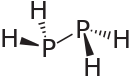Diphosphane
View on Wikipedia
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Diphosphane
| |||
| Systematic IUPAC name
Diphosphane (substitutive) Tetrahydridodiphosphorus(P—P) (additive) | |||
| Other names
Diphosphine
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| P2H4 | |||
| Melting point | −99 °C (−146 °F; 174 K) | ||
| Boiling point | 63.5 °C (146.3 °F; 336.6 K) (Extrapolated, decomposes) | ||
| Related compounds | |||
Other anions
|
ammonia hydrazine triazane | ||
Other cations
|
diphosphines | ||
Related Binary Phosphorus halides
|
diphosphorus tetrafluoride diphosphorus tetrachloride diphosphorus tetrabromide diphosphorus tetraiodide | ||
Related compounds
|
phosphane triphosphane diphosphene diphosphenes | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Diphosphane, or diphosphine, is an inorganic compound with the chemical formula P2H4. This colourless liquid is one of several binary phosphorus hydrides. It is the impurity that typically causes samples of phosphine to ignite in air.
Properties, preparation, reactions
[edit]Diphosphane adopts the gauche conformation (like hydrazine, less symmetrical than shown in the image) with a P−P distance of 2.219 angstroms. It is nonbasic, unstable at room temperature, and spontaneously flammable in air. It is only poorly soluble in water but dissolves in organic solvents. Its 1H NMR spectrum consists of 32 lines resulting from an A2XX'A'2 splitting system.[1]
Diphosphane is produced by the hydrolysis of calcium monophosphide, which can be described as the Ca2+ derivative of P4−2. According to an optimized procedure, hydrolysis of 400 g of CaP at −30 °C gives about 20 g of product, slightly contaminated with phosphine.[citation needed]
Reaction of diphosphane with butyllithium affords a variety of condensed polyphosphine compounds.
Organic diphosphanes
[edit]A variety of organic derivatives of diphosphane are known, but asymmetric diphosphanes are only stable at cryogenic temperatures. Otherwise, the substituents facily redistribute on the phosphorus centers to give a mixture of products. On the other hand, there appears to be a substantial barrier to chiral inversion.[2]
The central bond is weak, and easily adds substituents.[3]
The simplest synthesis method heats a phosphorus halide and a phosphane:
- Ph2PCl + HPPh2 → Ph2P−PPh2 + HCl↑
Alkali metals can replace the hydrogen in that reaction (i.e., a dialkylphosphide), and in some rare cases a dialkylamine can replace the halide. Symmetric diphosphanes are easily prepared by reductive coupling, e.g. tetraphenyldiphosphine from chlorodiphenylphosphine:
- 2 ClPPh2 + 2 Na → Ph2P−PPh2 + 2 NaCl
Ultraviolet radiation decomposes mercury(II) dialkylphosphides to the metal and a dialkylphosphane.[4]
The methyl compound P2Me4 is prepared by the reduction of Me2P(S)−P(S)Me2, which is produced by methylation of thiophosphoryl chloride with methylmagnesium bromide.[5]
See also
[edit]References
[edit]- ^ Marianne Baudler, Klaus Glinka (1993). "Monocyclic and polycyclic phosphines". Chem. Rev. 93 (4): 1623–1667. doi:10.1021/cr00020a010.
- ^ Phosphorus: Chemistry, Biochemistry and Technology, Sixth Edition, 2013, D.E.C. Corbridge, CRC Pres, Taylor Francis Group, ISBN 978-1-4398-4088-7. pp. 421-422.
- ^ Corbridge 2013, p. 422.
- ^ Corbridge 2013, pp. 421–422.
- ^ Butter, S. A.; Chatt, J. (1974). "Ethylenebis(dimethylphosphine)". Inorganic Syntheses. Vol. 15. p. 185. doi:10.1002/9780470132463.ch41. ISBN 9780470132463.
{{cite book}}:|journal=ignored (help)