Recent from talks
Nothing was collected or created yet.
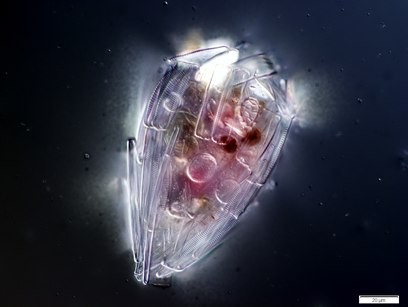
Testate amoebae
View on Wikipedia
Testate amoebae (formerly thecamoebians, Testacea or Thecamoeba) are a polyphyletic group of unicellular amoeboid protists, which differ from naked amoebae in the presence of a test that partially encloses the cell, with an aperture from which the pseudopodia emerge, that provides the amoeba with shelter from predators and environmental conditions.
The test of some species is produced entirely by the amoeba and may be organic, siliceous or calcareous depending on the species (autogenic tests), whereas in other cases the test is made up of particles of sediment collected by the amoeba which are then agglutinated together by secretions from within the cell (xenogenic tests). A few taxa (Hyalospheniidae) can build either type, depending on the circumstances and availability of foreign material.[1]
The assemblage referred to as "testate amoebae" is actually composed of several, unrelated groups of organisms. However, some features they all share that have been used to group them together include the presence of a test (regardless of its composition) and pseudopodia that do not anastomose.[2]
Testate amoebae can be found in most freshwater environments, including lakes, rivers, cenotes,[3] as well as mires and soils.
The strong and resistant nature of the tests allows them to be preserved long after the amoeba has died. These characteristics, along with the sensitivity that some species display to changes in environmental conditions (such as temperature, pH, and conductivity), has sparked their use as bioindicators and paleoclimate proxies in recent years.[4]
Gallery
[edit]| Part of a series related to |
| Biomineralization |
|---|
 |
-
Naked amoeba (i. e. not testate) showing food vacuoles and ingested diatom
-
Shell or test of a testate amoeba, Arcella sp.
-
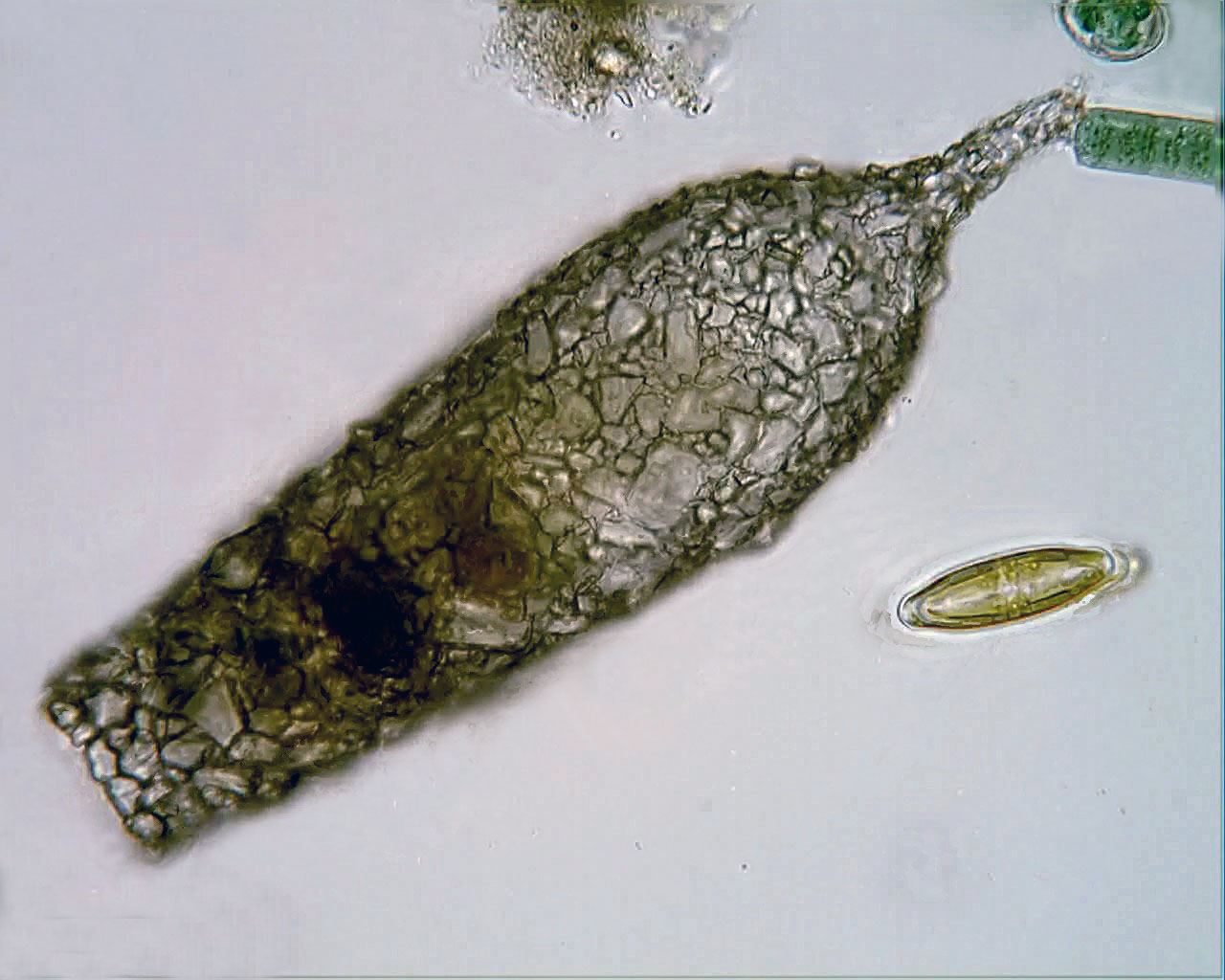
Xenogenic testate amoeba covered in diatoms (from Penard's Amoeba Collection)
-
Euglypha tuberculata, a species with a siliceous autogenic test
-
The autogenic test of Arcella discoides, made up of organic plates
-
Shell of Cyphoderia ampulla, composed of circular, siliceous plates produced by the amoeba
Taxonomy and classification
[edit]Testate amoebae are a polyphyletic assemblage. The main testate amoebae groups are the lobose Tubulinea, which include Arcellinida, Difflugina and Phryganellina (within the Amoebozoa),[5] and the filose Euglyphida (within the SAR supergroup),[6] although there are smaller groups that also include other testate amoebae.[7]
Order Arcellinida
[edit]- Family Arcellidae
- Arcella - Ehrenberg 1832
- Antarcella - Deflandre 1928
- Family Netzeliidae
- Netzelia - Ogden 1979
- Family Hyalospheniidae
- Quadrulella - Cockerell 1909
- Hyalosphenia - Stein 1859
- Alocodera - Jung 1942
- Apodera - Loeblich & Tappan 1961
- Certesella - Loeblich & Tappan 1961
- Porosia - Jung 1942
- Nebela - Leidy 1874
- Padaungiella - Lara & Todorov 2012
- Family Microchlamyiidae
- Microchlamys - Cockerell 1911
- Spumochlamys - Kudryavtsev & Hausmann 2007
- Family Plagiopyxidae
- Bullinularia - Deflandre 1953
- Geoplagiopyxis - Chardez 1961
- Protoplagiopyxis - Bonnet 1962
- Paracentropyxis - Bonnet 1960
- Plagiopyxis - Penard 1910
- Hoogenraadia - Gauthier-Lievre & Thomas 1958
- Planhoogenraadia - Bonnet 1977
- Family Cryptodifflugiidae
- Cryptodifflugia - Penard 1890
- Wailesella - Deflandre 1928
- Family Microcoryciidae
- Amphizonella - Greeff 1866
- Diplochlamys - Greeff 1888
- Microcorycia - Cockerell 1911
- Penardochlamys - Deflandre 1953
- Zonomyxa - Nusslin 1882
- Parmulina - Penard 1902
- Family Phryganellidae
- Phryganella - Penard 1902
- Family Lamtopyxidae
- Lamtopyxis - Bonnet 1974
- Family Distomatopyxidae
- Distomatopyxis - Bonnet 1964
- Family Paraquadrulidae
- Paraquadrula - Deflandre 1932
- Lamtoquadrula - Bonnet 1974
- Family Centropyxidae
- Centropyxis - Stein 1857
- Proplagiopyxis - Schonborn 1964
- Family Trigonopyxidae
- Trigonopyxis - Penard 1912
- Cyclopyxis - Deflandre 1929
- Geopyxella - Bonnet & Thomas 1955
- Cornuapyxis - Couteaux and Chardez 1981
- Incertae sedis
- Argynnia - Vucetich 1974
- Awerintzewia - Schouteden 1906
- Cucurbitella - Penard 1902
- Difflugia - Leclerc 1815
- Geamphorella - Bonnet 1959
- Heleopera - Leidy 1879
- Jungia - Loeblich and Tappan 1961
- Lagenodifflugia - Medioli & Scott 1983
- Leptochlamys - West 1901
- Lesquereusia - Schlumberger 1845
- Maghrebia - Gauthier-Lievre & Thomas 1960
- Mediolus - Patterson 2014
- Microquadrula - Golemansky 1968
- Oopyxis - Jung 1942
- Pentagonia - Gauthier-Lievre & Thomas 1960
- Physochila - Jung 1942
- Pomoriella - Golemansky 1970
- Pontigulasia - Rhumbler 1896
- Protocucurbitella - Gauthier-Lievre & Thomas 1960
- Pseudawerintzewia - Bonnet 1959
- Pseudonebela - Gauthier-Lievre 1953
- Pyxidicula - Ehrenberg 1838
- Schoenbornia - Decloitre 1964
- Schwabia - Jung 1942
- Sexangularia - Awerintzew 1906
- Suiadifflugia - Green 1975
- Zivkovicia - Ogden 1987
- Ellipsopyxis - Bonnet 1965
- Ellipsopyxella - Bonnet 1975
Order Euglyphida
[edit]
|
|
|
Other Cercozoa
[edit]
|
|
|
Order Stramenopila
[edit]
|
|
Unclassified testate amoebae
[edit]- Paramphitrema - Valkanov 1970
The following table includes a few examples of testate amoebae genera, and reflects their position within the classification by Adl et al. (2012),[7] where five supergroups (Amoebozoa, Opisthokonta, Excavata, SAR and Archaeplastida) were proposed to classify all eukaryotes. This classification purposefully avoids the use of Linnaean higher category names (phylum, class, order, family). While it has been noted that the names that Adl et al. provide for the clades may result confusing or uninformative regarding the relative degree of phenotypic distinctiveness amongst groups when used in isolation,[8] this system avoids creating superfluous ranks where unnecessary and provides stable group names that can be retained even when a group is moved to a different lineage, as is often the case with protists, as their classification remains in constant review.[7]
| Amoebozoa | Tubulinea | Arcellinida | Arcellina | Amphizonella - Arcella - Microchlamys - Microcorycia - Spumochlamys | |||
|---|---|---|---|---|---|---|---|
| Difflugina | Bullinularia - Centropyxis - Difflugia - Distomatopyxis - Heleopera - Hyalosphenia - Lesquereusia - Nebela - Paraquadrula - Pontigulasia -
Plagiopyxis - Quadrulella - Trigonopyxis | ||||||
| Phryganellina | Cryptodifflugia - Phryganella - Wailesella | ||||||
| Discosea | Himatismenida | Cochliopodium | |||||
| SAR Supergroup | Stramenopila | Labyrinthulomycetes | Amphitremida | Amphitrema - Archerella | |||
| Rhizaria | Cercozoa | Thecofilosea | Cryomonadida | Rhizaspididae | Capsellina - Rhizaspis - Rhogostoma | ||
| Ventricleftida | Ventrifissura - Verrucomonas | ||||||
| Imbricatea | Silicofilosea | Euglyphida | Euglyphidae | Euglypha - Scutiglypha | |||
| Assulinidae | Assulina - Placocista - Valkanovia | ||||||
| Trinematidae | Corythion - Playfairina - Puytoracia - Trinema | ||||||
| Cyphoderidae | Campascus - Corythionella - Cyphoderia - Messemvriella - Pseudocorythion - Schaudinnula. | ||||||
| Paulinellidae | Ovulinata - Paulinella | ||||||
Traditionally, those species that form large networks of anastomosing pseudopodia, despite some of them having tests, are not counted amongst testate amoebae; this comprises genus Gromia and the Foraminifera (both in Rhizaria).[2]
Notes
[edit]The Thecamoebida (Amoebozoa), with the genus Thecamoeba, despite their name, do not have tests.
Euglyphid testate amoebae are closely related to the Foraminifera.[9]
External links
[edit]- Microworld - World of ameboid organisms - A database of both testate and naked amoebae with over 6,700 microphotographs and videos and over 1,700 species descriptions, as well as dichotomous and visual keys for identification.
References
[edit]- ^ B., Scott, D. (2001). Monitoring in coastal environments using Foraminifera and Thecamoebian indicators. Medioli, F. S., Schafer, Charles T. Cambridge: Cambridge University Press. ISBN 978-0-521-56173-0. OCLC 70724931.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ a b Kosakyan, Anush; Gomaa, Fatma; Lara, Enrique; Lahr, Daniel J.G. (2016). "Current and future perspectives on the systematics, taxonomy and nomenclature of testate amoebae". European Journal of Protistology. 55 (Pt B): 105–117. doi:10.1016/j.ejop.2016.02.001. hdl:11380/1302106. PMID 27004416.
- ^ Sigala, Itzel; Lozano-García, Socorro; Escobar, Jaime; Pérez, Liseth; Gallegos-Neyra, Elvia (2016-06-28). "Testate Amoebae (Amebozoa: Arcellinida) in Tropical Lakes of Central Mexico" (PDF). Revista de Biología Tropical. 64 (1): 377–397. doi:10.15517/rbt.v64i1.18004. ISSN 2215-2075. PMID 28862826.
- ^ Mitchell, Edward A. D.; Charman, Daniel J.; Warner, Barry G. (2008-08-01). "Testate amoebae analysis in ecological and paleoecological studies of wetlands: past, present and future" (PDF). Biodiversity and Conservation. 17 (9): 2115–2137. doi:10.1007/s10531-007-9221-3. ISSN 0960-3115. S2CID 27912982.
- ^ Ralf Meisterfeld: Arcellinida, In: John J. Lee, Gordon F. Leedale, Phyllis Bradbury (Hrsg.): Illustrated Guide to the Protozoa, 2nd Edition. Vol. 2, Society of Protozoologists, Lawrence, Kansas 2000, ISBN 1-891276-23-9, pp. 827-860
- ^ Ralf Meisterfeld: Testate amoebae with filopodia , In: John J. Lee, Gordon F. Leedale, Phyllis Bradbury (Hrsg.): The Illustrated Guide to the Protozoa, 2nd Edition. Vol. 2, Society of Protozoologists, Lawrence, Kansas 2000, ISBN 1-891276-23-9, pp. 1054-1084
- ^ a b c Adl, Sina M.; Simpson, Alastair G. B.; Lane, Christopher E.; Lukeš, Julius; Bass, David; Bowser, Samuel S.; Brown, Matthew W.; Burki, Fabien; Dunthorn, Micah (2012-09-01). "The Revised Classification of Eukaryotes". Journal of Eukaryotic Microbiology. 59 (5): 429–514. doi:10.1111/j.1550-7408.2012.00644.x. ISSN 1550-7408. PMC 3483872. PMID 23020233.
- ^ Ruggiero, Michael A.; Gordon, Dennis P.; Orrell, Thomas M.; Bailly, Nicolas; Bourgoin, Thierry; Brusca, Richard C.; Cavalier-Smith, Thomas; Guiry, Michael D.; Kirk, Paul M. (2015-04-29). "A Higher Level Classification of All Living Organisms". PLOS ONE. 10 (4) e0119248. Bibcode:2015PLoSO..1019248R. doi:10.1371/journal.pone.0119248. ISSN 1932-6203. PMC 4418965. PMID 25923521.
- ^ Testate amoebae as environmental indicators (PDF)
Bibliography
[edit]- Medioli, F.S.; Scott, D.B.; Collins, E.; Asioli, S.; Reinhardt, E.G. (1999). The thecamoebian bibliography. Palaeontologia Electronica, 3: 1-161, [1].
- Medioli, F.S.; Bonnet, L.; Scott, D.B.; Medioli, B.E. (2003). The thecamoebian bibliography: 2nd edition. Palaeontologia Electronica, 61: 1-107, [2].