland or goiter, evoking the almond-like shape of these lymphoid structures in the throat.[75] Abscess originates from Latin abscessus, the past participle of abscēdere ("to go away" or "separate"), which in ancient medical theory described the "departure" of harmful humors as pus.[76] Historically, the condition has been known by the synonym quinsy, a term prevalent in English medical and lay usage until the early 20th century. Quinsy stems from Middle English quinesye, borrowed from Old French quinencie and Medieval Latin quinancia, tracing back to Ancient Greek kynanchē—a compound of kyn- (from kyōn, "dog") and anchein ("to strangle" or "choke")—capturing the sensation of throat constriction akin to a dog's collar.[77][78] The precise nomenclature "peritonsillar abscess" gained standardization in 19th-century medical texts, with the adjective peritonsillar entering English usage in the 1870s to accurately denote the suppurative collection in the space surrounding the tonsillar capsule.[79] Earlier or alternative designations, such as "paratonsillar abscess," appeared occasionally but were deemed anatomically imprecise, as the infection develops in the peritonsillar space rather than simply adjacent to the tonsil. In other languages, equivalent terms mirror this anatomical focus; for instance, the French "abcès périamygdalien" employs péri- (around) and amygdalien (from Latin amygdala, "almond," highlighting the tonsil's shape).[80]Notable cases
Historical cases
Pope Adrian IV (c. 1100–1159), the only English pope, reportedly died from quinsy, a peritonsillar abscess, though some accounts suggest he choked on a fly in his wine.[81] French Renaissance philosopher Michel de Montaigne (1533–1592) died at age 59 from quinsy, which caused throat inflammation and deprived him of speech in his final days.[82]Modern cases
In 2017, English singer and actor Lee Ryan of the band Blue was hospitalized for quinsy as a complication of tonsillitis, requiring treatment and rest.[83] Reality television personality Bianca Gascoigne was hospitalized for quinsy in 2016 and again in 2018 in Dubai, describing severe throat pain and swelling.[84] Actress Crissy Rock was rushed to hospital in 2019 for a throat abscess diagnosed as quinsy, undergoing drainage due to intense pain.[85]References
- https://en.wiktionary.org/wiki/quinsy

