Recent from talks
Nothing was collected or created yet.
Ethanolamine
View on Wikipedia | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Aminoethan-1-ol[1] | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.004.986 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H7NO | |
| Molar mass | 61.084 g·mol−1 |
| Appearance | Viscous colourless liquid |
| Odor | Unpleasant ammonia-like odour |
| Density | 1.0117 g/cm3 |
| Melting point | 10.3 °C (50.5 °F; 283.4 K) |
| Boiling point | 170 °C (338 °F; 443 K) |
| Miscible | |
| Vapor pressure | 64 Pa (20 °C)[2] |
| Acidity (pKa) | 9.50[3] |
Refractive index (nD)
|
1.4539 (20 °C)[4] |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H302, H312, H314, H332, H335, H412[5] | |
| P261, P273, P303+P361+P353, P305+P351+P338[5] | |
| NFPA 704 (fire diamond) | |
| Flash point | 85 °C (185 °F; 358 K) (closed cup) |
| 410 °C (770 °F; 683 K) | |
| Explosive limits | 5.5–17% |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
|
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA: 3 ppm (6 mg/m3)[6] |
REL (Recommended)
|
|
IDLH (Immediate danger)
|
30 ppm[6] |
| Safety data sheet (SDS) | Sigma[5] |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
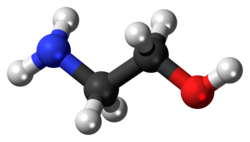
Ethanolamine (2-aminoethanol, monoethanolamine, ETA, or MEA) is a naturally occurring organic chemical compound with the formula HOCH
2CH
2NH
2 or C
2H
7NO.[8] The molecule is bifunctional, containing both a primary amine and a primary alcohol. Ethanolamine is a colorless, viscous liquid with an odor reminiscent of ammonia.[9]
Ethanolamine is commonly called monoethanolamine or MEA in order to be distinguished from diethanolamine (DEA) and triethanolamine (TEOA). The ethanolamines comprise a group of amino alcohols. A class of antihistamines is identified as ethanolamines, which includes carbinoxamine, clemastine, dimenhydrinate, chlorphenoxamine, diphenhydramine and doxylamine.[10]
History
[edit]Ethanolamines, or in particular, their salts, were discovered by Charles Adolphe Wurtz in 1860[11] by heating 2-chloroethanol with ammonia solution while studying derivatives of ethylene oxide he discovered a year earlier.[12] He wasn't able to separate the salts or isolate any free bases.
In 1897 Ludwig Knorr developed the modern industrial route (see below) and separated the products, including MEA, by fractional distillation, for the first time studying their properties.[13]
None of the ethanolamines were of any commercial importance until after the WWII industrial production of ethylene oxide took off.[12]
Occurrence in nature
[edit]MEA molecules are a component in the formation of cellular membranes and are thus a molecular building block for life. Ethanolamine is the second-most-abundant head group for phospholipids, substances found in biological membranes (particularly those of prokaryotes); e.g., phosphatidylethanolamine. It is also used in messenger molecules such as palmitoylethanolamide, which has an effect on CB1 receptors.[14]
MEA was thought to exist only on Earth and on certain asteroids, but in 2021 evidence was found that these molecules exist in interstellar space.[15]
Ethanolamine is biosynthesized by decarboxylation of serine:[16]
- HOCH
2CH(CO
2H)NH
2 → HOCH
2CH
2NH
2 + CO2
Derivatives of ethanolamine are widespread in nature; e.g., lipids, as precursor of a variety of N-acylethanolamines (NAEs), that modulate several animal and plant physiological processes such as seed germination, plant–pathogen interactions, chloroplast development and flowering,[17] as well as precursor, combined with arachidonic acid C
20H
32O
2 20:4, ω-6), to form the endocannabinoid anandamide (AEA: C
22H
37NO
2; 20:4, ω-6).[18]
MEA is biodegraded by ethanolamine ammonia-lyase, a B12-dependent enzyme. It is converted to acetaldehyde and ammonia via initial H-atom abstraction.[19]
- H2NCH2CH2OH → NH3 + CH3CHO
Industrial production
[edit]Monoethanolamine is produced by treating ethylene oxide with aqueous ammonia; the reaction also produces diethanolamine and triethanolamine. The ratio of the products can be controlled by the stoichiometry of the reactants.[20]
Applications
[edit]MEA is used as feedstock in the production of detergents, emulsifiers, polishes, pharmaceuticals, corrosion inhibitors, and chemical intermediates.[9]
For example, reacting ethanolamine with ammonia gives ethylenediamine, a precursor of the commonly used chelating agent, EDTA.[20]
Gas stream scrubbing
[edit]Monoethanolamines can scrub combusted-coal, combusted-methane and combusted-biogas flue emissions of carbon dioxide (CO2) very efficiently. MEA carbon dioxide scrubbing is also used to regenerate the air on submarines.
Solutions of MEA in water are used as a gas stream scrubbing liquid in amine treaters.[21] For example, aqueous MEA is used to remove carbon dioxide (CO2) and hydrogen sulfide (H2S) from various gas streams; e.g., flue gas and sour natural gas.[22] The MEA ionizes dissolved acidic compounds, making them polar and considerably more soluble.
MEA scrubbing solutions can be recycled through a regeneration unit. When heated, MEA, being a rather weak base, will release dissolved H2S or CO2 gas resulting in a pure MEA solution.[20][23]
Other uses
[edit]In pharmaceutical formulations, MEA is used primarily for buffering or preparation of emulsions. MEA can be used as pH regulator in cosmetics.[24]
It is an injectable sclerosant as a treatment option of symptomatic hemorrhoids. 2–5 ml of ethanolamine oleate can be injected into the mucosa just above the hemorrhoids to cause ulceration and mucosal fixation thus preventing hemorrhoids from descending out of the anal canal.
It is also an ingredient in cleaning fluid for automobile windshields.[25]
pH-control amine
[edit]Ethanolamine is often used for alkalinization of water in steam cycles of power plants, including nuclear power plants with pressurized water reactors. This alkalinization is performed to control corrosion of metal components. ETA (or sometimes a similar organic amine; e.g., morpholine) is selected because it does not accumulate in steam generators (boilers) and crevices due to its volatility, but rather distributes relatively uniformly throughout the entire steam cycle. In such application, ETA is a key ingredient of so-called "all-volatile treatment" of water (AVT).[citation needed]
Reactions
[edit]Upon reaction with carbon dioxide, 2 equivalents of ethanolamine react through the intermediacy of carbonic acid to form a carbamate salt,[26] which when heated usually reforms back to ethanolamine and carbon dioxide but occasionally can also cyclizate to 2-oxazolidone, generating amine gas treatment wastes.[27]
References
[edit]- ^ a b Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. pp. 649, 717. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
For example, the name 'ethanolamine', which is still widely used, is badly constructed because of the presence of two suffixes; it is not an alternative to the preferred IUPAC name, '2-aminoethan-1-ol'.
- ^ "Ethanolamine MSDS" (PDF). Acros Organics. Archived from the original (PDF) on 2011-07-15.
- ^ Hall, H.K. (1957). "Correlation of the Base Strengths of Amines". J. Am. Chem. Soc. 79 (20): 5441–4. Bibcode:1957JAChS..79.5441H. doi:10.1021/ja01577a030.
- ^ Reitmeier, R.E.; Sivertz, V.; Tartar, H.V. (1940). "Some Properties of Monoethanolamine and its Aqueous Solutions". Journal of the American Chemical Society. 62 (8): 1943–44. Bibcode:1940JAChS..62.1943R. doi:10.1021/ja01865a009.
- ^ a b c Sigma-Aldrich Co., Ethanolamine. Retrieved on 2018-05-24.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0256". National Institute for Occupational Safety and Health (NIOSH).
- ^ "Ethanolamine". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ "National Library of Medicine. PubChem. Ethanolomine". NIH, National Library of Medicine. Retrieved September 5, 2021.
- ^ a b Martin Ernst; Johann-Peter Melder; Franz Ingo Berger; Christian Koch (2022). "Ethanolamines and Propanolamines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a10_001.pub2. ISBN 978-3-527-30673-2.
- ^ Cough, Cold, and Allergy Preparation Toxicity at eMedicine
- ^ Wurtz, A. (1860). "Synthese sauerstoffhaltiger Basen". Justus Liebigs Annalen der Chemie. 114 (1): 51–54. doi:10.1002/jlac.18601140106. ISSN 0075-4617.
- ^ a b U.S. patent 9227912B2
- ^ Knorr, Ludwig (1897). "Ueber den Amidoäthylalkohol". Berichte der Deutschen Chemischen Gesellschaft. 30 (1): 909–915. doi:10.1002/cber.189703001178. ISSN 0365-9496.
- ^ Calignano, A; La Rana, G; Piomelli, D (2001). "Antinociceptive activity of the endogenous fatty acid amide, palmitylethanolamide". European Journal of Pharmacology. 419 (2–3): 191–8. doi:10.1016/S0014-2999(01)00988-8. PMID 11426841.
- ^ "First evidence of cell membrane molecules in space". Astronomy Magazine. May 28, 2021. Retrieved September 4, 2021.
- ^ "Phosphatidylethanolamine and related lipids". AOCS. Archived from the original on 2012-08-21. Retrieved 2015-08-09.
- ^ Coutinho, Bruna G.; Mevers, Emily; Schaefer, Amy L.; Pelletier, Dale A.; Harwood, Caroline S.; Clardy, Jon; Greenberg, E. Peter (2018-09-25). "A plant-responsive bacterial-signaling system senses an ethanolamine derivative". Proceedings of the National Academy of Sciences of the United States of America. 115 (39): 9785–9790. Bibcode:2018PNAS..115.9785C. doi:10.1073/pnas.1809611115. ISSN 0027-8424. PMC 6166808. PMID 30190434.
- ^ Marzo, V. Di; Petrocellis, L. De; Sepe, N.; Buono, A. (1996-06-15). "Biosynthesis of anandamide and related acylethanolamides in mouse J774 macrophages and N18 neuroblastoma cells". Biochemical Journal. 316 (Pt 3): 977–84. doi:10.1042/bj3160977. PMC 1217444. PMID 8670178.
- ^ Shibata, Naoki; Tamagaki, Hiroko; Hieda, Naoki; Akita, Keita; Komori, Hirofumi; Shomura, Yasuhito; Terawaki, Shin-Ichi; Mori, Koichi; Yasuoka, Noritake; Higuchi, Yoshiki; Toraya, Tetsuo (2010). "Crystal Structures of Ethanolamine Ammonia-lyase Complexed with Coenzyme B12 Analogs and Substrates". Journal of Biological Chemistry. 285 (34): 26484–26493. doi:10.1074/jbc.M110.125112. PMC 2924083. PMID 20519496.
- ^ a b c Weissermel, Klaus; Arpe, Hans-Jürgen; Lindley, Charlet R.; Hawkins, Stephen (2003). "Chap. 7. Oxidation Products of Ethylene". Industrial Organic Chemistry. Wiley-VCH. pp. 159–161. ISBN 3-527-30578-5.
- ^ Chremos A, et al. (August 2015). "Modelling the phase and chemical equilibria of aqueous solutions of alkanolamines and carbon dioxide using the SAFT-γ SW group contribution approach". Fluid Phase Equilibria. 407: 280–297. doi:10.1016/j.fluid.2015.07.052. hdl:10044/1/25382.
- ^ Emergency and Continuous Exposure Guidance Levels for Selected Submarine Contaminants. 2007. doi:10.17226/11170. ISBN 978-0-309-09225-8.
- ^ "Ethanolamine". Occupational Safety & Health Administration. Archived from the original on 2013-05-03. Retrieved 2008-05-11.
- ^ Carrasco, F. (2009). "Ingredientes Cosméticos". Diccionario de Ingredientes Cosméticos 4ª Ed. www.imagenpersonal.net. p. 306. ISBN 978-84-613-4979-1.
- ^ Federal Motor Vehicle Safety Standards. U.S. Department of Transportation, National Highway Traffic Safety Administration. 1994. p. Part 571; S 108—PRE 128.
- ^ Lu, Yanyue; Liao, Anping; Yun, Zhuge; Liang, Yanqing; Yao, Qinmei (2014). "Absorption of Carbon Dioxide in Ethanolamine Solutions". Asian Journal of Chemistry. 26 (1): 39–42. doi:10.14233/ajchem.2014.15301.
- ^ Salim, S. R. S. (2021-03-01). "Treatment of amine wastes generated in industrial processes". IOP Conference Series: Materials Science and Engineering. 1092 (1) 012051. Bibcode:2021MS&E.1092a2051S. doi:10.1088/1757-899x/1092/1/012051. ISSN 1757-8981.
External links
[edit]Ethanolamine
View on GrokipediaProperties
Physical properties
Ethanolamine (C₂H₇NO) is a bifunctional organic compound with a molecular weight of 61.08 g/mol. It appears as a colorless, viscous liquid at room temperature, exhibiting an ammonia-like odor. This physical state influences its handling in industrial and laboratory settings, where it remains liquid over a moderate temperature range. Key physical characteristics include a melting point of 10.3 °C and a boiling point of 170.8 °C, indicating stability under ambient conditions but requiring caution during heating processes. The density is 1.012 g/cm³ at 20 °C, contributing to its syrupy consistency. Ethanolamine is fully miscible with water and ethanol, facilitating its use in aqueous solutions, while it shows limited solubility in organic solvents such as ether. Additional properties include a refractive index of 1.4538 at 20 °C, a flash point of 85 °C, and a vapor pressure of 0.4 mmHg at 20 °C, which inform safety protocols for storage and transport.[1] Its viscosity measures approximately 18.0 cP at 25 °C, and the specific heat capacity is about 3.20 J/g·K. Regarding acid-base behavior, the pKa of the amine group is 9.50, while that of the alcohol group is approximately 15.5.| Property | Value | Conditions |
|---|---|---|
| Molecular formula | C₂H₇NO | - |
| Molecular weight | 61.08 g/mol | - |
| Appearance | Colorless viscous liquid, ammonia-like odor | Room temperature |
| Melting point | 10.3 °C | - |
| Boiling point | 170.8 °C | 760 mmHg |
| Density | 1.012 g/cm³ | 20 °C |
| Refractive index | 1.4538 | 20 °C |
| Flash point | 85 °C | Closed cup |
| Vapor pressure | 0.4 mmHg | 20 °C |
| Viscosity | 18.0 cP | 25 °C |
| Specific heat capacity | 3.20 J/g·K | Liquid phase |
| pKa (amine) | 9.50 | 25 °C |
| pKa (alcohol) | ≈15.5 | - |
| Solubility in water | Fully miscible | - |
| Solubility in ethanol | Fully miscible | - |
| Solubility in ether | Slightly soluble | - |
Chemical properties
Ethanolamine is a bifunctional molecule possessing a primary amine (-NH₂) group and a primary alcohol (-OH) group attached to adjacent carbon atoms. This arrangement confers amphoteric character, enabling it to behave as a base via the amine (with the pKa of its conjugate acid at 9.5 at 25°C) and as a weak acid via the alcohol (pKa ≈ 15.5).[1][6] The molecule's ability to form extensive hydrogen bonds, both intramolecularly and intermolecularly, through its -NH₂ and -OH groups results in strong associations that elevate its boiling point to 170°C—significantly higher than expected for a compound of its 61 g/mol molecular weight.[1] Ethanolamine demonstrates good chemical stability under ambient conditions but is hygroscopic, readily absorbing moisture and carbon dioxide from air to form carbonates. It undergoes slow oxidative degradation in the presence of oxygen, yielding products such as glycolaldehyde and glyoxal. Thermal decomposition begins above approximately 200°C, producing hazardous gases including nitrogen oxides and carbon oxides.[7][8] Key spectroscopic properties reflect the contributions of its amine and alcohol functionalities. Infrared (IR) spectra exhibit broad overlapping stretches for O-H and N-H at 3200–3600 cm⁻¹, N-H bending near 1600 cm⁻¹, and C-O stretching around 1050–1100 cm⁻¹. In ¹H nuclear magnetic resonance (NMR), the CH₂NH₂ protons appear at 2.7–3.1 ppm and CH₂OH at 3.5–3.8 ppm (in D₂O), while ¹³C NMR shows distinct signals at ≈42 ppm (CH₂NH₂) and ≈61 ppm (CH₂OH). Ultraviolet (UV) absorption is weak, with a maximum near 226 nm attributable to n→σ* transitions.[9][1]Nomenclature and Structure
Naming conventions
Ethanolamine is systematically named based on its bifunctional nature, incorporating both an alcohol and an amine group. The preferred IUPAC name is 2-aminoethan-1-ol, reflecting the higher precedence of the hydroxy group as the principal characteristic group, with the amino substituent at position 2 on the ethane chain.[1] An alternative retained IUPAC name for general nomenclature is 2-aminoethanol.[10] When the amine function is considered principal, the systematic name becomes 2-hydroxyethanamine, though this is less commonly used due to seniority rules favoring alcohols.[11] In common usage, the compound is referred to as ethanolamine or monoethanolamine (MEA), the latter emphasizing its role as the simplest member of the ethanolamines series to distinguish it from diethanolamine and triethanolamine, which feature additional hydroxyethyl groups on the nitrogen atom.[1] The etymology of "ethanolamine" stems directly from its structural components: "ethanol" for the C2H4OH moiety and "amine" for the NH2 group, a convention that highlights its hybrid organic functional groups. Standard chemical identifiers for ethanolamine include the CAS Registry Number 141-43-5 and the EC (EINECS) number 205-483-3, which are used internationally for regulatory and inventory purposes.[10] These identifiers ensure precise referencing in scientific literature, patents, and safety data sheets across the ethanolamine family.[11]Molecular structure and isomers
Ethanolamine, with the molecular formula C₂H₇NO, possesses the structural formula HOCH₂CH₂NH₂, consisting of a vicinal amino alcohol where the hydroxyl and amino groups are attached to adjacent carbon atoms in an ethylene chain.[1] The C-O bond length is approximately 1.43 Å, and the C-N bond length is about 1.47 Å, based on computational geometry optimizations, while the C-C bond is around 1.53 Å; bond angles at the methylene carbons approach tetrahedral values of 109°–110° as determined by electron diffraction and ab initio calculations.[13] These structural parameters reflect the single bonds typical of aliphatic alcohols and primary amines, with no unusual distortions in the gas phase or solution. The molecule contains no stereocenters, rendering it achiral and incapable of existing as enantiomers.[1] Conformational flexibility arises primarily around the central C-C bond, allowing gauche and anti arrangements of the OH and NH₂ groups; the gauche conformer predominates due to stabilizing intramolecular hydrogen bonding between the oxygen and nitrogen atoms, with studies showing gauche populations exceeding 80% across various solvents. Within the ethanolamine series, diethanolamine (HN(CH₂CH₂OH)₂) and triethanolamine (N(CH₂CH₂OH)₃) serve as key derivatives, featuring additional hydroxyethyl groups on the nitrogen atom rather than true positional isomers of the parent compound. Tautomerism is negligible, as the stable amino-alcohol form lacks viable alternatives like enol or imine tautomers under standard conditions.Synthesis and Production
Laboratory synthesis
Ethanolamine can be synthesized in the laboratory through the nucleophilic substitution of ethylene chlorohydrin with ammonia, a classical method first demonstrated by Charles-Adolphe Wurtz in 1860 by heating the reactants in a sealed tube with aqueous ammonia. The reaction is represented as: This process is typically conducted at 100–150 °C under pressure to maintain ammonia in solution, producing ethanolamine alongside ammonium chloride as a byproduct.[14] The crude mixture is treated with a base such as sodium hydroxide to liberate the free ethanolamine, followed by extraction into an organic solvent or direct distillation for isolation. A more contemporary laboratory approach involves the ring-opening reaction of ethylene oxide with ammonia, adapted from larger-scale processes but suitable for benchtop execution with controlled addition of reactants. The primary reaction is: Excess ammonia (molar ratio of 10:1 to 20:1) is employed to favor monoethanolamine formation over di- and triethanolamines, with the reaction performed at 50–100 °C and 5–15 bar in a pressure reactor. Typical laboratory yields for monoethanolamine reach 70–80% based on ethylene oxide conversion, which is nearly complete under these conditions.[15][16] Purification of the product mixture commonly entails fractional distillation under reduced pressure, exploiting the boiling point differences: ethanolamine at approximately 170 °C, diethanolamine at 268 °C, and triethanolamine at 335 °C (at atmospheric pressure). This step isolates high-purity ethanolamine while recycling unreacted ammonia.[16] An alternative route utilizes the reduction of glycolamide (2-hydroxyacetamide), prepared from glycolic acid and ammonia, using lithium aluminum hydride in ether or catalytic hydrogenation with Raney nickel. The carbonyl group is reduced to a methylene unit, yielding ethanolamine after workup and purification. This method provides good yields (around 60–75%) but is less frequently used due to the availability of simpler precursors.[17] Reduction of serine derivatives, such as the ethyl ester of N-protected serine, with lithium aluminum hydride followed by deprotection, offers another synthetic option, though it requires additional steps for side-chain management and typically achieves moderate yields (50–70%).Industrial production
Ethanolamine is produced industrially on a large scale through the liquid-phase reaction of ethylene oxide with excess aqueous ammonia, typically at temperatures of 50–100 °C and pressures of 1–10 atm.[18] This exothermic reaction yields a mixture of monoethanolamine (MEA), diethanolamine (DEA), and triethanolamine (TEA) in proportions that can be adjusted by controlling the ammonia-to-ethylene oxide molar ratio (often 10:1 to 20:1) and reaction conditions, with a typical unoptimized ratio approaching 1:1:1 by weight.[19][20] The process operates without catalysts, relying on water in the aqueous ammonia (45–55 wt%) to facilitate the reaction and excess ammonia to enhance selectivity toward MEA while minimizing higher analogs.[19] Byproducts are minimized through recycling of unreacted ammonia and small amounts of MEA and DEA to the reactor, achieving overall yields of 98–99%.[19] Separation of the product mixture occurs via fractional distillation in a series of columns, leveraging the significant differences in boiling points (MEA at 170 °C, DEA at 269 °C, TEA at 335 °C) to isolate high-purity fractions after initial ammonia stripping and water removal.[21][20] Global production of ethanolamines totals approximately 2.5 million metric tons annually in the 2020s, led by major manufacturers such as Dow Chemical Company and BASF SE.[22] The process economics are influenced by feedstocks, with ethylene oxide derived from the catalytic oxidation of ethylene and ammonia synthesized via the energy-intensive Haber-Bosch process.[20]Natural Occurrence and Biological Role
Occurrence in nature
Ethanolamine has been detected in the interstellar medium, highlighting its potential role as a prebiotic compound. In 2021, observations using the IRAM 30 m and Yebes 40 m radio telescopes identified it in the molecular cloud G+0.693–0.027, part of the Sagittarius B2 complex near the galactic center, with column densities on the order of 10^{13} to 10^{14} cm^{-2}. This marks the first astronomical detection of ethanolamine in space, suggesting formation pathways involving neutral-neutral reactions or ion-molecule processes in star-forming regions.[5] Extraterrestrial samples also reveal its presence in natural geological contexts. Ethanolamine was identified in the Almahata Sitta meteorite, fragments of the ureilite from asteroid 2008 TC3 that fell in Sudan in 2008, at a concentration of 19 ppb. Its detection alongside other amino acids and amines indicates possible abiotic origins through processes like Strecker synthesis or formamide hydrolysis in asteroidal materials.[23] On Earth, ethanolamine appears in trace amounts in certain natural settings, such as a degradation product from phospholipid breakdown in plants under phosphate starvation. During such conditions, phospholipases hydrolyze phosphatidylethanolamine, releasing free ethanolamine to facilitate phosphorus recycling.[24]Biological functions
Ethanolamine serves as a crucial component in the biosynthesis of phosphatidylethanolamine (PE), a major glycerophospholipid that constitutes approximately 20-25% of the total phospholipid mass in eukaryotic cell membranes, contributing to membrane structure, fluidity, and curvature.[25] PE is particularly enriched in the inner leaflet of the plasma membrane and plays essential roles in cellular processes such as membrane fusion, endocytosis, and the assembly of membrane proteins.[26] In eukaryotic cells, ethanolamine is incorporated into PE primarily through the Kennedy pathway, a de novo biosynthetic route. The process begins with the phosphorylation of ethanolamine by ethanolamine kinase to form phosphoethanolamine, followed by the action of CTP:phosphoethanolamine cytidylyltransferase to generate CDP-ethanolamine, and concludes with the transfer of the phosphoethanolamine moiety to diacylglycerol by ethanolaminephosphotransferase, yielding PE.[26] This pathway is conserved across eukaryotes and is vital for maintaining phospholipid homeostasis, with disruptions leading to impaired membrane integrity and cellular dysfunction.[27] In neurological contexts, ethanolamine functions as a precursor to choline through methylation pathways in certain neural cells, enabling the subsequent synthesis of acetylcholine, a key neurotransmitter involved in synaptic transmission and cognitive processes.[28] This conversion supports neurotransmission by providing substrates for cholinergic signaling, and ethanolamine-derived PE in neuronal membranes further facilitates ion channel function and vesicle trafficking essential for neural activity.[29] In microbiology, ethanolamine is essential for the growth and survival of various bacteria, particularly pathogens, where it acts as a nutrient source derived from host cell membranes. Mutants deficient in ethanolamine utilization exhibit reduced growth rates and attenuated virulence in host environments, highlighting its role in bacterial adaptation and colonization.[30] For instance, in phylogenetically diverse bacteria such as Escherichia coli and Listeria monocytogenes, ethanolamine catabolism supports metabolic fitness during infection.[31] Bacterial metabolism of ethanolamine often involves oxidation to glycolaldehyde by ethanolamine oxidase, an enzyme that catalyzes the reaction with oxygen to produce glycolaldehyde, ammonia, and hydrogen peroxide, providing carbon and energy sources under nutrient-limited conditions. This oxidase-dependent pathway is observed in species like Pseudomonas, contrasting with the more common ammonia-lyase route but underscoring ethanolamine's versatility in prokaryotic metabolism.[32]Applications
Gas stream scrubbing
Ethanolamine, commonly referred to as monoethanolamine (MEA), is primarily utilized in aqueous solutions at concentrations of 10-30 wt% for the absorption of acidic gases such as carbon dioxide (CO₂) and hydrogen sulfide (H₂S) in amine gas treating processes, particularly in natural gas processing to purify raw gas streams.[33][34] This method, known as gas sweetening, involves contacting the gas stream with the MEA solution in an absorber column, where the amines react chemically with the acid gases to form soluble compounds.[35] The absorption mechanism relies on the formation of a reversible carbamate with CO₂, following the zwitterion pathway: This reaction enables high reactivity and selectivity for CO₂ and H₂S removal. The loaded solution, or "rich amine," is then directed to a regenerator (stripper) column where heat is applied at 100-120 °C to reverse the reaction, releasing the captured gases for venting or further processing while recovering the lean amine for reuse.[36][37] MEA exhibits a high absorption capacity of approximately 0.5 mol CO₂ per mol MEA, stemming from the stoichiometry of the carbamate formation, and is a standard solvent in the majority of amine-based gas treating operations due to its rapid kinetics and effectiveness in bulk acid gas removal.[38][34] To optimize performance, MEA is often blended with diethanolamine (DEA) or methyldiethanolamine (MDEA); for instance, MEA-DEA mixtures enhance absorption rates, while MEA-MDEA formulations improve H₂S selectivity and reduce corrosion in selective acid gas removal scenarios.[39] These systems typically consume 2-3 GJ of energy per ton of CO₂ captured, primarily for steam generation in the regenerator.[40] In post-combustion CO₂ capture applications at power plants, MEA-based scrubbing has been scaled to facilities handling capacities up to several million tons of CO₂ per year, such as proposed projects capturing flue gas from coal-fired units, demonstrating its viability for large-scale carbon mitigation.[41]Other industrial uses
Ethanolamine functions as a surfactant and emulsifier in the formulation of detergents and cleaning products, where it enhances wetting and foaming properties. It is commonly incorporated into shampoos, soaps, and household cleaners at concentrations typically ranging from 5% to 10%, contributing to effective soil removal and product stability.[42][43] In the pharmaceutical sector, ethanolamine serves as a key intermediate in the synthesis of ethanolamine-class antihistamines, such as diphenhydramine, which are used to treat allergic reactions and insomnia. Additionally, it acts as a pH adjuster and buffering agent in various drug formulations, ensuring stability and compatibility.[44][45] Ethanolamine finds application in cosmetics and personal care products for pH adjustment, helping to maintain optimal alkalinity in formulations like hair dyes and lotions. In hair dyes, it often replaces ammonia to swell the hair cuticle and facilitate color penetration, while its humectant properties aid in moisture retention in lotions and creams.[46][47] In the textile industry, ethanolamine is utilized as a softening agent to impart antistatic and smooth properties to fabrics during pre- and post-treatment processes. It also serves as a wetting agent and dye stabilizer, promoting even color distribution and improved dye uptake on fibers. Similar roles extend to paper production, where it acts as a softening additive in pulp processing and dye formulations.[48][49] Beyond these, ethanolamine is employed in herbicide formulations, notably as the ethanolamine salt of glyphosate, which enhances solubility and efficacy in agricultural applications. In cement manufacturing, ethanolamine salts function as grinding aids, reducing surface tension to improve dispersion and mill efficiency while influencing hydration rates for better mechanical properties. In metalworking fluids, it provides corrosion inhibition, pH stabilization, and alkalinity boosting, extending fluid life and protecting metal surfaces during machining.[50][51][52]Reactions
Reactions with acids and CO₂
Ethanolamine, a primary amine with a pKa of 9.5 for its conjugate acid at 25 °C, readily undergoes protonation in acidic media to form the ethanolammonium ion (HOCH₂CH₂NH₃⁺).[1] This acid-base reaction is characterized by the equilibrium HOCH₂CH₂NH₂ + H⁺ ⇌ HOCH₂CH₂NH₃⁺, where the protonation constant is derived from the pKa value reported in standard compilations of organic base dissociation constants. The resulting conjugate acid is a stable cationic species that influences ethanolamine's behavior in aqueous solutions, particularly in buffering applications. Ethanolamine forms salts with various acids, including inorganic and organic species. With hydrochloric acid, it produces ethanolamine hydrochloride, a white crystalline solid with a melting point of 82–84 °C, density of approximately 1.07 g/cm³ at 20–25 °C, and solubility in water of approximately 33 g/100 mL at 20 °C, though it is only slightly soluble in DMSO when heated.[53][54][55] This salt is hygroscopic and used in biochemical assays due to its role as a buffering agent near physiological pH. With fatty acids, such as oleic or stearic acid, ethanolamine reacts to form alkanolamine soaps, which are neutral carboxylates (e.g., HOCH₂CH₂NH₃⁺ RCOO⁻, where R is a long-chain alkyl group).[43] These soaps exhibit emulsifying properties, aiding in the dispersion of oils in water-based formulations, and are valued for their mildness compared to alkali metal soaps in personal care products.[56] A key reaction of ethanolamine involves carbon dioxide, where two molecules of the amine react to form a carbamate salt via the zwitterionic mechanism: This carbamate formation is exothermic, with an enthalpy of reaction approximately -85.4 kJ/mol CO₂ in aqueous solution at typical absorption conditions.[57] The equilibrium constant for carbamate formation (K_c = [HOCH₂CH₂NH₃⁺][HOCH₂CH₂NHCOO⁻] / [HOCH₂CH₂NH₂]²[CO₂]) is about 12.5 M⁻¹ at 298 K, decreasing with increasing temperature (e.g., to 4.8 M⁻¹ at 328 K), which reflects the endothermic nature of the reverse dissociation.[58] Kinetically, the reaction proceeds via a zwitterion intermediate, with the overall second-order rate constant for the forward reaction around 4.6 × 10³ M⁻¹ s⁻¹ at 293 K in aqueous media, governed by the base-catalyzed hydrolysis of CO₂ followed by amine attack. These properties underpin ethanolamine's use in CO₂ scrubbing processes. Ethanolamine reacts analogously with hydrogen sulfide (H₂S) through proton transfer, forming ethanolammonium hydrosulfide (HOCH₂CH₂NH₃⁺ HS⁻) in aqueous solutions, similar to its interaction with CO₂ but without carbamate formation. The reaction is rapid and essentially irreversible under absorption conditions due to the weak acidity of H₂S (pKa ≈ 7), yielding stable sulfide salts that facilitate H₂S removal from gas streams.[59] The mechanism involves direct protonation of the amine by H₂S, with equilibrium favoring the salt at low partial pressures of H₂S.[60]Other reactions
Ethanolamine undergoes nucleophilic substitution reactions with alkyl halides, primarily at the nitrogen atom, leading to N-alkylated derivatives such as N-methylethanolamine upon treatment with methyl iodide.[61] This N-alkylation can be achieved selectively under phase transfer catalysis conditions, where the stoichiometry of reagents influences regioselectivity toward monoalkylation products, minimizing over-alkylation or O-alkylation side products.[62] The hydroxyl group of ethanolamine can participate in esterification reactions with carboxylic acids to form 2-(acyloxy)ethylamines, but the presence of the adjacent amine group poses significant challenges, as it promotes competing amide formation or intramolecular O-to-N acyl migration, converting the initial ester to an amide.[63] To achieve selective esterification, temporary protection of the amine functionality, such as through protonation or acylation, is often required prior to reaction with the carboxylic acid under acidic catalysis. Oxidation of ethanolamine typically targets the C-C bond adjacent to the amino and hydroxyl groups, yielding glycolaldehyde as the primary product. Sodium periodate serves as an effective oxidant for this cleavage, proceeding rapidly in aqueous media at neutral to mildly acidic pH to produce glycolaldehyde and ammonia.[64] Enzymatic oxidation by ethanolamine oxidase, found in certain bacteria, also converts ethanolamine to glycolaldehyde under physiological conditions, facilitating its role in metabolic pathways.[65] Additionally, exposure to air leads to slow auto-oxidation, generating degradation products like amino acids through radical-mediated processes. In polyurethane synthesis, ethanolamine acts as a difunctional precursor, reacting primarily at the hydroxyl group with isocyanates to form urethane linkages, while the amine group can form urea linkages, enabling its use as a chain extender or crosslinker in polymer networks.[66] This reactivity contributes to the formation of rigid polyurethane foams when combined with polyisocyanates and catalysts, enhancing mechanical properties through hydrogen bonding in the resulting urethanes.[67] Thermal dehydration of ethanolamine at elevated temperatures above 200 °C, typically 350–450 °C in the gas phase over solid catalysts like metal oxides, cyclizes the molecule to ethyleneimine (aziridine) via intramolecular elimination of water.[68] This process operates under reduced pressure to favor the volatile product, achieving high selectivity with catalysts that balance acidity and basicity to promote dehydration over decomposition.[69]Safety, Toxicity, and Environmental Impact
Health and safety
Ethanolamine is corrosive to the skin and eyes, causing severe burns and irritation upon contact. It is also an irritant to the respiratory tract when inhaled. The oral LD50 in rats is 1.72 g/kg, indicating moderate acute toxicity via ingestion.[70][1] Exposure to ethanolamine primarily occurs through inhalation, skin contact, or ingestion in occupational settings. The odor threshold is approximately 2.6 ppm, providing some warning of presence, though it may not detect levels above exposure limits. The Occupational Safety and Health Administration (OSHA) permissible exposure limit (PEL) is 3 ppm (6 mg/m³) as an 8-hour time-weighted average (TWA), while the American Conference of Governmental Industrial Hygienists (ACGIH) threshold limit value (TLV) is 3 ppm TWA with a short-term exposure limit (STEL) of 6 ppm. The National Institute for Occupational Safety and Health (NIOSH) immediately dangerous to life or health (IDLH) value is 30 ppm.[71][1][72] Safe handling of ethanolamine requires personal protective equipment (PPE), including chemical-resistant gloves, safety goggles, and protective clothing to prevent skin and eye contact. It should be used in well-ventilated areas or under a fume hood to minimize inhalation risks. Storage must occur in a cool, dry, ventilated location away from heat sources and incompatible materials, such as strong oxidizers, which can lead to violent reactions.[70] In case of exposure, first aid measures include immediately flushing affected skin or eyes with large amounts of water for at least 15 minutes and seeking medical attention. For inhalation, move the person to fresh air and provide oxygen if breathing is difficult; professional medical evaluation is recommended. If ingested, do not induce vomiting and obtain immediate medical help.[70] Ethanolamine is not classified as a carcinogen by the International Agency for Research on Cancer (IARC Group 3) and shows minimal reproductive toxicity based on available data.[73]Environmental considerations
Ethanolamine is readily biodegradable in aerobic environments, with studies demonstrating greater than 70% degradation within 28 days under OECD 301 test conditions, such as the modified Sturm test (301B) and closed bottle test (301D). This rapid breakdown occurs primarily through microbial action, resulting in mineralization to carbon dioxide and biomass. Its low bioaccumulation potential further limits long-term ecological buildup, as indicated by an octanol-water partition coefficient (log Kow) of -1.31 and a bioconcentration factor (BCF) estimated at 3.2, both well below thresholds for concern (log Kow < 3 and BCF < 100).[1] Ecotoxicity assessments reveal moderate impacts on aquatic life. For fish, the 96-hour LC50 values range from 114 to 196 mg/L in species like rainbow trout (Oncorhynchus mykiss), indicating potential acute toxicity at elevated concentrations. Algal species show higher sensitivity, with 72-hour EC50 values around 15 mg/L for green algae such as Pseudokirchneriella subcapitata, suggesting inhibition of growth at levels exceeding 10 mg/L. These effects are attributed to ethanolamine's interference with cellular processes in primary producers, though chronic risks are mitigated by its biodegradability.[70][1] Regulatory frameworks address ethanolamine's environmental release. In the European Union, it is registered under REACH (EC 205-483-3), classified as harmful to aquatic life with long-lasting effects (Aquatic Chronic 3, H412), requiring risk assessments for manufacturers and importers exceeding one tonne annually. In the United States, it is listed on the TSCA inventory as an active substance, subject to reporting under the Clean Water Act for industrial discharges. Effluent limitations for industrial discharges, including those from gas processing, are site-specific under NPDES permits and may include restrictions on ammonia and related compounds to protect receiving waters.[74][75] Industrial emissions of ethanolamine primarily stem from gas scrubbing and chemical manufacturing, but its low environmental persistence minimizes accumulation. In water, aerobic biodegradation half-lives range from 10 to 58 hours, driven by first-order kinetics in activated sludge systems, ensuring rapid removal in wastewater treatment plants (75-90% efficiency). Atmospheric half-lives are similarly short, around 11-27 hours due to photodegradation.[76][77] Sustainability efforts focus on reducing ethanolamine's environmental footprint in CO2 capture applications. Greener alternatives, such as deep eutectic solvents (e.g., choline chloride-based mixtures) and piperazine-promoted amines, offer lower volatility, reduced energy for regeneration, and decreased degradation products compared to traditional 30% aqueous ethanolamine solutions. Process recycling, including thermal reclaiming and ion-exchange purification of spent amine streams, recovers up to 95% of ethanolamine, minimizing waste and emissions in closed-loop systems.[78][79]History
Discovery
Ethanolamine was first synthesized in 1860 by the French chemist Charles-Adolphe Wurtz, who prepared it and its salts by heating ethylene chlorohydrin with aqueous ammonia in a sealed tube.[80] This laboratory synthesis marked the initial identification of the compound as a simple amino alcohol, though the reaction also produced diethanolamine and triethanolamine as byproducts. Wurtz's work built on his earlier studies of ethylene derivatives and ammonia reactions, providing one of the earliest examples of synthesizing beta-amino alcohols.[81] However, the first deliberate isolation of ethanolamine from natural sources occurred in 1884 by biochemist Johann Ludwig Wilhelm Thudichum, who extracted it from brain cephalin lipids during saponification and hydrolysis processes. Thudichum mistakenly considered the isolated ethanolamine a decomposition artifact of choline but documented its presence in brain tissue, linking it to lipid structures.[82] The first isolation of the free base ethanolamine was achieved in 1897 by German chemist Ludwig Knorr at the University of Jena, who reacted ethylene chlorohydrin with ammonia to produce the compound.[2] This method laid the groundwork for later industrial processes, though initial production remained limited to small-scale applications. Structural confirmation of ethanolamine came in the 1880s through degradation studies on cephalin by Thudichum and contemporaries, involving hydrolysis and oxidation to yield identifiable fragments like glycolic acid and ammonia, verifying the 2-aminoethanol formula.[83] These analyses established its role as the polar head group in phosphatidylethanolamine. Initially named "colamine," a term derived from its association with "glue-like" lipid extracts (from Greek kolla, meaning glue), reflecting its origin in viscous brain substances.[84]Commercial development
Commercial production of ethanolamine began in the early 1930s via reaction of ammonia with ethylene halohydrin, but it gained significant scale only after World War II in the 1940s and 1950s, coinciding with the expansion of ethylene oxide manufacturing.[85] The modern ethylene oxide-ammonia route was integrated into large-scale petrochemical operations, improving efficiency and reducing costs compared to earlier halohydrin methods, positioning ethanolamine as a key intermediate in chemical manufacturing.[86] By the 1960s, production expanded rapidly to meet demand in detergents and gas treating applications, where ethanolamine served as a surfactant precursor and acid gas absorbent, respectively. The 1970s oil crisis further accelerated its use in gas scrubbing to purify natural gas streams, enhancing recovery and compliance with stricter environmental standards amid energy shortages.[87] In the 1980s, research intensified on ethanolamine-based solvents for CO₂ capture, marking its entry into flue gas treatment processes.[88] The 2000s saw a focus on green chemistry applications, with ethanolamine incorporated into sustainable formulations for eco-friendly detergents and reduced-emission processes.[89] Today, ethanolamine production is tightly integrated with the global ethylene industry, relying on ethylene oxide as a feedstock, and experiences annual growth of 5.2% (as of 2024) driven primarily by demand in gas purification and carbon capture and storage (CCS) technologies for mitigating industrial emissions.[90]References
- https://www.[sigmaaldrich](/page/Sigma-Aldrich).com/US/en/product/aldrich/15014




