Recent from talks
Integrin
Knowledge base stats:
Talk channels stats:
Members stats:
Integrin
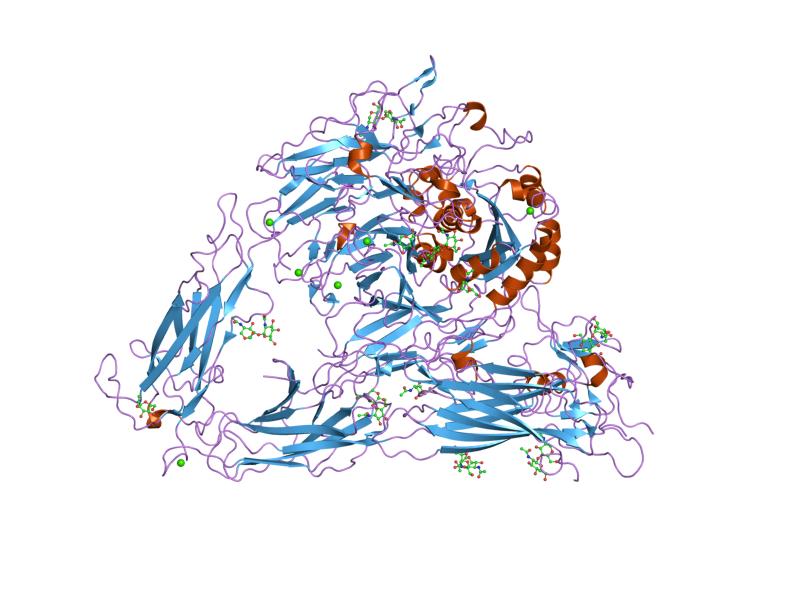
Integrins are transmembrane receptors that help cell–cell and cell–extracellular matrix (ECM) adhesion. Upon ligand binding, integrins activate signal transduction pathways that mediate cellular signals such as regulation of the cell cycle, organization of the intracellular cytoskeleton, and movement of new receptors to the cell membrane. The presence of integrins allows rapid and flexible responses to events at the cell surface (e.g. signal platelets to initiate an interaction with coagulation factors).
Several types of integrins exist, and one cell generally has multiple different types on its surface. Integrins are found in all animals while integrin-like receptors are found in plant cells.
Integrins work alongside other proteins such as cadherins, the immunoglobulin superfamily cell adhesion molecules, selectins and syndecans, to mediate cell–cell and cell–matrix interaction. Ligands for integrins include fibronectin, vitronectin, collagen and laminin.
Integrins are obligate heterodimers composed of α and β subunits. Several genes code for multiple isoforms of these subunits, which gives rise to an array of unique integrins with varied activity. In mammals, integrins are assembled from eighteen α and eight β subunits, in Drosophila five α and two β subunits, and in Caenorhabditis nematodes two α subunits and one β subunit. The α and β subunits are both class I transmembrane proteins, so each penetrates the plasma membrane once, and can possess several cytoplasmic domains.
Variants of some subunits are formed by differential RNA splicing; for example, four variants of the beta-1 subunit exist. Through different combinations of the α and β subunits, 24 unique mammalian integrins are generated, excluding splice- and glycosylation variants.
Integrin subunits span the cell membrane and have short cytoplasmic domains of 40–70 amino acids. The exception is the beta-4 subunit, which has a cytoplasmic domain of 1,088 amino acids, one of the largest of any membrane protein. Outside the cell membrane, the α and β chains lie close together along a length of about 23 nm; the final 5 nm N-termini of each chain forms a ligand-binding region for the ECM. They have been compared to lobster claws, although they don't actually "pinch" their ligand, they chemically interact with it at the insides of the "tips" of their "pinchers".
The molecular mass of the integrin subunits can vary from 90 kDa to 160 kDa. Beta subunits have four cysteine-rich repeated sequences. Both α and β subunits bind several divalent cations. The role of divalent cations in the α subunit is unknown, but may stabilize the folds of the protein. The cations in the β subunits are more interesting: they are directly involved in coordinating at least some of the ligands that integrins bind.
Integrins can be categorized in multiple ways. For example, some α chains have an additional structural element (or "domain") inserted toward the N-terminal, the alpha-A domain (so called because it has a similar structure to the A-domains found in the protein von Willebrand factor; it is also termed the α-I domain). Integrins carrying this domain either bind to collagens (e.g. integrins α1 β1, and α2 β1), or act as cell-cell adhesion molecules (integrins of the β2 family). This α-I domain is the binding site for ligands of such integrins. Those integrins that don't carry this inserted domain also have an A-domain in their ligand binding site, but this A-domain is found on the β subunit.
Hub AI
Integrin AI simulator
(@Integrin_simulator)
Integrin
Integrins are transmembrane receptors that help cell–cell and cell–extracellular matrix (ECM) adhesion. Upon ligand binding, integrins activate signal transduction pathways that mediate cellular signals such as regulation of the cell cycle, organization of the intracellular cytoskeleton, and movement of new receptors to the cell membrane. The presence of integrins allows rapid and flexible responses to events at the cell surface (e.g. signal platelets to initiate an interaction with coagulation factors).
Several types of integrins exist, and one cell generally has multiple different types on its surface. Integrins are found in all animals while integrin-like receptors are found in plant cells.
Integrins work alongside other proteins such as cadherins, the immunoglobulin superfamily cell adhesion molecules, selectins and syndecans, to mediate cell–cell and cell–matrix interaction. Ligands for integrins include fibronectin, vitronectin, collagen and laminin.
Integrins are obligate heterodimers composed of α and β subunits. Several genes code for multiple isoforms of these subunits, which gives rise to an array of unique integrins with varied activity. In mammals, integrins are assembled from eighteen α and eight β subunits, in Drosophila five α and two β subunits, and in Caenorhabditis nematodes two α subunits and one β subunit. The α and β subunits are both class I transmembrane proteins, so each penetrates the plasma membrane once, and can possess several cytoplasmic domains.
Variants of some subunits are formed by differential RNA splicing; for example, four variants of the beta-1 subunit exist. Through different combinations of the α and β subunits, 24 unique mammalian integrins are generated, excluding splice- and glycosylation variants.
Integrin subunits span the cell membrane and have short cytoplasmic domains of 40–70 amino acids. The exception is the beta-4 subunit, which has a cytoplasmic domain of 1,088 amino acids, one of the largest of any membrane protein. Outside the cell membrane, the α and β chains lie close together along a length of about 23 nm; the final 5 nm N-termini of each chain forms a ligand-binding region for the ECM. They have been compared to lobster claws, although they don't actually "pinch" their ligand, they chemically interact with it at the insides of the "tips" of their "pinchers".
The molecular mass of the integrin subunits can vary from 90 kDa to 160 kDa. Beta subunits have four cysteine-rich repeated sequences. Both α and β subunits bind several divalent cations. The role of divalent cations in the α subunit is unknown, but may stabilize the folds of the protein. The cations in the β subunits are more interesting: they are directly involved in coordinating at least some of the ligands that integrins bind.
Integrins can be categorized in multiple ways. For example, some α chains have an additional structural element (or "domain") inserted toward the N-terminal, the alpha-A domain (so called because it has a similar structure to the A-domains found in the protein von Willebrand factor; it is also termed the α-I domain). Integrins carrying this domain either bind to collagens (e.g. integrins α1 β1, and α2 β1), or act as cell-cell adhesion molecules (integrins of the β2 family). This α-I domain is the binding site for ligands of such integrins. Those integrins that don't carry this inserted domain also have an A-domain in their ligand binding site, but this A-domain is found on the β subunit.