Recent from talks
Nothing was collected or created yet.
Blood pressure
View on Wikipedia| Blood pressure | |
|---|---|
 A healthcare worker measuring blood pressure using a sphygmomanometer. | |
| MeSH | D001794 |
| MedlinePlus | 007490 |
| LOINC | 35094-2 |
Blood pressure (BP) is the pressure of circulating blood against the walls of blood vessels. Most of this pressure results from the heart pumping blood through the circulatory system. When used without qualification, the term "blood pressure" refers to the pressure in a brachial artery, where it is most commonly measured. Blood pressure is usually expressed in terms of the systolic pressure (maximum pressure during one heartbeat) over diastolic pressure (minimum pressure between two heartbeats) in the cardiac cycle. It is measured in millimetres of mercury (mmHg) above the surrounding atmospheric pressure, or in kilopascals (kPa). The difference between the systolic and diastolic pressures is known as pulse pressure,[1] while the average pressure during a cardiac cycle is known as mean arterial pressure.[2]
Blood pressure is one of the vital signs—together with respiratory rate, heart rate, oxygen saturation, and body temperature—that healthcare professionals use in evaluating a patient's health. Normal resting blood pressure in an adult is approximately 120 millimetres of mercury (16 kPa) systolic over 80 millimetres of mercury (11 kPa) diastolic, denoted as "120/80 mmHg". Globally, the average blood pressure, age standardized, has remained about the same since 1975 to the present,[when?] at approximately 127/79 mmHg in men and 122/77 mmHg in women, although these average data mask significantly diverging regional trends.[3]
Traditionally, a health-care worker measured blood pressure non-invasively by auscultation (listening) through a stethoscope for sounds in one arm's artery as the artery is squeezed, closer to the heart, by an aneroid gauge or a mercury-tube sphygmomanometer.[4] Auscultation is still generally considered to be the gold standard of accuracy for non-invasive blood pressure readings in clinic.[5] However, semi-automated methods have become common, largely due to concerns about potential mercury toxicity,[6] although cost, ease of use and applicability to ambulatory blood pressure or home blood pressure measurements have also influenced this trend.[7] Early automated alternatives to mercury-tube sphygmomanometers were often seriously inaccurate, but modern devices validated to international standards achieve an average difference between two standardized reading methods of 5 mm Hg or less, and a standard deviation of less than 8 mm Hg.[7] Most of these semi-automated methods measure blood pressure using oscillometry (measurement by a pressure transducer in the cuff of the device of small oscillations of intra-cuff pressure accompanying heartbeat-induced changes in the volume of each pulse).[8]
Blood pressure is influenced by cardiac output, systemic vascular resistance, blood volume and arterial stiffness, and varies depending on person's situation, emotional state, activity and relative health or disease state. In the short term, blood pressure is regulated by baroreceptors, which act via the brain to influence the nervous and the endocrine systems.
Blood pressure that is too low is called hypotension, pressure that is consistently too high is called hypertension, and normal pressure is called normotension.[9] Both hypertension and hypotension have many causes and may be of sudden onset or of long duration. Long-term hypertension is a risk factor for many diseases, including stroke, heart disease, and kidney failure. Long-term hypertension is more common than long-term hypotension.
Classification, normal and abnormal values
[edit]Systemic arterial pressure
[edit]Blood pressure measurements can be influenced by circumstances of measurement.[10] Guidelines use different thresholds for office (also known as clinic), home (when the person measures their own blood pressure at home), and ambulatory blood pressure (using an automated device over a 24-hour period).[10]
| Categories | Systolic blood pressure, mmHg | and/or | Diastolic blood pressure, mmHg | ||||
|---|---|---|---|---|---|---|---|
| Method | Office | Home | 24h ambulatory | Office | Home | 24h ambulatory | |
| American College of Cardiology/American Heart Association (2017)[11] | |||||||
| Normal | <120 | <120 | <115 | and | <80 | <80 | <75 |
| Elevated | 120–129 | 120–129 | 115–124 | and | <80 | <80 | <75 |
| Hypertension, stage 1 | 130–139 | 130–134 | 125–129 | or | 80–89 | 80–84 | 75–79 |
| Hypertension, stage 2 | ≥140 | ≥135 | ≥130 | or | ≥90 | ≥85 | ≥80 |
| European Society of Cardiology (2024)[10] | |||||||
| Non-elevated | <120 | <120 | <115 | and | <70 | <70 | <65 |
| Elevated | 120–139 | 120–134 | 115–129 | and | 70–89 | 70–84 | 65–79 |
| Hypertension | ≥140 | ≥135 | ≥130 | or | ≥90 | ≥85 | ≥80 |
| European Society of Hypertension/International Society of Hypertension (2023)[12] | |||||||
| Optimal | <120 | — | — | and | <80 | — | — |
| Normal | 120–129 | — | — | and/or | 80–84 | — | — |
| High normal | 130–139 | — | — | and/or | 85–89 | — | — |
| Hypertension, grade 1 | 140–159 | ≥135 | ≥130 | and/or | 90–99 | ≥85 | ≥80 |
| Hypertension, grade 2 | 160–179 | — | — | and/or | 100–109 | — | — |
| Hypertension, grade 3 | ≥180 | — | — | and/or | ≥110 | — | — |
The risk of cardiovascular disease increases progressively above 90 mmHg, especially among women.[10]
Observational studies demonstrate that people who maintain arterial pressures at the low end of these pressure ranges have much better long-term cardiovascular health. There is an ongoing medical debate over what is the optimal level of blood pressure to target when using drugs to lower blood pressure with hypertension, particularly in older people.[13]
Blood pressure fluctuates from minute to minute and normally shows a circadian rhythm over a 24-hour period,[14] with highest readings in the early morning and evenings and lowest readings at night.[15][16] Loss of the normal fall in blood pressure at night is associated with a greater future risk of cardiovascular disease and there is evidence that night-time blood pressure is a stronger predictor of cardiovascular events than day-time blood pressure.[17] Blood pressure varies over longer time periods (months to years) and this variability predicts adverse outcomes.[18] Blood pressure also changes in response to temperature, noise, emotional stress, consumption of food or liquid, dietary factors, physical activity, changes in posture (such as standing-up), drugs, and disease.[19] The variability in blood pressure and the better predictive value of ambulatory blood pressure measurements has led some authorities, such as the National Institute for Health and Care Excellence (NICE) in the UK, to advocate for the use of ambulatory blood pressure as the preferred method for diagnosis of hypertension.[20]

Various other factors, such as age and sex, also influence a person's blood pressure. Differences between left-arm and right-arm blood pressure measurements tend to be small. However, occasionally there is a consistent difference greater than 10 mmHg which may need further investigation, e.g. for peripheral arterial disease, obstructive arterial disease or aortic dissection.[21][22][23][24]
There is no accepted diagnostic standard for hypotension, although pressures less than 90/60 are commonly regarded as hypotensive.[25] In practice blood pressure is considered too low only if symptoms are present.[26]
Systemic arterial pressure and age
[edit]Fetal blood pressure
[edit]In pregnancy, it is the fetal heart and not the mother's heart that builds up the fetal blood pressure to drive blood through the fetal circulation. The blood pressure in the fetal aorta is approximately 30 mmHg at 20 weeks of gestation, and increases to approximately 45 mmHg at 40 weeks of gestation.[27]
The average blood pressure for full-term infants:[28]
- Systolic 65–95 mmHg
- Diastolic 30–60 mmHg
Childhood
[edit]| Stage | Approximate age | Systolic BP, mmHg |
Diastolic BP, mmHg |
|---|---|---|---|
| Infants | 0–12 months | 75–100 | 50–70 |
| Toddlers and preschoolers | 1–5 years | 80–110 | 50–80 |
| School age | 6–12 years | 85–120 | 50–80 |
| Adolescents | 13–18 years | 95–140 | 60–90 |
In children the normal ranges for blood pressure are lower than for adults and depend on height.[30] Reference blood pressure values have been developed for children in different countries, based on the distribution of blood pressure in children of these countries.[31]
Aging adults
[edit]In adults in most societies, systolic blood pressure tends to rise from early adulthood onward, up to at least age 70;[32][33] diastolic pressure tends to begin to rise at the same time but start to fall earlier in mid-life, approximately age 55.[33] Mean blood pressure rises from early adulthood, plateauing in mid-life, while pulse pressure rises quite markedly after the age of 40. Consequently, in many older people, systolic blood pressure often exceeds the normal adult range,[33] if the diastolic pressure is in the normal range this is termed isolated systolic hypertension. The rise in pulse pressure with age is attributed to increased stiffness of the arteries.[34] An age-related rise in blood pressure is not considered healthy and is not observed in some isolated unacculturated communities.[35]
Systemic venous pressure
[edit]| Site | Normal pressure range (in mmHg)[36] | |
|---|---|---|
| Central venous pressure | 3–8 | |
| Right ventricular pressure | systolic | 15–30 |
| diastolic | 3–8 | |
| Pulmonary artery pressure | systolic | 15–30 |
| diastolic | 4–12 | |
| Pulmonary vein/ |
2–15 | |
| Left ventricular pressure | systolic | 100–140 |
| diastolic | 3–12 | |
Blood pressure generally refers to the arterial pressure in the systemic circulation. However, measurement of pressures in the venous system and the pulmonary vessels plays an important role in intensive care medicine but requires invasive measurement of pressure using a catheter.
Venous pressure is the vascular pressure in a vein or in the atria of the heart. It is much lower than arterial pressure, with common values of 5 mmHg in the right atrium and 8 mmHg in the left atrium.
Variants of venous pressure include:
- Central venous pressure, which is a good approximation of right atrial pressure,[37] which is a major determinant of right ventricular end diastolic volume. (However, there can be exceptions in some cases.)[38]
- The jugular venous pressure (JVP) is the indirectly observed pressure over the venous system. It can be useful in the differentiation of different forms of heart and lung disease.
- The portal venous pressure is the blood pressure in the portal vein. It is normally 5–10 mmHg[39]
Pulmonary pressure
[edit]Normally, the pressure in the pulmonary artery is about 15 mmHg at rest.[40]
Increased blood pressure in the capillaries of the lung causes pulmonary hypertension, leading to interstitial edema if the pressure increases to above 20 mmHg, and to pulmonary edema at pressures above 25 mmHg.[41]
Aortic pressure
[edit]Aortic pressure, also called central aortic blood pressure, or central blood pressure, is the blood pressure at the root of the aorta. Elevated aortic pressure has been found to be a more accurate predictor of both cardiovascular events and mortality, as well as structural changes in the heart, than has peripheral blood pressure (such as measured through the brachial artery).[42][43] Traditionally it involved an invasive procedure to measure aortic pressure, but now there are non-invasive methods of measuring it indirectly without a significant margin of error.[44][45]
Certain researchers have argued for physicians to begin using aortic pressure, as opposed to peripheral blood pressure, as a guide for clinical decisions.[46][43] The way antihypertensive drugs impact peripheral blood pressure can often be very different from the way they impact central aortic pressure.[47]
Mean systemic pressure
[edit]If the heart is stopped, blood pressure falls, but it does not fall to zero. The remaining pressure measured after cessation of the heart beat and redistribution of blood throughout the circulation is termed the mean systemic pressure or mean circulatory filling pressure;[48] typically this is proximally ~7 mmHg.[48]
Disorders of blood pressure
[edit]Disorders of blood pressure control include high blood pressure, low blood pressure, and blood pressure that shows excessive or maladaptive fluctuation.
High blood pressure
[edit]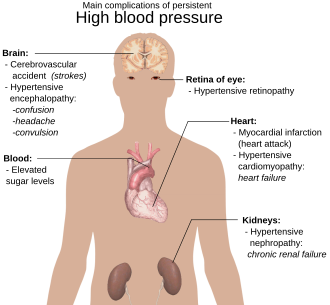
Arterial hypertension can be an indicator of other problems and may have long-term adverse effects. Sometimes it can be an acute problem, such as in a hypertensive emergency when blood pressure is more than 180/120 mmHg.[49]
Levels of arterial pressure put mechanical stress on the arterial walls. Higher pressures increase heart workload and progression of unhealthy tissue growth (atheroma) that develops within the walls of arteries. The higher the pressure, the more stress that is present and the more atheroma tend to progress and the heart muscle tends to thicken, enlarge and become weaker over time.
Persistent hypertension is one of the risk factors for strokes, heart attacks, heart failure, and arterial aneurysms, and is the leading cause of chronic kidney failure.[49] Even moderate elevation of arterial pressure leads to shortened life expectancy.[49] At severely high pressures, mean arterial pressures 50% or more above average, a person can expect to live no more than a few years unless appropriately treated.[50] For people with high blood pressure, higher heart rate variability (HRV) is a risk factor for atrial fibrillation.[51]
Both high systolic pressure and high pulse pressure (the numerical difference between systolic and diastolic pressures) are risk factors.[49] Elevated pulse pressure has been found to be a stronger independent predictor of cardiovascular events, especially in older populations, than has systolic, diastolic, or mean arterial pressure.[52][53][54][55] In some cases, it appears that a decrease in excessive diastolic pressure can actually increase risk, probably due to the increased difference between systolic and diastolic pressures (ie. widened pulse pressure). If systolic blood pressure is elevated (>140 mmHg) with a normal diastolic blood pressure (<90 mmHg), it is called isolated systolic hypertension and may present a health concern.[49][56] According to the 2017[57] American Heart Association blood pressure guidelines state that a systolic blood pressure of 130–139 mmHg with a diastolic pressure of 80–89 mmHg is "stage one hypertension".[49]
For those with heart valve regurgitation, a change in its severity may be associated with a change in diastolic pressure. In a study of people with heart valve regurgitation that compared measurements two weeks apart for each person, there was an increased severity of aortic and mitral regurgitation when diastolic blood pressure increased, whereas when diastolic blood pressure decreased, there was a decreased severity.[58]
Low blood pressure
[edit]Blood pressure that is too low is known as hypotension. This is a medical concern if it causes signs or symptoms, such as dizziness, fainting, or in extreme cases in medical emergencies, circulatory shock.[59] Causes of low arterial pressure include sepsis, hypovolemia, bleeding, cardiogenic shock, reflex syncope, hormonal abnormalities such as Addison's disease, and eating disorders – particularly anorexia nervosa and bulimia.[60]
Orthostatic hypotension
[edit]A large fall in blood pressure upon standing (typically a systolic/diastolic blood pressure decrease of >20/10 mmHg) is termed orthostatic hypotension (postural hypotension) and represents a failure of the body to compensate for the effect of gravity on the circulation. Standing results in an increased hydrostatic pressure in the blood vessels of the lower limbs. The consequent distension of the veins below the diaphragm (venous pooling) causes ~500 ml of blood to be relocated from the chest and upper body. This results in a rapid decrease in central blood volume and a reduction of ventricular preload which in turn reduces stroke volume, and mean arterial pressure. Normally this is compensated for by multiple mechanisms, including activation of the autonomic nervous system which increases heart rate, myocardial contractility and systemic arterial vasoconstriction to preserve blood pressure and elicits venous vasoconstriction to decrease venous compliance. Decreased venous compliance also results from an intrinsic myogenic increase in venous smooth muscle tone in response to the elevated pressure in the veins of the lower body.
Other compensatory mechanisms include the veno-arteriolar axon reflex, the 'skeletal muscle pump' and 'respiratory pump'. Together these mechanisms normally stabilize blood pressure within a minute or less.[61] If these compensatory mechanisms fail and arterial pressure and blood flow decrease beyond a certain point, the perfusion of the brain becomes critically compromised (i.e., the blood supply is not sufficient), causing lightheadedness, dizziness, weakness or fainting.[62] Usually this failure of compensation is due to disease, or drugs that affect the sympathetic nervous system.[61] A similar effect is observed following the experience of excessive gravitational forces (G-loading), such as routinely experienced by aerobatic or combat pilots 'pulling Gs' where the extreme hydrostatic pressures exceed the ability of the body's compensatory mechanisms.
Variable or fluctuating blood pressure
[edit]Some fluctuation or variation in blood pressure is normal. Variation in blood pressure that is significantly greater than the norm is known as labile hypertension and is associated with increased risk of cardiovascular disease[63] brain small vessel disease,[64] and dementia[65] independent of the average blood pressure level. Recent evidence from clinical trials has also linked variation in blood pressure to mortality,[66][67] stroke,[68] heart failure,[69] and cardiac changes that may give rise to heart failure.[70] These data have prompted discussion of whether excessive variation in blood pressure should be treated, even among normotensive older adults.[71]
Older individuals and those who had received blood pressure medications are more likely to exhibit larger fluctuations in pressure,[72] and there is some evidence that different antihypertensive agents have different effects on blood pressure variability;[65] whether these differences translate to benefits in outcome is uncertain.[65]
Physiology
[edit]


During each heartbeat, blood pressure varies between a maximum (systolic) and a minimum (diastolic) pressure.[73][unreliable medical source] The blood pressure in the circulation is principally due to the pumping action of the heart.[74] However, blood pressure is also regulated by neural regulation from the brain (see Hypertension and the brain), as well as osmotic regulation from the kidney. Differences in mean blood pressure drive the flow of blood around the circulation. The rate of mean blood flow depends on both blood pressure and the resistance to flow presented by the blood vessels. In the absence of hydrostatic effects (e.g. standing), mean blood pressure decreases as the circulating blood moves away from the heart through arteries and capillaries due to viscous losses of energy. Mean blood pressure drops over the whole circulation, although most of the fall occurs along the small arteries and arterioles.[75] Pulsatility also diminishes in the smaller elements of the arterial circulation, although some transmitted pulsatility is observed in capillaries.[76] Gravity affects blood pressure via hydrostatic forces (e.g., during standing), and valves in veins, breathing, and pumping from contraction of skeletal muscles also influence blood pressure, particularly in veins.[74]
Hemodynamics
[edit]A simple view of the hemodynamics of systemic arterial pressure is based around mean arterial pressure (MAP) and pulse pressure. Most influences on blood pressure can be understood in terms of their effect on cardiac output,[77] systemic vascular resistance, or arterial stiffness (the inverse of arterial compliance). Cardiac output is the product of stroke volume and heart rate. Stroke volume is influenced by 1) the end-diastolic volume or filling pressure of the ventricle acting via the Frank–Starling mechanism—this is influenced by blood volume; 2) cardiac contractility; and 3) afterload, the impedance to blood flow presented by the circulation.[78] In the short-term, the greater the blood volume, the higher the cardiac output. This has been proposed as an explanation of the relationship between high dietary salt intake and increased blood pressure; however, responses to increased dietary sodium intake vary between individuals and are highly dependent on autonomic nervous system responses and the renin–angiotensin system,[79][80][81] changes in plasma osmolarity may also be important.[82] In the longer-term the relationship between volume and blood pressure is more complex.[83] In simple terms, systemic vascular resistance is mainly determined by the caliber of small arteries and arterioles. The resistance attributable to a blood vessel depends on its radius as described by the Hagen-Poiseuille's equation (resistance∝1/radius4). Hence, the smaller the radius, the higher the resistance. Other physical factors that affect resistance include: vessel length (the longer the vessel, the higher the resistance), blood viscosity (the higher the viscosity, the higher the resistance)[84] and the number of vessels, particularly the smaller numerous, arterioles and capillaries. The presence of a severe arterial stenosis increases resistance to flow, however this increase in resistance rarely increases systemic blood pressure because its contribution to total systemic resistance is small, although it may profoundly decrease downstream flow.[85] Substances called vasoconstrictors reduce the caliber of blood vessels, thereby increasing blood pressure. Vasodilators (such as nitroglycerin) increase the caliber of blood vessels, thereby decreasing arterial pressure. In the longer term a process termed remodeling also contributes to changing the caliber of small blood vessels and influencing resistance and reactivity to vasoactive agents.[86][87] Reductions in capillary density, termed capillary rarefaction, may also contribute to increased resistance in some circumstances.[88]
In practice, each individual's autonomic nervous system and other systems regulating blood pressure, notably the kidney,[89] respond to and regulate all these factors so that, although the above issues are important, they rarely act in isolation and the actual arterial pressure response of a given individual can vary widely in the short and long term.
Pulse pressure
[edit]
The pulse pressure is the difference between the measured systolic and diastolic pressures,[90]
The pulse pressure is a consequence of the pulsatile nature of the cardiac output, i.e. the heartbeat. The magnitude of the pulse pressure is usually attributed to the interaction of the stroke volume of the heart, the compliance (ability to expand) of the arterial system—largely attributable to the aorta and large elastic arteries—and the resistance to flow in the arterial tree.[90]
Clinical significance of pulse pressure
[edit]A healthy pulse pressure is around 40 mmHg.[1] A pulse pressure that is consistently 60 mmHg or greater is likely to be associated with disease, and a pulse pressure of 50 mmHg or more increases the risk of cardiovascular disease as well as other complications such as eye and kidney disease.[52] Pulse pressure is considered low if it is less than 25% of the systolic. (For example, if the systolic pressure is 120 mmHg, then the pulse pressure would be considered low if it is less than 30 mmHg, since 30 is 25% of 120.)[91] A very low pulse pressure can be a symptom of disorders such as congestive heart failure.[52]
Elevated pulse pressure has been found to be a stronger independent predictor of cardiovascular events, especially in older populations, than has systolic, diastolic, or mean arterial pressure.[52][53] This increased risk exists for both men and women and even when no other cardiovascular risk factors are present. The increased risk also exists even in cases in which diastolic pressure decreases over time while systolic remains steady.[55][54]
A meta-analysis in 2000 showed that a 10 mmHg increase in pulse pressure was associated with a 20% increased risk of cardiovascular mortality, and a 13% increase in risk for all coronary end points. The study authors also noted that, while risks of cardiovascular end points do increase with higher systolic pressures, at any given systolic blood pressure the risk of major cardiovascular end points increases, rather than decreases, with lower diastolic levels. This suggests that interventions that lower diastolic pressure without also lowering systolic pressure (and thus lowering pulse pressure) could actually be counterproductive.[92] There are no drugs currently approved to lower pulse pressure, although some antihypertensive drugs may modestly lower pulse pressure, while in some cases a drug that lowers overall blood pressure may actually have the counterproductive side effect of raising pulse pressure.[93]
Pulse pressure can both widen or narrow in people with sepsis depending on the degree of hemodynamic compromise. A pulse pressure of over 70 mmHg in sepsis is correlated with an increased chance of survival and a more positive response to IV fluids.[94][95]
Mean arterial pressure
[edit]Mean arterial pressure (MAP) is the average of blood pressure over a cardiac cycle and is determined by the cardiac output (CO), systemic vascular resistance (SVR), and central venous pressure (CVP):[2][96][97]
In practice, the contribution of CVP (which is small) is generally ignored and so
MAP is often estimated from measurements of the systolic pressure, and the diastolic pressure, [97] using the equation:
where k = 0.333 although other values for k have been advocated.[98][99]
Regulation of blood pressure
[edit]The endogenous, homeostatic regulation of arterial pressure is not completely understood, but the following mechanisms of regulating arterial pressure have been well-characterized:
- Baroreceptor reflex: Baroreceptors in the high pressure receptor zones detect changes in arterial pressure. These baroreceptors send signals ultimately to the medulla of the brain stem, specifically to the rostral ventrolateral medulla (RVLM). The medulla, by way of the autonomic nervous system, adjusts the mean arterial pressure by altering both the force and speed of the heart's contractions, as well as the systemic vascular resistance. The most important arterial baroreceptors are located in the left and right carotid sinuses and in the aortic arch.[100]
- Renin–angiotensin system (RAS): This system is generally known for its long-term adjustment of arterial pressure. This system allows the kidney to compensate for loss in blood volume or drops in arterial pressure by activating an endogenous vasoconstrictor known as angiotensin II.
- Aldosterone release: This steroid hormone is released from the adrenal cortex in response to activation of the renin-angiotensin system, high serum potassium levels, or elevated adrenocorticotropic hormone (ACTH). Renin converts angiotensinogen to angiotensin I, which is converted by angiotensin converting enzyme to angiotensin II. Angiotensin II then signals to the adrenal cortex to release aldosterone.[101] Aldosterone stimulates sodium retention and potassium excretion by the kidneys and the consequent salt and water retention increases plasma volume, and indirectly, arterial pressure. Aldosterone may also exert direct pressor effects on vascular smooth muscle and central effects on sympathetic nervous system activity.[102]
- Baroreceptors in low pressure receptor zones (mainly in the venae cavae and the pulmonary veins, and in the atria) result in feedback by regulating the secretion of antidiuretic hormone (ADH/vasopressin), renin and aldosterone. The resultant increase in blood volume results in an increased cardiac output by the Frank–Starling law of the heart, in turn increasing arterial blood pressure.
These different mechanisms are not necessarily independent of each other, as indicated by the link between the RAS and aldosterone release. When blood pressure falls many physiological cascades commence in order to return the blood pressure to a more appropriate level.
- The blood pressure fall is detected by a decrease in blood flow and thus a decrease in glomerular filtration rate (GFR).
- Decrease in GFR is sensed as a decrease in Na+ levels by the macula densa.
- The macula densa causes an increase in Na+ reabsorption, which causes water to follow in via osmosis and leads to an ultimate increase in plasma volume. Further, the macula densa releases adenosine which causes constriction of the afferent arterioles.
- At the same time, the juxtaglomerular cells sense the decrease in blood pressure and release renin.
- Renin converts angiotensinogen (inactive form) to angiotensin I (active form).
- Angiotensin I flows in the bloodstream until it reaches the capillaries of the lungs where angiotensin-converting enzyme (ACE) acts on it to convert it into angiotensin II.
- Angiotensin II is a vasoconstrictor that will increase blood flow to the heart and subsequently the preload, ultimately increasing the cardiac output.
- Angiotensin II also causes an increase in the release of aldosterone from the adrenal glands.
- Aldosterone further increases the Na+ and H2O reabsorption in the distal convoluted tubule of the nephron.
The RAS is targeted pharmacologically by ACE inhibitors and angiotensin II receptor antagonists (also known as angiotensin receptor blockers; ARB). The aldosterone system is directly targeted by aldosterone antagonists. The fluid retention may be targeted by diuretics; the antihypertensive effect of diuretics is due to its effect on blood volume. Generally, the baroreceptor reflex is not targeted in hypertension because if blocked, individuals may experience orthostatic hypotension and fainting.
Measurement
[edit]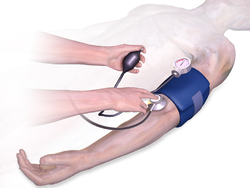

Arterial pressure is most commonly measured via a sphygmomanometer, which uses the height of a column of mercury, or an aneroid gauge, to reflect the blood pressure by auscultation.[4] The most common automated blood pressure measurement technique is based on the oscillometric method.[103] Fully automated oscillometric measurement has been available since 1981.[104] This principle has recently been used to measure blood pressure with a smartphone.[105] Measuring pressure invasively, by penetrating the arterial wall to take the measurement, is much less common and usually restricted to a hospital setting. Novel methods to measure blood pressure without penetrating the arterial wall, and without applying any pressure on patient's body are being explored,[106] for example, cuffless measurements that uses only optical sensors.[107]
In office blood pressure measurement, terminal digit preference is common. According to one study, approximately 40% of recorded measurements ended with the digit zero, whereas "without bias, 10%–20% of measurements are expected to end in zero"[108]
In animals
[edit]Blood pressure levels in non-human mammals may vary depending on the species. Heart rate differs markedly, largely depending on the size of the animal (larger animals have slower heart rates).[109] The giraffe has a distinctly high arterial pressure of about 190 mm Hg, enabling blood perfusion through the 2 metres (6 ft 7 in)-long neck to the head.[110] In other species subjected to orthostatic blood pressure, such as arboreal snakes, blood pressure is higher than in non-arboreal snakes.[111] A heart near to the head (short heart-to-head distance) and a long tail with tight integument favor blood perfusion to the head.[112][113]
As in humans, blood pressure in animals differs by age, sex, time of day, and environmental circumstances:[114][115] measurements made in laboratories or under anesthesia may not be representative of values under free-living conditions. Rats, mice, dogs and rabbits have been used extensively to study the regulation of blood pressure.[116]
| Species | Blood pressure mm Hg |
Heart rate beats per minute | |
|---|---|---|---|
| Systolic | Diastolic | ||
| Calves | 140 | 70 | 75–146 |
| Cats | 155 | 68 | 100–259 |
| Dogs | 161 | 51 | 62–170 |
| Goats | 140 | 90 | 80–120 |
| Guinea-pigs | 140 | 90 | 240–300 |
| Mice | 120 | 75 | 580–680 |
| Pigs | 169 | 55 | 74–116 |
| Rabbits | 118 | 67 | 205–306 |
| Rats | 153 | 51 | 305–500 |
| Rhesus monkeys | 160 | 125 | 180–210 |
| Sheep | 140 | 80 | 63–210 |
Hypertension in cats and dogs
[edit]Hypertension in cats and dogs is generally diagnosed if the blood pressure is greater than 150 mm Hg (systolic),[117] although sight hounds have higher blood pressures than most other dog breeds; a systolic pressure greater than 180 mmHg is considered abnormal in these dogs.[118]
See also
[edit]References
[edit]- ^ a b Homan TD, Bordes SJ, Cichowski E (12 July 2022). "Physiology, Pulse Pressure". StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. PMID 29494015. Retrieved 2019-07-21 – via NCBI Bookshelf.
- ^ a b Mayet J, Hughes A (September 2003). "Cardiac and vascular pathophysiology in hypertension". Heart. 89 (9): 1104–1109. doi:10.1136/heart.89.9.1104. PMC 1767863. PMID 12923045.
- ^ Zhou B, Bentham J, Di Cesare M, Bixby H, Danaei G, Cowan MJ, et al. (NCD Risk Factor Collaboration (NCD-RisC)) (January 2017). "Worldwide trends in blood pressure from 1975 to 2015: a pooled analysis of 1479 population-based measurement studies with 19·1 million participants". Lancet. 389 (10064): 37–55. doi:10.1016/S0140-6736(16)31919-5. PMC 5220163. PMID 27863813.
- ^ a b Booth J (November 1977). "A short history of blood pressure measurement". Proceedings of the Royal Society of Medicine. 70 (11): 793–799. doi:10.1177/003591577707001112. PMC 1543468. PMID 341169.
- ^ Grim CE, Grim CM (March 2016). "Auscultatory BP: still the gold standard". Journal of the American Society of Hypertension. 10 (3): 191–193. doi:10.1016/j.jash.2016.01.004. PMID 26839183.
- ^ O'Brien E (January 2001). "Blood pressure measurement is changing!". Heart. 85 (1): 3–5. doi:10.1136/heart.85.1.3. PMC 1729570. PMID 11119446.
- ^ a b Ogedegbe G, Pickering T (November 2010). "Principles and techniques of blood pressure measurement". Cardiology Clinics. 28 (4): 571–586. doi:10.1016/j.ccl.2010.07.006. PMC 3639494. PMID 20937442.
- ^ Alpert BS, Quinn D, Gallick D (December 2014). "Oscillometric blood pressure: a review for clinicians". Journal of the American Society of Hypertension. 8 (12): 930–938. doi:10.1016/j.jash.2014.08.014. PMID 25492837.
- ^ Newman WA, ed. (2012). Dorland's illustrated medical dictionary (32nd ed.). Philadelphia, PA: Saunders/Elsevier. ISBN 978-1-4160-6257-8. OCLC 706780870.
- ^ a b c d McEvoy, John William; McCarthy, Cian P; Bruno, Rosa Maria; Brouwers, Sofie; Canavan, Michelle D; et al. (2024-08-30). "2024 ESC Guidelines for the management of elevated blood pressure and hypertension: Developed by the task force on the management of elevated blood pressure and hypertension of the European Society of Cardiology (ESC) and endorsed by the European Society of Endocrinology (ESE) and the European Stroke Organisation (ESO)". European Heart Journal. 45 (38): 3912–4018. doi:10.1093/eurheartj/ehae178. ISSN 0195-668X. PMID 39210715.
- ^ Whelton, Paul K; Carey, Robert M; Mancia, Giuseppe; Kreutz, Reinhold; Bundy, Joshua D; Williams, Bryan (2022-09-14). "Harmonization of the American College of Cardiology/American Heart Association and European Society of Cardiology/European Society of Hypertension Blood Pressure/Hypertension Guidelines". European Heart Journal. 43 (35): 3302–3311. doi:10.1093/eurheartj/ehac432. ISSN 0195-668X. PMC 9470378. PMID 36100239.
- ^ Mancia, Giuseppe; Kreutz, Reinhold; Brunström, Mattias; Burnier, Michel; Grassi, Guido; et al. (2023-12-01). "2023 ESH Guidelines for the management of arterial hypertension The Task Force for the management of arterial hypertension of the European Society of Hypertension: Endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA)". Journal of Hypertension. 41 (12): 1874–2071. doi:10.1097/HJH.0000000000003480. hdl:11379/603005. ISSN 1473-5598. PMID 37345492.
- ^ Yusuf S, Lonn E (November 2016). "The SPRINT and the HOPE-3 Trial in the Context of Other Blood Pressure-Lowering Trials". JAMA Cardiology. 1 (8): 857–858. doi:10.1001/jamacardio.2016.2169. PMID 27602555.
- ^ Smolensky MH, Hermida RC, Portaluppi F (June 2017). "Circadian mechanisms of 24-hour blood pressure regulation and patterning". Sleep Medicine Reviews. 33: 4–16. doi:10.1016/j.smrv.2016.02.003. PMID 27076261.
- ^ van Berge-Landry HM, Bovbjerg DH, James GD (October 2008). "Relationship between waking-sleep blood pressure and catecholamine changes in African-American and European-American women". Blood Pressure Monitoring. 13 (5): 257–262. doi:10.1097/MBP.0b013e3283078f45. PMC 2655229. PMID 18799950.
Table2: Comparison of ambulatory blood pressures and urinary norepinephrine and epinephrine excretion measured at work, home, and during sleep between European–American (n = 110) and African–American (n = 51) women
- ^ van Berge-Landry HM, Bovbjerg DH, James GD (October 2008). "Relationship between waking-sleep blood pressure and catecholamine changes in African-American and European-American women". Blood Pressure Monitoring. 13 (5): 257–262. doi:10.1097/MBP.0b013e3283078f45. PMC 2655229. PMID 18799950. NIHMS90092.
- ^ Hansen TW, Li Y, Boggia J, Thijs L, Richart T, Staessen JA (January 2011). "Predictive role of the nighttime blood pressure". Hypertension. 57 (1): 3–10. doi:10.1161/HYPERTENSIONAHA.109.133900. PMID 21079049.
- ^ Rothwell PM (June 2011). "Does blood pressure variability modulate cardiovascular risk?". Current Hypertension Reports. 13 (3): 177–186. doi:10.1007/s11906-011-0201-3. PMID 21465141. S2CID 207331784.
- ^ Engel BT, Blümchen G, eds. (1992). Temporal Variations of the Cardiovascular System. Berlin, Heidelberg: Springer Berlin Heidelberg. ISBN 978-3-662-02748-6. OCLC 851391490.
- ^ National Clinical Guideline Centre (UK) (2011). Hypertension: The Clinical Management of Primary Hypertension in Adults: Update of Clinical Guidelines 18 and 34. National Institute for Health and Clinical Excellence: Guidance. London: Royal College of Physicians (UK). PMID 22855971. Archived from the original on 2022-08-12. Retrieved 2019-01-28.
- ^ Eguchi K, Yacoub M, Jhalani J, Gerin W, Schwartz JE, Pickering TG (February 2007). "Consistency of blood pressure differences between the left and right arms". Archives of Internal Medicine. 167 (4): 388–393. doi:10.1001/archinte.167.4.388. PMID 17325301.
- ^ Agarwal R, Bunaye Z, Bekele DM (March 2008). "Prognostic significance of between-arm blood pressure differences". Hypertension. 51 (3): 657–662. CiteSeerX 10.1.1.540.5836. doi:10.1161/HYPERTENSIONAHA.107.104943. PMID 18212263. S2CID 1101762.
- ^ Clark CE, Campbell JL, Evans PH, Millward A (December 2006). "Prevalence and clinical implications of the inter-arm blood pressure difference: A systematic review". Journal of Human Hypertension. 20 (12): 923–931. doi:10.1038/sj.jhh.1002093. PMID 17036043.
- ^ Clark CE, Warren FC, Boddy K, McDonagh ST, Moore SF, Goddard J, et al. (February 2021). "Associations Between Systolic Interarm Differences in Blood Pressure and Cardiovascular Disease Outcomes and Mortality: Individual Participant Data Meta-Analysis, Development and Validation of a Prognostic Algorithm: The INTERPRESS-IPD Collaboration". Hypertension. 77 (2): 650–661. doi:10.1161/HYPERTENSIONAHA.120.15997. PMC 7803446. PMID 33342236.
- ^ Sharma S, Bhattacharya PT (2018). "Hypotension". StatPearls. StatPearls Publishing. PMID 29763136. Archived from the original on 2020-03-17. Retrieved 2018-12-23.
- ^ Mayo Clinic staff (2009-05-23). "Low blood pressure (hypotension) – Causes". MayoClinic.com. Mayo Foundation for Medical Education and Research. Archived from the original on 2021-11-17. Retrieved 2010-10-19.
- ^ Struijk PC, Mathews VJ, Loupas T, Stewart PA, Clark EB, Steegers EA, Wladimiroff JW (October 2008). "Blood pressure estimation in the human fetal descending aorta". Ultrasound in Obstetrics & Gynecology. 32 (5): 673–681. doi:10.1002/uog.6137. hdl:1765/30025. PMID 18816497. S2CID 23575926.
- ^ Sharon SM, Emily SM (2006). Foundations of Maternal-Newborn Nursing (4th ed.). Philadelphia: Elsevier. p. 476.
- ^ Pediatric Age Specific Archived 2017-05-16 at the Wayback Machine, p. 6. Revised 6/10. By Theresa Kirkpatrick and Kateri Tobias. UCLA Health System
- ^ National Heart Lung and Blood Institute. "Blood pressure tables for children and adolescents". Archived from the original on 2014-06-18. Retrieved 2008-09-23. (The median blood pressure is given by the 50th percentile and hypertension is defined by the 95th percentile for a given age, height, and sex.)
- ^ Chiolero A (March 2014). "The quest for blood pressure reference values in children". Journal of Hypertension. 32 (3): 477–479. doi:10.1097/HJH.0000000000000109. PMID 24477093. S2CID 1949314.
- ^ Wills AK, Lawlor DA, Matthews FE, Sayer AA, Bakra E, Ben-Shlomo Y, et al. (June 2011). "Life course trajectories of systolic blood pressure using longitudinal data from eight UK cohorts". PLOS Medicine. 8 (6) e1000440. doi:10.1371/journal.pmed.1000440. PMC 3114857. PMID 21695075.
- ^ a b c Franklin SS, Gustin W, Wong ND, Larson MG, Weber MA, Kannel WB, Levy D (July 1997). "Hemodynamic patterns of age-related changes in blood pressure. The Framingham Heart Study". Circulation. 96 (1): 308–315. doi:10.1161/01.CIR.96.1.308. PMID 9236450. S2CID 40209177.
- ^ Franklin SS (2008-05-01). "Beyond blood pressure: Arterial stiffness as a new biomarker of cardiovascular disease". Journal of the American Society of Hypertension. 2 (3): 140–151. doi:10.1016/j.jash.2007.09.002. PMID 20409896.
- ^ Gurven M, Blackwell AD, Rodríguez DE, Stieglitz J, Kaplan H (July 2012). "Does blood pressure inevitably rise with age?: longitudinal evidence among forager-horticulturalists". Hypertension. 60 (1): 25–33. doi:10.1161/HYPERTENSIONAHA.111.189100. PMC 3392307. PMID 22700319.
- ^ Table 30-1 in: Goers TA, Klingensmith ME, Chen LE, Glasgow SC (2008). The Washington Manual of Surgery. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. ISBN 978-0-7817-7447-5.
- ^ "Central Venous Catheter Physiology". Archived from the original on 2008-08-21. Retrieved 2009-02-27.
- ^ Tkachenko BI, Evlakhov VI, Poyasov IZ (October 2002). "Independence of changes in right atrial pressure and central venous pressure". Bulletin of Experimental Biology and Medicine. 134 (4): 318–320. doi:10.1023/A:1021931508946. PMID 12533747. S2CID 23726657.
- ^ "Esophageal Varices: Article Excerpt by: Samy A Azer". eMedicine. Archived from the original on 2008-10-07. Retrieved 2011-08-22.
- ^ "What Is Pulmonary Hypertension?". From Diseases and Conditions Index (DCI). National Heart, Lung, and Blood Institute. September 2008. Archived from the original on 27 April 2012. Retrieved 6 April 2009.
- ^ Adair OV (2001). "Chapter 41". Cardiology secrets (2nd ed.). Philadelphia: Hanley & Belfus. p. 210. ISBN 978-1-56053-420-4.
- ^ Roman, Mary J.; Devereux, Richard B.; Kizer, Jorge R.; Lee, Elisa T.; Galloway, James M.; Ali, Tauqeer; Umans, Jason G.; Howard, Barbara V. (2007). "Central Pressure More Strongly Relates to Vascular Disease and Outcome Than Does Brachial Pressure". Hypertension. 50 (1). Ovid Technologies (Wolters Kluwer Health): 197–203. doi:10.1161/hypertensionaha.107.089078. ISSN 0194-911X. PMID 17485598.
- ^ a b Kesten, Steven; Qasem, Ahmad; Avolio, Alberto (2022-10-20). "Viewpoint: The Case for Non-Invasive Central Aortic Pressure Monitoring in the Management of Hypertension". Artery Research. 28 (4): 128–139. doi:10.1007/s44200-022-00023-z. ISSN 1876-4401.
- ^ Avolio, Alberto (2008). "Central Aortic Blood Pressure and Cardiovascular Risk: A Paradigm Shift?". Hypertension. 51 (6): 1470–1471. doi:10.1161/HYPERTENSIONAHA.107.108910. ISSN 0194-911X. PMID 18426994.
- ^ Chen, Chen-Huan; Nevo, Erez; Fetics, Barry; Pak, Peter H.; Yin, Frank C.P.; Maughan, W. Lowell; Kass, David A. (1997). "Estimation of Central Aortic Pressure Waveform by Mathematical Transformation of Radial Tonometry Pressure". Circulation. 95 (7). Ovid Technologies (Wolters Kluwer Health): 1827–1836. doi:10.1161/01.cir.95.7.1827. ISSN 0009-7322. PMID 9107170.
- ^ McEniery, Carmel M.; Cockcroft, John R.; Roman, Mary J.; Franklin, Stanley S.; Wilkinson, Ian B. (23 Jan 2014). "Central blood pressure: current evidence and clinical importance". European Heart Journal. 35 (26). Oxford University Press (OUP): 1719–1725. doi:10.1093/eurheartj/eht565. ISSN 1522-9645. PMC 4155427. PMID 24459197.
- ^ The CAFE Investigators; CAFE Steering Committee and Writing Committee; Williams, Bryan; Lacy, Peter S.; Thom, Simon M.; Cruickshank, Kennedy; Stanton, Alice; Collier, David; Hughes, Alun D.; Thurston, H.; O'Rourke, Michael (2006-03-07). "Differential Impact of Blood Pressure–Lowering Drugs on Central Aortic Pressure and Clinical Outcomes: Principal Results of the Conduit Artery Function Evaluation (CAFE) Study". Circulation. 113 (9): 1213–1225. doi:10.1161/CIRCULATIONAHA.105.595496. ISSN 0009-7322. PMID 16476843.
- ^ a b Rothe CF (February 1993). "Mean circulatory filling pressure: its meaning and measurement". Journal of Applied Physiology. 74 (2): 499–509. doi:10.1152/jappl.1993.74.2.499. PMID 8458763.
- ^ a b c d e f g "The facts about high blood pressure". American Heart Association. 2023. Archived from the original on 14 May 2023. Retrieved 14 May 2023.
- ^ Guyton AC (2006). Textbook of Medical Physiology (11th ed.). Philadelphia: Elsevier Saunders. p. 220. ISBN 978-0-7216-0240-0.
- ^ Kim SH, Lim KR, Chun KJ (2022). "Higher heart rate variability as a predictor of atrial fibrillation in patients with hypertensione". Scientific Reports. 12 (1) 3702. Bibcode:2022NatSR..12.3702K. doi:10.1038/s41598-022-07783-3. PMC 8904557. PMID 35260686.
- ^ a b c d "Pulse pressure". Cleveland Clinic. 28 July 2021. Retrieved 10 February 2023.
- ^ a b Mitchell, Gary F.; Izzo, Joseph L.; Lacourcière, Yves; Ouellet, Jean-Pascal; Neutel, Joel; Qian, Chunlin; Kerwin, Linda J.; Block, Alan J.; Pfeffer, Marc A. (25 Jun 2002). "Omapatrilat Reduces Pulse Pressure and Proximal Aortic Stiffness in Patients With Systolic Hypertension". Circulation. 105 (25). Ovid Technologies (Wolters Kluwer Health): 2955–2961. doi:10.1161/01.cir.0000020500.77568.3c. ISSN 0009-7322. PMID 12081987. S2CID 7092379.
- ^ a b Benetos, Athanase; Safar, Michel; Rudnichi, Annie; Smulyan, Harold; Richard, Jacques-Lucien; Ducimetière, Pierre; Guize, Louis (1997). "Pulse Pressure". Hypertension. 30 (6). Ovid Technologies (Wolters Kluwer Health): 1410–1415. doi:10.1161/01.hyp.30.6.1410. ISSN 0194-911X. PMID 9403561.
- ^ a b Franklin, Stanley S.; Khan, Shehzad A.; Wong, Nathan D.; Larson, Martin G.; Levy, Daniel (27 Jul 1999). "Is Pulse Pressure Useful in Predicting Risk for Coronary Heart Disease?". Circulation. 100 (4). Ovid Technologies (Wolters Kluwer Health): 354–360. doi:10.1161/01.cir.100.4.354. ISSN 0009-7322. PMID 10421594.
- ^ "Isolated systolic hypertension: A health concern?". MayoClinic.com. Archived from the original on 2013-12-28. Retrieved 2018-01-25.
- ^ Tan JL, Thakur K (2022). "Systolic Hypertension". StatPearls. Treasure Island (FL): StatPearls Publishing. PMID 29494079. Retrieved 2022-10-03.
- ^ Gottdiener JS, Panza JA, St John Sutton M, Bannon P, Kushner H, Weissman NJ (July 2002). "Testing the test: the reliability of echocardiography in the sequential assessment of valvular regurgitation". American Heart Journal. 144 (1): 115–121. doi:10.1067/mhj.2002.123139. PMID 12094197.
- ^ "Diseases and conditions index – hypotension". National Heart Lung and Blood Institute. September 2008. Archived from the original on 2012-04-27. Retrieved 2008-09-16.
- ^ Braunwald E, Bonow RO (2012). Braunwald's heart disease: a textbook of cardiovascular medicine (9th ed.). Philadelphia: Saunders. ISBN 978-1-4377-0398-6. OCLC 671465395.
- ^ a b Ricci F, De Caterina R, Fedorowski A (August 2015). "Orthostatic Hypotension: Epidemiology, Prognosis, and Treatment". Journal of the American College of Cardiology. 66 (7): 848–860. doi:10.1016/j.jacc.2015.06.1084. PMID 26271068.
- ^ Franco Folino A (2007). "Cerebral autoregulation and syncope". Progress in Cardiovascular Diseases. 50 (1): 49–80. doi:10.1016/j.pcad.2007.01.001. PMID 17631437.
- ^ Stevens SL, Wood S, Koshiaris C, Law K, Glasziou P, Stevens RJ, McManus RJ (August 2016). "Blood pressure variability and cardiovascular disease: systematic review and meta-analysis". BMJ. 354 i4098. doi:10.1136/bmj.i4098. PMC 4979357. PMID 27511067.
- ^ Tully PJ, Yano Y, Launer LJ, Kario K, Nagai M, Mooijaart SP, et al. (January 2020). "Association Between Blood Pressure Variability and Cerebral Small-Vessel Disease: A Systematic Review and Meta-Analysis". Journal of the American Heart Association. 9 (1) e013841. doi:10.1161/JAHA.119.013841. PMC 6988154. PMID 31870233.
- ^ a b c Messerli FH, Hofstetter L, Rimoldi SF, Rexhaj E, Bangalore S (May 2019). "Risk Factor Variability and Cardiovascular Outcome: JACC Review Topic of the Week". Journal of the American College of Cardiology. 73 (20): 2596–2603. doi:10.1016/j.jacc.2019.02.063. PMID 31118154.
- ^ Chiriacò M, Pateras K, Virdis A, Charakida M, Kyriakopoulou D, Nannipieri M, et al. (December 2019). "Association between blood pressure variability, cardiovascular disease and mortality in type 2 diabetes: A systematic review and meta-analysis". Diabetes, Obesity & Metabolism. 21 (12): 2587–2598. doi:10.1111/dom.13828. hdl:11568/996646. PMID 31282073. S2CID 195829708.
- ^ Nuyujukian DS, Newell MS, Zhou JJ, Koska J, Reaven PD (May 2022). "Baseline blood pressure modifies the role of blood pressure variability in mortality: Results from the ACCORD trial". Diabetes, Obesity & Metabolism. 24 (5): 951–955. doi:10.1111/dom.14649. PMC 8986598. PMID 35014154. S2CID 245896131.
- ^ Muntner P, Whittle J, Lynch AI, Colantonio LD, Simpson LM, Einhorn PT, et al. (September 2015). "Visit-to-Visit Variability of Blood Pressure and Coronary Heart Disease, Stroke, Heart Failure, and Mortality: A Cohort Study". Annals of Internal Medicine. 163 (5): 329–338. doi:10.7326/M14-2803. PMC 5021508. PMID 26215765.
- ^ Nuyujukian DS, Koska J, Bahn G, Reaven PD, Zhou JJ (July 2020). "Blood Pressure Variability and Risk of Heart Failure in ACCORD and the VADT". Diabetes Care. 43 (7): 1471–1478. doi:10.2337/dc19-2540. hdl:10150/641980. PMC 7305004. PMID 32327422.
- ^ Nwabuo CC, Yano Y, Moreira HT, Appiah D, Vasconcellos HD, Aghaji QN, et al. (July 2020). "Association Between Visit-to-Visit Blood Pressure Variability in Early Adulthood and Myocardial Structure and Function in Later Life". JAMA Cardiology. 5 (7): 795–801. doi:10.1001/jamacardio.2020.0799. PMC 7160747. PMID 32293640.
- ^ Parati G, Ochoa JE, Lombardi C, Bilo G (March 2013). "Assessment and management of blood-pressure variability". Nature Reviews. Cardiology. 10 (3): 143–155. doi:10.1038/nrcardio.2013.1. PMID 23399972. S2CID 22425558.
- ^ Brickman AM, Reitz C, Luchsinger JA, Manly JJ, Schupf N, Muraskin J, et al. (May 2010). "Long-term blood pressure fluctuation and cerebrovascular disease in an elderly cohort". Archives of Neurology. 67 (5): 564–569. doi:10.1001/archneurol.2010.70. PMC 2917204. PMID 20457955.
- ^ "Normal Blood Pressure Ranges in Adults". svollop.com. 2023-03-16. Archived from the original on 2023-03-16. Retrieved 2023-03-21.
- ^ a b Caro CG (1978). The Mechanics of The Circulation. Oxford [Oxfordshire]: Oxford University Press. ISBN 978-0-19-263323-1.
- ^ Klabunde R (2005). Cardiovascular Physiology Concepts. Lippincott Williams & Wilkins. pp. 93–94. ISBN 978-0-7817-5030-1.
- ^ Mahler F, Muheim MH, Intaglietta M, Bollinger A, Anliker M (June 1979). "Blood pressure fluctuations in human nailfold capillaries". The American Journal of Physiology. 236 (6): H888 – H893. doi:10.1152/ajpheart.1979.236.6.H888. PMID 443454.
- ^ Guyton AC (December 1981). "The relationship of cardiac output and arterial pressure control". Circulation. 64 (6): 1079–1088. doi:10.1161/01.cir.64.6.1079. PMID 6794930.
- ^ Milnor WR (May 1975). "Arterial impedance as ventricular afterload". Circulation Research. 36 (5): 565–570. doi:10.1161/01.res.36.5.565. PMID 1122568.
- ^ Freis ED (April 1976). "Salt, volume and the prevention of hypertension". Circulation. 53 (4): 589–595. doi:10.1161/01.CIR.53.4.589. PMID 767020.
- ^ Caplea A, Seachrist D, Dunphy G, Ely D (April 2001). "Sodium-induced rise in blood pressure is suppressed by androgen receptor blockade". American Journal of Physiology. Heart and Circulatory Physiology. 4. 280 (4): H1793 – H1801. doi:10.1152/ajpheart.2001.280.4.H1793. PMID 11247793. S2CID 12069178.
- ^ Houston MC (January 1986). "Sodium and hypertension. A review". Archives of Internal Medicine. 1. 146 (1): 179–185. doi:10.1001/archinte.1986.00360130217028. PMID 3510595.
- ^ Kanbay M, Aslan G, Afsar B, Dagel T, Siriopol D, Kuwabara M, et al. (October 2018). "Acute effects of salt on blood pressure are mediated by serum osmolality". Journal of Clinical Hypertension. 20 (10): 1447–1454. doi:10.1111/jch.13374. PMC 8030773. PMID 30232829.
- ^ Titze J, Luft FC (June 2017). "Speculations on salt and the genesis of arterial hypertension". Kidney International. 91 (6): 1324–1335. doi:10.1016/j.kint.2017.02.034. PMID 28501304.
- ^ Lee AJ (December 1997). "The role of rheological and haemostatic factors in hypertension". Journal of Human Hypertension. 11 (12): 767–776. doi:10.1038/sj.jhh.1000556. PMID 9468002.
- ^ Coffman JD (December 1988). "Pathophysiology of obstructive arterial disease". Herz. 13 (6): 343–350. PMID 3061915.
- ^ Korner PI, Angus JA (1992). "Structural determinants of vascular resistance properties in hypertension. Haemodynamic and model analysis". Journal of Vascular Research. 29 (4): 293–312. doi:10.1159/000158945. PMID 1391553.
- ^ Mulvany MJ (January 2012). "Small artery remodelling in hypertension". Basic & Clinical Pharmacology & Toxicology. 110 (1): 49–55. doi:10.1111/j.1742-7843.2011.00758.x. PMID 21733124.
- ^ de Moraes R, Tibirica E (2017). "Early Functional and Structural Microvascular Changes in Hypertension Related to Aging". Current Hypertension Reviews. 13 (1): 24–32. doi:10.2174/1573402113666170413095508. PMID 28412915.
- ^ Guyton AC, Coleman TG, Cowley AV, Scheel KW, Manning RD, Norman RA (May 1972). "Arterial pressure regulation. Overriding dominance of the kidneys in long-term regulation and in hypertension". The American Journal of Medicine. 52 (5): 584–594. doi:10.1016/0002-9343(72)90050-2. PMID 4337474.
- ^ a b Klabunde RE (2007). "Cardiovascular Physiology Concepts – Pulse Pressure". Archived from the original on 2009-10-18. Retrieved 2008-10-02.
- ^ Liaw SY, Scherpbier A, Klainin-Yobas P, Rethans JJ (September 2011). "A review of educational strategies to improve nurses' roles in recognizing and responding to deteriorating patients". International Nursing Review. 58 (3): 296–303. doi:10.1111/j.1466-7657.2011.00915.x. PMID 21848774.
- ^ Blacher J, Staessen JA, Girerd X, Gasowski J, Thijs L, Liu L, et al. (April 2000). "Pulse pressure not mean pressure determines cardiovascular risk in older hypertensive patients". Archives of Internal Medicine. 160 (8): 1085–1089. doi:10.1001/archinte.160.8.1085. PMID 10789600.
- ^ Cushman, William C.; Materson, Barry J.; Williams, David W.; Reda, Domenic J. (1 Oct 2001). "Pulse Pressure Changes With Six Classes of Antihypertensive Agents in a Randomized, Controlled Trial". Hypertension. 38 (4): 953–957. doi:10.1161/hy1001.096212. PMID 11641316.
- ^ Khilnani P, Singhi S, Lodha R, Santhanam I, Sachdev A, Chugh K, Jaishree M, Ranjit S, Ramachandran B, Ali U, Udani S, Uttam R, Deopujari S (January 2010). "Pediatric Sepsis Guidelines: Summary for resource-limited countries". Indian J Crit Care Med. 14 (1): 41–52. doi:10.4103/0972-5229.63029. PMC 2888329. PMID 20606908.
- ^ Al-Khalisy H, Nikiforov I, Jhajj M, Kodali N, Cheriyath P (11 December 2015). "A widened pulse pressure: a potential valuable prognostic indicator of mortality in patients with sepsis. J Community Hosp Intern Med Perspect". J Community Hosp Intern Med Perspect. 5 (6) 29426. doi:10.3402/jchimp.v5.29426. PMC 4677588. PMID 26653692.
- ^ Granger JP, Hall JE (2007). "Role of the Kidney in Hypertension". Comprehensive Hypertension. Elsevier. pp. 241–263. doi:10.1016/b978-0-323-03961-1.50026-x. ISBN 978-0-323-03961-1.
- ^ a b Klabunde RE (2007). "Cardiovascular Physiology Concepts – Mean Arterial Pressure". Archived from the original on 2009-10-02. Retrieved 2008-09-29.
- ^ Bos WJ, Verrij E, Vincent HH, Westerhof BE, Parati G, van Montfrans GA (April 2007). "How to assess mean blood pressure properly at the brachial artery level". Journal of Hypertension. 25 (4): 751–755. doi:10.1097/HJH.0b013e32803fb621. PMID 17351365. S2CID 23155959.
- ^ Meaney E, Alva F, Moguel R, Meaney A, Alva J, Webel R (July 2000). "Formula and nomogram for the sphygmomanometric calculation of the mean arterial pressure". Heart. 84 (1): 64. doi:10.1136/heart.84.1.64. PMC 1729401. PMID 10862592.
- ^ Klabunde RE (2007). "Cardiovascular Physiology Concepts – Arterial Baroreceptors". Archived from the original on 2009-12-23. Retrieved 2008-09-09.
- ^ Fountain J, Lappin SL (January 2022). "Physiology, Renin Angiotensin System.". StatPearls. Treasure Island, FL: StatPearls Publishing. PMID 29261862. Archived from the original on 29 April 2019. Retrieved 18 November 2022.
- ^ Feldman RD (January 2014). "Aldosterone and blood pressure regulation: recent milestones on the long and winding road from electrocortin to KCNJ5, GPER, and beyond". Hypertension. 63 (1): 19–21. doi:10.1161/HYPERTENSIONAHA.113.01251. PMID 24191283.
- ^ Forouzanfar M, Dajani HR, Groza VZ, Bolic M, Rajan S, Batkin I (2015-01-01). "Oscillometric Blood Pressure Estimation: Past, Present, and Future". IEEE Reviews in Biomedical Engineering. 8: 44–63. Bibcode:2015IRBE....8...44F. doi:10.1109/RBME.2015.2434215. PMID 25993705. S2CID 8940215.
- ^ "Apparatus and method for measuring blood pressure". Archived from the original on 2022-10-26. Retrieved 2019-01-12 – via Google patents.
- ^ Chandrasekhar A, Kim CS, Naji M, Natarajan K, Hahn JO, Mukkamala R (March 2018). "Smartphone-based blood pressure monitoring via the oscillometric finger-pressing method". Science Translational Medicine. 10 (431) eaap8674. doi:10.1126/scitranslmed.aap8674. PMC 6039119. PMID 29515001.
- ^ Solà J, Delgado-Gonzalo R (2019). The Handbook of Cuffless Blood Pressure Monitoring. Springer International Publishing. ISBN 978-3-030-24701-0. Archived from the original on 2021-06-17. Retrieved 2020-01-29.
- ^ Sola J, Bertschi M, Krauss J (September 2018). "Measuring Pressure: Introducing oBPM, the Optical Revolution for Blood Pressure Monitoring". IEEE Pulse. 9 (5): 31–33. Bibcode:2018IPuls...9e..31S. doi:10.1109/MPUL.2018.2856960. PMID 30273141. S2CID 52893219.
- ^ Foti KE, Appel LJ, Matsushita K, Coresh J, Alexander GC, Selvin E (May 2021). "Digit Preference in Office Blood Pressure Measurements, United States 2015-2019". American Journal of Hypertension. 34 (5): 521–530. doi:10.1093/ajh/hpaa196. PMC 8628654. PMID 33246327.
- ^ Prothero JW (2015-10-22). The Design of Mammals: A Scaling Approach. Cambridge: Cambridge University Press. ISBN 978-1-107-11047-2. OCLC 907295832.
- ^ Brøndum E, Hasenkam JM, Secher NH, Bertelsen MF, Grøndahl C, Petersen KK, et al. (October 2009). "Jugular venous pooling during lowering of the head affects blood pressure of the anesthetized giraffe". American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 297 (4): R1058 – R1065. doi:10.1152/ajpregu.90804.2008. PMID 19657096.
- ^ Seymour RS, Lillywhite HB (December 1976). "Blood pressure in snakes from different habitats". Nature. 264 (5587): 664–666. Bibcode:1976Natur.264..664S. doi:10.1038/264664a0. PMID 1004612. S2CID 555576.
- ^ Nasoori A, Taghipour A, Shahbazzadeh D, Aminirissehei A, Moghaddam S (September 2014). "Heart place and tail length evaluation in Naja oxiana, Macrovipera lebetina, and Montivipera latifii". Asian Pacific Journal of Tropical Medicine. 7S1: S137 – S142. doi:10.1016/s1995-7645(14)60220-0. PMID 25312108.
- ^ Seymour RS (1987). "Scaling of cardiovascular physiology in snakes". American Zoologist. 27 (1): 97–109. doi:10.1093/icb/27.1.97. ISSN 0003-1569.
- ^ a b Gross DR (2009). Animal Models in Cardiovascular Research (3rd ed.). Dordrecht: Springer. p. 5. ISBN 978-0-387-95962-7. OCLC 432709394.
- ^ Brown S, Atkins C, Bagley R, Carr A, Cowgill L, Davidson M, et al. (2007). "Guidelines for the identification, evaluation, and management of systemic hypertension in dogs and cats". Journal of Veterinary Internal Medicine. 21 (3): 542–558. doi:10.1111/j.1939-1676.2007.tb03005.x. PMID 17552466.
- ^ Lerman LO, Chade AR, Sica V, Napoli C (September 2005). "Animal models of hypertension: an overview". The Journal of Laboratory and Clinical Medicine. 146 (3): 160–173. doi:10.1016/j.lab.2005.05.005. PMID 16131455.
- ^ "AKC Canine Health Foundation | Hypertension in Dogs". www.akcchf.org. Archived from the original on 2022-10-03. Retrieved 2022-10-03.
- ^ Acierno MJ, Brown S, Coleman AE, Jepson RE, Papich M, Stepien RL, Syme HM (November 2018). "ACVIM consensus statement: Guidelines for the identification, evaluation, and management of systemic hypertension in dogs and cats". Journal of Veterinary Internal Medicine. 32 (6): 1803–1822. doi:10.1111/jvim.15331. PMC 6271319. PMID 30353952.
External links
[edit] Media related to Blood pressure at Wikimedia Commons
Media related to Blood pressure at Wikimedia Commons
Blood pressure
View on GrokipediaFundamentals
Definition and Types
Blood pressure is the force exerted by circulating blood against the walls of arteries as the heart pumps it through the body, primarily reflecting the hydrostatic pressure within the major arterial system. This pressure is measured in millimeters of mercury (mmHg) and arises mainly from the mechanical action of the heart ejecting blood into the compliant arterial tree.[4] The hydrostatic component represents the physical force generated by cardiac output and vascular resistance, driving blood flow throughout the circulation.[2] Additionally, osmotic pressure—particularly the oncotic pressure exerted by plasma proteins like albumin—contributes by counteracting fluid leakage from vessels, thereby helping to sustain intravascular volume and indirectly supporting overall blood pressure levels.[5] The primary types of arterial blood pressure are systolic, diastolic, and mean arterial pressure, each characterizing different phases of the cardiac cycle. Systolic pressure denotes the maximum pressure attained in large arteries during ventricular contraction (systole), when blood is forcefully ejected from the heart.[4] Diastolic pressure, in contrast, is the minimum pressure occurring during ventricular relaxation (diastole), when the heart refills between beats.[4] Mean arterial pressure (MAP) provides an average value over the entire cardiac cycle, calculated as the integral of arterial pressure across systole and diastole, and serves as a key indicator of perfusion adequacy to organs.[6] Blood pressure is distinguished into systemic and pulmonary types based on the circulatory pathway. Systemic blood pressure refers to the higher pressures in the arteries distributing oxygenated blood to the body's tissues via the left ventricle.[2] Pulmonary blood pressure, driven by the right ventricle, involves lower pressures in the pulmonary arteries that carry deoxygenated blood to the lungs for gas exchange, typically about one-sixth of systemic levels to accommodate the lung's delicate vasculature.[7] The concept of blood pressure measurement originated in the early 18th century, with English clergyman and scientist Stephen Hales conducting the first direct recording in 1733 by inserting a brass tube into a horse's carotid artery and observing the blood's rise in a vertical glass tube connected to it.[8]Basic Physiology
Blood pressure is fundamentally generated and maintained by the cardiovascular system, which consists of the heart acting as the central pump and the network of blood vessels serving as conduits for blood flow. The heart propels blood through rhythmic contractions, producing a cardiac output that represents the volume of blood ejected per minute, typically around 5 liters in a resting adult. Arteries carry oxygenated blood away from the heart under high pressure, veins return deoxygenated blood to the heart under lower pressure, and capillaries facilitate exchange of nutrients, gases, and wastes between blood and tissues. This closed system ensures continuous circulation, with blood pressure arising from the force exerted by the heart against vascular resistance.[9] Hemodynamics describes the physical principles governing blood flow and pressure, analogous to Ohm's law in electricity, where mean arterial pressure (MAP) equals cardiac output (CO) multiplied by total peripheral resistance (TPR): MAP = CO × TPR. Cardiac output is the product of heart rate and stroke volume, while TPR arises primarily from the arterioles due to their narrow lumens and smooth muscle control, though the entire vascular tree contributes. This relationship highlights how pressure gradients drive flow from high-pressure regions (aorta) to low-pressure areas (right atrium), with blood viscosity and vessel geometry influencing overall dynamics.[9][10] Blood flow in vessels is predominantly laminar, characterized by smooth, layered movement parallel to the vessel walls, which minimizes energy loss and shear stress on endothelial cells. In contrast, turbulent flow involves chaotic eddies and mixing, occurring rarely in normal physiology such as during peak systole in the aorta or at vessel bifurcations, and is quantified by the Reynolds number (Re = ρVD/μ, where ρ is blood density, V is velocity, D is diameter, and μ is viscosity); laminar flow prevails when Re < 2000. Resistance to flow in individual vessels follows Poiseuille's law for laminar conditions in rigid tubes: , where η is blood viscosity, L is vessel length, and r is radius, emphasizing that resistance is inversely proportional to the fourth power of the radius—thus, small changes in vessel diameter profoundly affect pressure.[10][9] Arteries exhibit vascular compliance and elasticity due to their layered structure, including elastin fibers in the media, allowing them to distend during systole and recoil during diastole, thereby buffering the intermittent pressure pulses from the heart to maintain steady downstream flow. This Windkessel effect transforms pulsatile cardiac ejection into more continuous perfusion of capillaries and tissues, with arterial compliance defined as the change in volume per change in pressure (C = ΔV/ΔP). Large elastic arteries like the aorta are particularly adept at this buffering, absorbing up to 50% of stroke volume during contraction.[9][11]Pressure Parameters
Arterial Pressure
Systemic arterial pressure is the force exerted by circulating blood on the walls of the arteries in the systemic circulation, which delivers oxygenated blood from the left ventricle to the body's tissues. It consists of two primary components: systolic pressure, the maximum pressure occurring during ventricular contraction, and diastolic pressure, the minimum pressure during ventricular relaxation. In healthy adults, systolic arterial pressure typically ranges from 90 to 120 mmHg, while diastolic pressure ranges from 60 to 80 mmHg, with values below 90/60 mmHg indicating hypotension and above 120/80 mmHg suggesting elevated pressure.[12][13][14] Aortic pressure, measured in the proximal aorta near the heart, represents the central arterial pressure where blood is ejected directly from the left ventricle. Due to this proximity, the mean arterial pressure is highest in the aorta, calculated as approximately one-third systolic plus two-thirds diastolic, and it declines gradually toward the peripheral arteries owing to frictional losses and increasing vascular resistance along the arterial tree. Although systolic pressure can exhibit amplification in peripheral sites due to wave reflections, the overall pressure profile ensures adequate perfusion while minimizing cardiac workload.[15][16] Pulse pressure, defined as the difference between systolic and diastolic pressures, normally ranges from 30 to 50 mmHg in adults and reflects arterial compliance and stroke volume. It is computed simply as systolic pressure minus diastolic pressure, providing insight into vascular health; for instance, a pulse pressure of 40 mmHg is typical for a reading of 120/80 mmHg. A widened pulse pressure exceeding 60 mmHg often signifies arterial stiffness, where reduced elasticity leads to higher systolic peaks and lower diastolic troughs, increasing cardiovascular risk.[17][16][18]Venous and Pulmonary Pressure
Venous pressure in the systemic circulation is significantly lower than arterial pressure, reflecting the role of veins as capacitance vessels that store and return blood to the heart. Central venous pressure (CVP), measured at the junction of the superior vena cava and right atrium, typically ranges from 0 to 8 mmHg in healthy individuals.[19] This low pressure serves as the primary determinant of venous return, acting as the back pressure that opposes the flow of blood from peripheral veins toward the heart; higher CVP reduces venous return, while lower values facilitate it.[20] In the pulmonary circulation, pressures are also low to accommodate the thin-walled alveoli and minimize the work of breathing. Normal pulmonary artery pressure includes a systolic value of 15 to 30 mmHg, diastolic of 4 to 12 mmHg, and mean of 9 to 18 mmHg.[21] These values are substantially lower than systemic arterial pressures due to the pulmonary vasculature's high compliance and low resistance, which prevent excessive strain on the lung tissue during gas exchange.[22] Mean systemic filling pressure (MSFP) represents the equilibrium pressure throughout the systemic circulation in the absence of blood flow, such as during cardiac arrest, and is approximately 7 mmHg in normovolemic humans.[23] This pressure arises primarily from the elastic recoil of the vascular walls and the volume of blood within the system, serving as a key indicator of circulatory filling status and the driving force for venous return under static conditions.[24] Compared to the arterial system, both venous and pulmonary circulations exhibit lower resistance and higher volume compliance, allowing them to handle larger blood volumes with minimal pressure changes. The venous system's compliance is about 30 times greater than that of arteries, enabling it to store up to 60-70% of total blood volume while maintaining low pressures.[25] Similarly, the pulmonary circuit's low resistance (about one-tenth of systemic) ensures efficient perfusion without compromising oxygenation.[26]Specialized Pressures
In the fetal circulation, systemic arterial blood pressure at term averages approximately 70/45 mmHg, with a mean of 55 mmHg, reflecting the underdeveloped vascular system and reliance on placental gas exchange.[27] Prior to birth, pulmonary vascular resistance remains elevated—approximately 8 to 10 times higher than systemic vascular resistance—due to factors such as fluid-filled lungs, low oxygen tension, and vasoconstrictive mediators, which minimizes pulmonary blood flow and directs most right ventricular output through the ductus arteriosus to the systemic circulation.[28] This high resistance ensures oxygenation via the placenta but requires a dramatic drop in pulmonary pressure postnatally for effective lung perfusion.[29] Portal venous pressure, which governs blood flow through the liver's portal system, normally ranges from 5 to 10 mmHg in healthy adults, exceeding inferior vena caval pressure by about 4 to 5 mmHg to facilitate nutrient-rich blood delivery from the splanchnic organs.[30] This low-pressure system supports sinusoidal filtration without excessive resistance, but elevations beyond 10 mmHg indicate portal hypertension, often linked to liver cirrhosis or thrombosis.[31] Intracranial pressure (ICP) interacts closely with systemic blood pressure to determine cerebral perfusion pressure (CPP), calculated as CPP = mean arterial pressure (MAP) minus ICP, ensuring adequate oxygen delivery to brain tissue.[32] In adults, normal ICP ranges from 7 to 15 mmHg in the supine position, maintained by cerebrospinal fluid dynamics and cerebral blood volume; when ICP rises above 20 mmHg, it can compress cerebral vessels, reducing CPP and risking ischemia unless compensated by elevated MAP.[33] This relationship underscores the brain's autoregulation, where CPP is typically kept between 60 and 150 mmHg to match metabolic demands.[32] In the microcirculation, capillary hydrostatic pressure drives fluid exchange across vessel walls and varies along the capillary length, typically ranging from 10 to 25 mmHg at heart level, with higher values (around 30-35 mmHg) at the arterial end and lower (10-15 mmHg) at the venous end.[34] This gradient, influenced by precapillary sphincter tone and interstitial forces, balances filtration and reabsorption per Starling's principle, preventing edema while nourishing tissues; for instance, in skin capillaries, pressures measured at the apex fall within 10.5 to 22.5 mmHg under normal conditions.[35]Normal Values and Variations
Norms by Age and Population
Blood pressure norms vary significantly across life stages, reflecting developmental changes in cardiovascular physiology. In the fetal period, blood pressure is low, with mean arterial pressure around 30-50 mmHg near term, but it rises rapidly postnatally as the circulatory system adapts to independent oxygenation. For newborns, average systolic blood pressure is approximately 60-80 mmHg, with values typically ranging from 60-90 mmHg in the first days of life, increasing to 70-100 mmHg by one month. Diastolic pressures follow a similar pattern, averaging 40-50 mmHg initially. These early norms are established through population-based studies of term infants, emphasizing the need for age-specific reference ranges to avoid misdiagnosis.[36] During childhood, blood pressure increases gradually with growth and body size, necessitating norms adjusted for age, sex, and height percentiles rather than fixed values. For example, in a 10-year-old child, normal systolic blood pressure falls between 100-120 mmHg and diastolic between 60-80 mmHg at the 50th-90th percentiles, with hypertension defined above the 95th percentile (e.g., >122/78 mmHg for boys of average height).[37] These percentile-based standards, derived from large cohorts like the National Health and Nutrition Examination Survey, account for the linear upward trend in both systolic and diastolic pressures from infancy through adolescence, reaching near-adult levels by late teens.[38] In adulthood, normal blood pressure is generally defined as less than 120/80 mmHg for both sexes. According to ACC/AHA guidelines, blood pressure categories are classified as normal (systolic <120 mmHg and diastolic <80 mmHg), elevated (systolic 120-129 mmHg and diastolic <80 mmHg), stage 1 hypertension (systolic 130-139 mmHg or diastolic 80-89 mmHg), and stage 2 hypertension (systolic ≥140 mmHg or diastolic ≥90 mmHg).[39] Examples include normal readings such as 115/75 mmHg or 118/76 mmHg, elevated at 125/78 mmHg, stage 1 hypertension at 135/85 mmHg, and stage 2 hypertension at 145/92 mmHg. For example, a blood pressure reading of 122/73 mmHg is classified as elevated (systolic 120-129 mmHg and diastolic <80 mmHg), not normal (<120/80 mmHg), and not hypertensive. It indicates a need for monitoring and lifestyle adjustments to prevent progression to hypertension. Conversely, a blood pressure reading of 110/70 mmHg is classified as normal (healthy) for a 59-year-old woman, according to American Heart Association and Mayo Clinic guidelines. Normal is defined as less than 120 mmHg systolic and less than 80 mmHg diastolic. Classification categories do not differ by age or gender for adults. This reading is below the average for women aged 40-59 (around 122/74 mmHg) but remains within the healthy range.[13][40][41] A concurrent pulse rate of 64 bpm is normal and good for adults, within the typical resting heart rate range of 60-100 bpm. In student physiology labs, resting readings for healthy young adults are commonly in the normal range, such as 110–130 mmHg systolic over 70–85 mmHg diastolic. The Japanese Society of Hypertension guidelines define normal blood pressure as systolic <130 mmHg and diastolic <85 mmHg, with optimal <120/80 mmHg.[42] Though mean values rise with age, peaking in middle age before a potential decline in extreme old age due to reduced vascular compliance. For adults aged 18-39, average systolic pressure is around 115-120 mmHg and diastolic 75-80 mmHg; by ages 60-79, systolic averages 130-140 mmHg while diastolic stabilizes or slightly decreases to 70-80 mmHg.[43] Women often exhibit lower pressures than men until menopause, after which differences narrow. Ethnic variations are notable, with individuals of African descent showing higher average systolic pressures (e.g., 5-10 mmHg above non-Hispanic whites) and earlier onset of elevated readings across adulthood.[44] In the elderly (aged 80+), average systolic values often reach 130-150 mmHg due to reduced vascular compliance, but normal blood pressure is still defined as less than 120/80 mmHg, with hypertension thresholds applying at 130/80 mmHg.[45] Recent ACC/AHA guidelines, updated in 2025, maintain the threshold for hypertension onset at 130/80 mmHg for adults, including older populations, while emphasizing individualized targets below 130/80 mmHg for most to reduce cardiovascular events, with considerations for frailty and limited life expectancy in those over 80 to ensure benefits outweigh harms. The 2025 guidelines incorporate the PREVENT equations for risk stratification to inform treatment initiation in those with stage 1 hypertension.[39] These standards, informed by trials like SPRINT, underscore the importance of accurate measurement to interpret norms reliably.[46]| Age Group | Typical Average Systolic (mmHg) | Typical Average Diastolic (mmHg) | Key Notes |
|---|---|---|---|
| Newborn (0-1 day) | 60-90 | 40-50 | Rapid postnatal rise; averages 64/41. Normal BP defined as <120/<80 mmHg for adults; age-specific for children. |
| Child (10 years) | 100-120 | 60-80 | 50th-90th percentiles; height-adjusted.[37] |
| Adult (18-39 years) | <120 | <80 | Lower in women pre-menopause. Normal BP defined as <120/<80 mmHg.[43] |
| Elderly (60+ years) | 120-140 | 70-80 | Systolic peaks mid-life, higher in African descent. Normal BP defined as <120/<80 mmHg.[44][45] |







