Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Enterococcus faecalis AI simulator
(@Enterococcus faecalis_simulator)
Hub AI
Enterococcus faecalis AI simulator
(@Enterococcus faecalis_simulator)
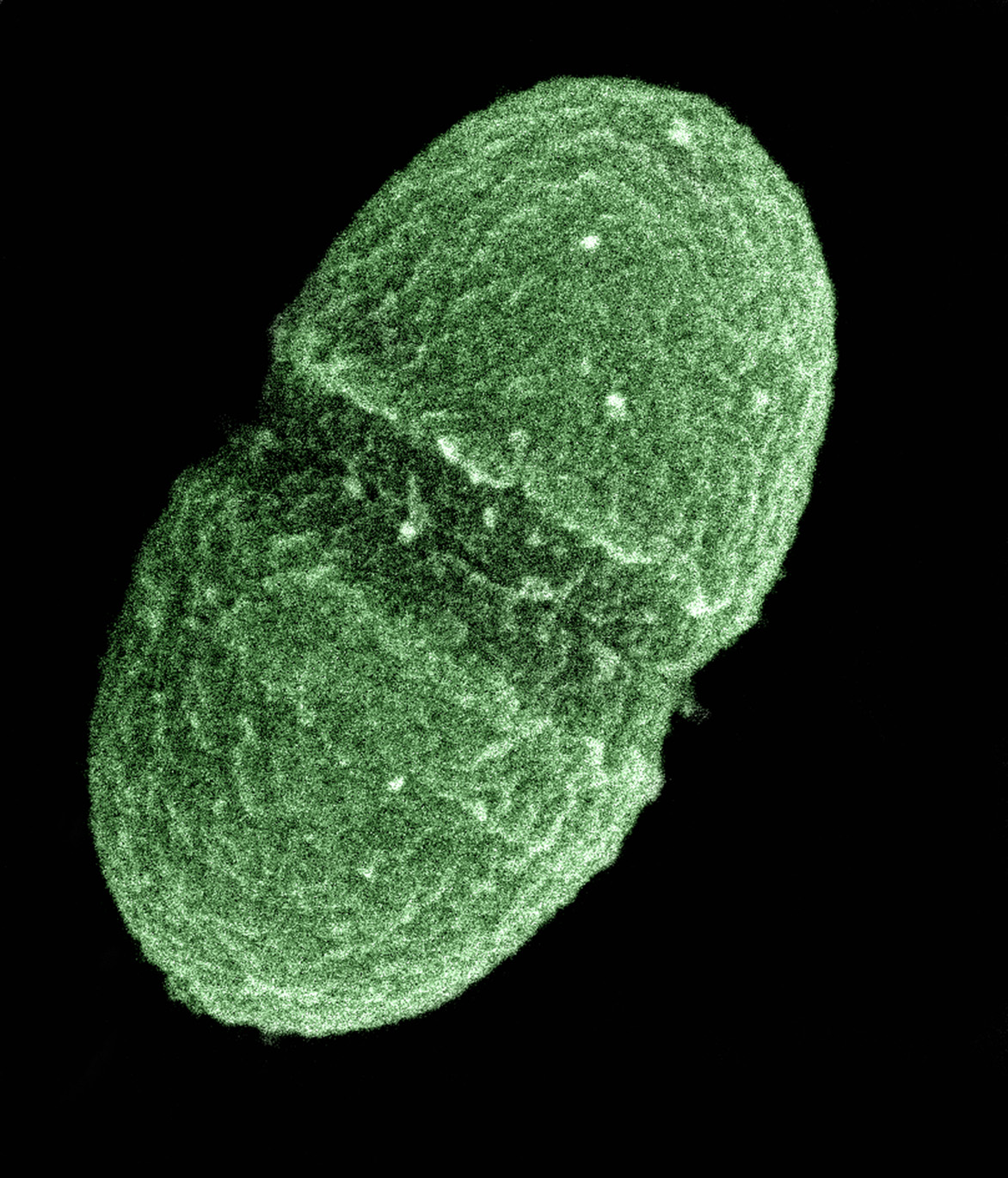
Enterococcus faecalis
Enterococcus faecalis – formerly classified as part of the group D Streptococcus, is a Gram-positive, commensal bacterium naturally inhabiting the gastrointestinal tracts of humans. Like other species in the genus Enterococcus, E. faecalis is found in healthy humans and can be used as a probiotic. The probiotic strains such as Symbioflor1 and EF-2001 are characterized by the lack of specific genes related to drug resistance and pathogenesis.
Despite its commensal role, E. faecalis is an opportunistic pathogen capable of causing severe infections, especially in the nosocomial (hospital) settings. Enterococcus spp. is among the leading causes of healthcare-associated infections ranging from endocarditis to urinary tract infections (UTIs). Hospital-acquired UTIs are associated with catheterization because catheters provide an ideal surface for biofilm formation, allowing E. faecalis to adhere, persist, and evade both the immune response and antibiotic treatment.
E. faecalis is able to grow in extreme environments due to its highly adaptive genome and lack of strong defense mechanisms. Its ability to easily acquire and transfer genes across species contributes to rising antibiotic resistance. E. faecalis exhibits intrinsic resistance to multiple antibiotics, including oxazolidinones, quinolones, and most β -lactams, such as cephalosporins.
E. faecalis has been frequently found in reinfected, root canal-treated teeth in prevalence values ranging from 30% to 90% of the cases. Re-infected root canal-treated teeth are about nine times more likely to harbor E. faecalis than cases of primary infections.
E. faecalis is a nonmotile microbe; it ferments glucose without gas production, and does not produce a catalase reaction with hydrogen peroxide. It produces a reduction of litmus milk, but does not liquefy gelatin. It shows consistent growth throughout nutrient broth which is consistent with being a facultative anaerobe. It catabolizes a variety of energy sources, including glycerol, lactate, malate, citrate, arginine, agmatine, and many keto acids. Enterococci survive very harsh environments, including extremely alkaline pH (9.6) and salt concentrations. They resist bile salts, detergents, heavy metals, ethanol, azide, and desiccation. They can grow in the range of 10 to 45 °C and survive at temperatures of 60 °C for 30 min.
In clinical settings, E. faecalis displays a relatively conserved metabolic profile compared to other enterococcal species. A recent large-scale study of urinary isolates from ICU patients showed that E. faecalis consistently metabolizes sorbitol, mannitol, amygdalin and sucrose but lacks the ability to utilize L-arabinose, melibiose, or raffinose—substrates readily used by E. faecium and E. durans. This substrate profile provides a reliable metabolic signature that can help distinguish E. faecalis from related species in diagnostic and research contexts.
E. faecalis is found in most healthy individuals, but can cause endocarditis and sepsis, urinary tract infections (UTIs), meningitis, and other infections in humans. Several virulence factors are thought to contribute to E. faecalis infections. A plasmid-encoded hemolysin, called the cytolysin, is important for pathogenesis in animal models of infection, and the cytolysin in combination with high-level gentamicin resistance is associated with a five-fold increase in risk of death in human bacteremia patients. A plasmid-encoded adhesin called "aggregation substance" is also important for virulence in animal models of infection.
E. faecalis contains a tyrosine decarboxylase enzyme capable of decarboxylating L-DOPA, a crucial drug in the treatment of Parkinson's disease. If L-DOPA is decarboxylated in the gut microbiome, it cannot pass through the blood-brain barrier and be decarboxylated in the brain to become dopamine.
Enterococcus faecalis
Enterococcus faecalis – formerly classified as part of the group D Streptococcus, is a Gram-positive, commensal bacterium naturally inhabiting the gastrointestinal tracts of humans. Like other species in the genus Enterococcus, E. faecalis is found in healthy humans and can be used as a probiotic. The probiotic strains such as Symbioflor1 and EF-2001 are characterized by the lack of specific genes related to drug resistance and pathogenesis.
Despite its commensal role, E. faecalis is an opportunistic pathogen capable of causing severe infections, especially in the nosocomial (hospital) settings. Enterococcus spp. is among the leading causes of healthcare-associated infections ranging from endocarditis to urinary tract infections (UTIs). Hospital-acquired UTIs are associated with catheterization because catheters provide an ideal surface for biofilm formation, allowing E. faecalis to adhere, persist, and evade both the immune response and antibiotic treatment.
E. faecalis is able to grow in extreme environments due to its highly adaptive genome and lack of strong defense mechanisms. Its ability to easily acquire and transfer genes across species contributes to rising antibiotic resistance. E. faecalis exhibits intrinsic resistance to multiple antibiotics, including oxazolidinones, quinolones, and most β -lactams, such as cephalosporins.
E. faecalis has been frequently found in reinfected, root canal-treated teeth in prevalence values ranging from 30% to 90% of the cases. Re-infected root canal-treated teeth are about nine times more likely to harbor E. faecalis than cases of primary infections.
E. faecalis is a nonmotile microbe; it ferments glucose without gas production, and does not produce a catalase reaction with hydrogen peroxide. It produces a reduction of litmus milk, but does not liquefy gelatin. It shows consistent growth throughout nutrient broth which is consistent with being a facultative anaerobe. It catabolizes a variety of energy sources, including glycerol, lactate, malate, citrate, arginine, agmatine, and many keto acids. Enterococci survive very harsh environments, including extremely alkaline pH (9.6) and salt concentrations. They resist bile salts, detergents, heavy metals, ethanol, azide, and desiccation. They can grow in the range of 10 to 45 °C and survive at temperatures of 60 °C for 30 min.
In clinical settings, E. faecalis displays a relatively conserved metabolic profile compared to other enterococcal species. A recent large-scale study of urinary isolates from ICU patients showed that E. faecalis consistently metabolizes sorbitol, mannitol, amygdalin and sucrose but lacks the ability to utilize L-arabinose, melibiose, or raffinose—substrates readily used by E. faecium and E. durans. This substrate profile provides a reliable metabolic signature that can help distinguish E. faecalis from related species in diagnostic and research contexts.
E. faecalis is found in most healthy individuals, but can cause endocarditis and sepsis, urinary tract infections (UTIs), meningitis, and other infections in humans. Several virulence factors are thought to contribute to E. faecalis infections. A plasmid-encoded hemolysin, called the cytolysin, is important for pathogenesis in animal models of infection, and the cytolysin in combination with high-level gentamicin resistance is associated with a five-fold increase in risk of death in human bacteremia patients. A plasmid-encoded adhesin called "aggregation substance" is also important for virulence in animal models of infection.
E. faecalis contains a tyrosine decarboxylase enzyme capable of decarboxylating L-DOPA, a crucial drug in the treatment of Parkinson's disease. If L-DOPA is decarboxylated in the gut microbiome, it cannot pass through the blood-brain barrier and be decarboxylated in the brain to become dopamine.