Recent from talks
Nothing was collected or created yet.
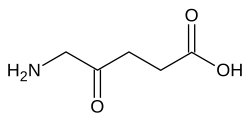
Aminolevulinic acid
View on Wikipedia
 | |
| Clinical data | |
|---|---|
| Trade names | Levulan, NatuALA, Ameluz, others |
| Other names | 5-aminolevulinic acid |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607062 |
| License data | |
| Routes of administration | Topical, By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.003.105 |
| Chemical and physical data | |
| Formula | C5H9NO3 |
| Molar mass | 131.131 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 118 °C (244 °F) |
| |
| |
| (verify) | |
δ-Aminolevulinic acid (also dALA, δ-ALA, 5ALA, 5-ALA or 5-aminolevulinic acid), an endogenous non-proteinogenic amino acid, is the first compound in the porphyrin synthesis pathway, the pathway that leads to heme[3] in mammals, as well as chlorophyll[4] in plants.
5-ALA is used in photodynamic detection and surgery of cancer.[5][6][7][8]
Medical uses
[edit]As a precursor of a photosensitizer, 5-ALA is also used as an add-on agent for photodynamic therapy.[9] In contrast to larger photosensitizer molecules, it is predicted by computer simulations to be able to penetrate tumor cell membranes.[10]
Cancer diagnosis
[edit]Photodynamic detection is the use of photosensitive drugs with a light source of the right wavelength for the detection of cancer, using fluorescence of the drug.[5] 5-ALA, or derivatives thereof, can be used to visualize bladder cancer by fluorescence imaging.[5]
Cancer treatment
[edit]Aminolevulinic acid is being studied for photodynamic therapy (PDT) in a number of types of cancer.[11] It is not currently a first line treatment for Barrett's esophagus.[12] Its use in brain cancer is currently experimental.[13] It has been studied in a number of gynecological cancers.[14]
Intra-operative cancer delineation
[edit]Aminolevulinic acid utilization is promising in the field of cancer delineation, particularly in the context of fluorescence-guided surgery. This compound is utilized to enhance the visualization of malignant tissues during surgical procedures.[7][8] The US FDA approved aminolevulinic acid hydrochloride (ALA HCL) for this use in 2017.[15]
When administered to patients, 5-ALA is metabolized to protoporphyrin IX (PpIX) preferentially in cancer cells, leading to their fluorescence under specific light wavelengths.[16] This fluorescence aids surgeons in real-time identification and precise removal of cancerous tissue, reducing the likelihood of leaving residual tumor cells behind. This innovative approach has shown success in various cancer types, including brain and spine gliomas, bladder cancer, and oral squamous cell carcinoma.[17][18][19]
5-ALA in gliomas
[edit]Aminolevulinic acid is indicated in adults for visualization of malignant tissue during surgery for malignant glioma (World Health Organization grade III and IV).[20]
Studies since 2006 have shown that the intraoperative use of this guiding method may reduce the tumour residual volume and prolong progression-free survival in people with malignant gliomas.[7][8]
Cytoreductive surgery has been considered to be benficial for patients with high-grade-gliomas[21]; it has resulted in significantly higher rate of complete resections in malignant gliomas, compared to the traditional white-light resections. The use of 5-ALA has been described as an essential technique, and as standard-of-care at many neurosurgical departments worldwide.[22]
Side effects
[edit]Side effects of administration may include liver damage and nerve problems.[12] Hyperthermia may also occur.[13] Deaths have also resulted.[12]
Biosynthesis
[edit]In non-photosynthetic eukaryotes such as animals, fungi, and protozoa, as well as the class Alphaproteobacteria of bacteria, it is produced by the enzyme ALA synthase, from glycine and succinyl-CoA. This reaction is known as the Shemin pathway, which occurs in mitochondria.[23]
In plants, algae, bacteria (except for the class Alphaproteobacteria) and archaea, it is produced from glutamic acid via glutamyl-tRNA and glutamate-1-semialdehyde. The enzymes involved in this pathway are glutamyl-tRNA synthetase, glutamyl-tRNA reductase, and glutamate-1-semialdehyde 2,1-aminomutase. This pathway is known as the C5 or Beale pathway.[24][25] In most plastid-containing species, glutamyl-tRNA is encoded by a plastid gene, and the transcription, as well as the following steps of C5 pathway, take place in plastids.[26]
Importance in humans
[edit]Activation of mitochondria
[edit]In humans, 5ALA is a precursor to heme.[3] Biosynthesized 5ALA goes through a series of transformations in the cytosol, finally being converted to protoporphyrin IX inside the mitochondria.[27][28] This protoporphyrin molecule chelates with iron in presence of enzyme ferrochelatase to produce heme.[27][28]
Heme increases the mitochondrial activity thereby helping in activation of respiratory system Krebs cycle and electron transport chain[29] leading to formation of adenosine triphosphate (ATP) for adequate supply of energy to the body.[29]
Accumulation of protoporphyrin IX
[edit]Cancer cells lack or have reduced ferrochelatase activity and this results in accumulation of protoporphyrin IX, a fluorescent substance that can easily be visualized.[5]
Induction of heme oxygenase-1 (HO-1)
[edit]Excess heme is converted in macrophages to biliverdin and ferrous ions by the enzyme HO-1. Biliverdin formed can be further converted to bilirubin and carbon monoxide.[30] Biliverdin and bilirubin are potent antioxidants and regulate important biological processes like inflammation, apoptosis, cell proliferation, fibrosis and angiogenesis.[30]
Plants
[edit]In plants, production of 5-ALA is the step on which the speed of synthesis of chlorophyll is regulated.[4] Plants that are fed by external 5-ALA accumulate toxic amounts of chlorophyll precursor, protochlorophyllide, indicating that the synthesis of this intermediate is not suppressed anywhere downwards in the chain of reaction. Protochlorophyllide is a strong photosensitizer in plants.[31] Controlled spraying of 5-ALA at lower doses (up to 150 mg/L) can however help protect plants from stress and encourage growth.[32]
References
[edit]- ^ "Levulan Kerastick Product information". Health Canada. 25 April 2012. Retrieved 4 June 2022.
- ^ "Gleolan Product information". Health Canada. 25 April 2012. Retrieved 4 June 2022.
- ^ a b Gardener LC, Cox TM (1988). "Biosynthesis of heme in immature erythroid cells". The Journal of Biological Chemistry. 263: 6676–6682. doi:10.1016/S0021-9258(18)68695-8.
- ^ a b Von Wettstein D, Gough S, Kannangara CG (July 1995). "Chlorophyll Biosynthesis". The Plant Cell. 7 (7): 1039–1057. doi:10.1105/tpc.7.7.1039. PMC 160907. PMID 12242396.
- ^ a b c d Wagnières, G.., Jichlinski, P., Lange, N., Kucera, P., Van den Bergh, H. (2014). Detection of Bladder Cancer by Fluorescence Cystoscopy: From Bench to Bedside - the Hexvix Story. Handbook of Photomedicine, 411-426.
- ^ Eyüpoglu IY, Buchfelder M, Savaskan NE (March 2013). "Surgical resection of malignant gliomas-role in optimizing patient outcome". Nature Reviews. Neurology. 9 (3): 141–151. doi:10.1038/nrneurol.2012.279. PMID 23358480. S2CID 20352840.
- ^ a b c Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ (May 2006). "Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial". The Lancet. Oncology. 7 (5): 392–401. doi:10.1016/s1470-2045(06)70665-9. PMID 16648043.
- ^ a b c Eyüpoglu IY, Hore N, Savaskan NE, Grummich P, Roessler K, Buchfelder M, et al. (2012). "Improving the extent of malignant glioma resection by dual intraoperative visualization approach". PLOS ONE. 7 (9) e44885. Bibcode:2012PLoSO...744885E. doi:10.1371/journal.pone.0044885. PMC 3458892. PMID 23049761.
- ^ Yew YW, Lai YC, Lim YL, Chong WS, Theng C (June 2016). "Photodynamic Therapy With Topical 5% 5-Aminolevulinic Acid for the Treatment of Truncal Acne in Asian Patients". Journal of Drugs in Dermatology. 15 (6): 727–732. PMID 27272080.
- ^ Erdtman E (2008). "Modelling the behavior of 5-aminolevulinic acid and its alkyl esters in a lipid bilayer". Chemical Physics Letters. 463 (1–3): 178–182. Bibcode:2008CPL...463..178E. doi:10.1016/j.cplett.2008.08.021.
- ^ Inoue K (February 2017). "5-Aminolevulinic acid-mediated photodynamic therapy for bladder cancer". International Journal of Urology. 24 (2): 97–101. doi:10.1111/iju.13291. PMID 28191719.
- ^ a b c Qumseya BJ, David W, Wolfsen HC (January 2013). "Photodynamic Therapy for Barrett's Esophagus and Esophageal Carcinoma". Clinical Endoscopy. 46 (1): 30–37. doi:10.5946/ce.2013.46.1.30. PMC 3572348. PMID 23423151.
- ^ a b Tetard MC, Vermandel M, Mordon S, Lejeune JP, Reyns N (September 2014). "Experimental use of photodynamic therapy in high grade gliomas: a review focused on 5-aminolevulinic acid". Photodiagnosis and Photodynamic Therapy. 11 (3): 319–330. doi:10.1016/j.pdpdt.2014.04.004. PMID 24905843. S2CID 38534019.
- ^ Shishkova N, Kuznetsova O, Berezov T (March 2012). "Photodynamic therapy for gynecological diseases and breast cancer". Cancer Biology & Medicine. 9 (1): 9–17. doi:10.3969/j.issn.2095-3941.2012.01.002. PMC 3643637. PMID 23691448.
- ^ FDA Approves Fluorescing Agent for Glioma Surgery.June 2017
- ^ Hadjipanayis CG, Widhalm G, Stummer W (November 2015). "What is the Surgical Benefit of Utilizing 5-Aminolevulinic Acid for Fluorescence-Guided Surgery of Malignant Gliomas?". Neurosurgery. 77 (5): 663–673. doi:10.1227/NEU.0000000000000929. PMC 4615466. PMID 26308630.
- ^ Maragkos GA, Schüpper AJ, Lakomkin N, Sideras P, Price G, Baron R, et al. (2021). "Fluorescence-Guided High-Grade Glioma Surgery More Than Four Hours After 5-Aminolevulinic Acid Administration". Frontiers in Neurology. 12 644804. doi:10.3389/fneur.2021.644804. PMC 7985355. PMID 33767664.
- ^ Albalkhi I, Shafqat A, Bin-Alamer O, Abou Al-Shaar AR, Mallela AN, Fernández-de Thomas RJ, et al. (December 2023). "Fluorescence-guided resection of intradural spinal tumors: a systematic review and meta-analysis". Neurosurgical Review. 47 (1) 10. doi:10.1007/s10143-023-02230-x. PMID 38085385. S2CID 266164983.
- ^ Filip P, Lerner DK, Kominsky E, Schupper A, Liu K, Khan NM, et al. (February 2024). "5-Aminolevulinic Acid Fluorescence-Guided Surgery in Head and Neck Squamous Cell Carcinoma". The Laryngoscope. 134 (2): 741–748. doi:10.1002/lary.30910. PMID 37540051. S2CID 260485667.
- ^ "Gliolan EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 6 January 2021.
- ^ Ewelt C, Nemes A, Senner V, Wölfer J, Brokinkel B, Stummer W, et al. (July 2015). "Fluorescence in neurosurgery: Its diagnostic and therapeutic use. Review of the literature". Journal of Photochemistry and Photobiology. B, Biology. 148: 302–309. Bibcode:2015JPPB..148..302E. doi:10.1016/j.jphotobiol.2015.05.002. PMID 26000742.
- ^ Hadjipanayis CG, Widhalm G, Stummer W (November 2015). "What is the Surgical Benefit of Utilizing 5-Aminolevulinic Acid for Fluorescence-Guided Surgery of Malignant Gliomas?". Neurosurgery. 77 (5): 663–673. doi:10.1227/NEU.0000000000000929. PMC 4615466. PMID 26308630.
- ^ Ajioka J, Soldati D, eds. (2007). Toxoplasma: molecular and cellular biology. Norfolk, England: Horizon Bioscience. p. 415. ISBN 978-1-904933-34-2.
- ^ Beale SI (August 1990). "Biosynthesis of the Tetrapyrrole Pigment Precursor, delta-Aminolevulinic Acid, from Glutamate". Plant Physiology. 93 (4): 1273–1279. doi:10.1104/pp.93.4.1273. PMC 1062668. PMID 16667613.
- ^ Willows RD (2004). "Chlorophylls". In Goodman RM (ed.). Encyclopaedia of Plant and Crop Science. Marcel Dekker. pp. 258–262. ISBN 0-8247-4268-0.
- ^ Biswal B, Krupinska K, Biswal UC, eds. (July 2013). Plastid development in leaves during growth and senescence. Dordrecht, The Netherlands: Springer. ISBN 9789400757233.
- ^ a b Malik Z, Djaldetti M (June 1979). "5-Aminolevulinic acid stimulation of porphyrin and hemoglobin synthesis by uninduced Friend erythroleukemic cells". Cell Differentiation. 8 (3): 223–233. doi:10.1016/0045-6039(79)90049-6. PMID 288514.
- ^ a b Olivo M, Bhuvaneswari R, Keogh I (July 2011). "Advances in bio-optical imaging for the diagnosis of early oral cancer". Pharmaceutics. 3 (3): 354–378. doi:10.3390/pharmaceutics3030354. PMC 3857071. PMID 24310585.
- ^ a b Ogura S, Maruyama K, Hagiya Y, Sugiyama Y, Tsuchiya K, Takahashi K, et al. (March 2011). "The effect of 5-aminolevulinic acid on cytochrome c oxidase activity in mouse liver". BMC Research Notes. 4 (4) 66. doi:10.1186/1756-0500-4-66. PMC 3068109. PMID 21414200.
- ^ a b Loboda A, Damulewicz M, Pyza E, Jozkowicz A, Dulak J (September 2016). "Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism". Cellular and Molecular Life Sciences. 73 (17): 3221–3247. doi:10.1007/s00018-016-2223-0. PMC 4967105. PMID 27100828.
- ^ Kotzabasis K, Senger H (1990). "The influence of 5-aminolevulinic acid on protochlorophyllide and protochlorophyll accumulation in dark-grown Scenedesmus". Z. Naturforsch. 45 (1–2): 71–73. doi:10.1515/znc-1990-1-212. S2CID 42965243.
- ^ Kosar F, Akram NA, Ashraf M (January 2015). "Exogenously-applied 5-aminolevulinic acid modulates some key physiological characteristics and antioxidative defense system in spring wheat (Triticum aestivum L.) seedlings under water stress". South African Journal of Botany. 96: 71–77. Bibcode:2015SAJB...96...71K. doi:10.1016/j.sajb.2014.10.015.

