Recent from talks
Nothing was collected or created yet.
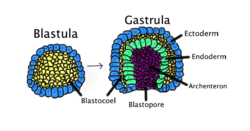
Gastrulation
View on Wikipedia| Gastrulation | |
|---|---|
 Gastrulation occurs when a blastula, made up of one layer, folds inward and enlarges to create a gastrula. This diagram is color-coded: ectoderm, blue; endoderm, green; blastocoel (the yolk sac), yellow; and archenteron (the primary gut), purple. | |
| Identifiers | |
| MeSH | D054262 |
| Anatomical terminology | |
Gastrulation is the stage in the early embryonic development of most animals, during which the blastula (a single-layered hollow sphere of cells), or in mammals, the blastocyst, is reorganized into a two-layered or three-layered embryo known as the gastrula.[1] Before gastrulation, the embryo is a continuous epithelial sheet of cells; by the end of gastrulation, the embryo has begun differentiation to establish distinct cell lineages, set up the basic axes of the body (e.g. dorsal–ventral, anterior–posterior), and internalized one or more cell types, including the prospective gut.[2]
Gastrula layers
[edit]In triploblastic organisms, the gastrula is trilaminar (three-layered). These three germ layers are the ectoderm (outer layer), mesoderm (middle layer), and endoderm (inner layer).[3][4] In diploblastic organisms, such as Cnidaria and Ctenophora, the gastrula has only ectoderm and endoderm. The two layers are also sometimes referred to as the hypoblast and epiblast.[5] Sponges do not go through the gastrula stage.
Gastrulation takes place after cleavage and the formation of the blastula, or blastocyst. Gastrulation is followed by organogenesis, when individual organs develop within the newly formed germ layers.[6] Each layer gives rise to specific tissues and organs in the developing embryo.
- The ectoderm gives rise to epidermis, the nervous system, and to the neural crest in vertebrates.[2]
- The endoderm gives rise to the epithelium of the digestive system and respiratory system, and organs associated with the digestive system, such as the liver and pancreas.[2]
- The mesoderm gives rise to many cell types such as muscle, bone, and connective tissue. In vertebrates, mesoderm derivatives include the notochord, the heart, blood and blood vessels, the cartilage of the ribs and vertebrae, and the dermis.[2][7]
Following gastrulation, cells in the body are either organized into sheets of connected cells (as in epithelia), or as a mesh of isolated cells, such as mesenchyme.[4][8]
Basic cell movements
[edit]Although gastrulation patterns exhibit enormous variation throughout the animal kingdom, they are unified by the five basic types of cell movements that occur during gastrulation:[2][9]
- Invagination
- Involution
- Ingression
- Delamination
- Epiboly
Etymology
[edit]The terms "gastrula" and "gastrulation" were coined by Ernst Haeckel, in his 1872 work "Biology of Calcareous Sponges".[10] Gastrula (literally, "little belly") is a neo-Latin diminutive based on the Ancient Greek γαστήρ gastḗr ("a belly").
Importance
[edit]Lewis Wolpert, pioneering developmental biologist in the field, has been credited for noting that "It is not birth, marriage, or death, but gastrulation which is truly the most important time in your life."[2][11]
Model systems
[edit]Gastrulation is highly variable across the animal kingdom but has underlying similarities. Gastrulation has been studied in many animals, but some models have been used for longer than others. Furthermore, it is easier to study development in animals that develop outside the mother. Model organisms whose gastrulation is understood in the greatest detail include the mollusc, sea urchin, frog, and chicken. A human model system is the gastruloid.
Protostomes versus deuterostomes
[edit]
The distinction between protostomes and deuterostomes is based on the direction in which the mouth (stoma) develops in relation to the blastopore. Protostome derives from the Greek word protostoma meaning "first mouth" (πρῶτος + στόμα) whereas Deuterostome's etymology is "second mouth" from the words second and mouth (δεύτερος + στόμα).[citation needed]
The major distinctions between deuterostomes and protostomes are found in embryonic development:
- Mouth/anus
- In protostome development, the first opening in development, the blastopore, becomes the animal's mouth.
- In deuterostome development, the blastopore becomes the animal's anus.
- Cleavage
- Protostomes have what is known as spiral cleavage which is determinate, meaning that the fate of the cells is determined as they are formed.
- Deuterostomes have what is known as radial cleavage that is indeterminate.
Sea urchins
[edit]Sea urchins have been important model organisms in developmental biology since the 19th century.[12] Their gastrulation is often considered the archetype for invertebrate deuterostomes.[13]
Sea urchins exhibit highly stereotyped cleavage patterns and cell fates. Maternally deposited mRNAs establish the organizing center of the sea urchin embryo. Canonical Wnt and Delta-Notch signaling progressively segregate progressive endoderm and mesoderm.[14]
The first cells to internalize are the primary mesenchyme cells (PMCs), which have a skeletogenic fate, which ingress during the blastula stage. Gastrulation – internalization of the prospective endoderm and non-skeletogenic mesoderm – begins shortly thereafter with invagination and other cell rearrangements the vegetal pole, which contribute approximately 30% to the final archenteron length. The gut's final length depends on cell rearrangements within the archenteron.[15]
Amphibians
[edit]The frog genus Xenopus has been used as a model organism for the study of gastrulation.[16]
Symmetry breaking
[edit]The sperm contributes one of the two mitotic asters needed to complete first cleavage. The sperm can enter anywhere in the animal half of the egg but its exact point of entry will break the egg's radial symmetry by organizing the cytoskeleton. Prior to first cleavage, the egg's cortex rotates relative to the internal cytoplasm by the coordinated action of microtubules, in a process known as cortical rotation. This displacement brings maternally loaded determinants of cell fate from the equatorial cytoplasm and vegetal cortex into contact, and together these determinants set up the organizer. Thus, the area on the vegetal side opposite the sperm entry point will become the organizer.[17] Hilde Mangold, working in the lab of Hans Spemann, demonstrated that this special "organizer" of the embryo is necessary and sufficient to induce gastrulation.[18][19][20]
The dorsal lip of the blastopore is the mechanical driver of gastrulation, and the first sign of invagination seen in the frog.[citation needed]
Germ layer differentiation
[edit]Specification of endoderm depends on rearrangement of maternally deposited determinants, leading to nuclearization of Beta-catenin. Mesoderm is induced by signaling from the presumptive endoderm to cells that would otherwise become ectoderm.[17]
Cell signaling
[edit]In the frog, Xenopus, one of the signals is retinoic acid (RA).[21] RA signaling in this organism can affect the formation of the endoderm and depending on the timing of the signaling, it can determine the fate whether its pancreatic, intestinal, or respiratory. Other signals such as Wnt and BMP also play a role in respiratory fate of the Xenopus by activating cell lineage tracers.[21]
Amniotes
[edit]Overview
[edit]In amniotes (reptiles, birds and mammals), gastrulation involves the creation of the blastopore, an opening into the archenteron. Note that the blastopore is not an opening into the blastocoel, the space within the blastula, but represents a new inpocketing that pushes the existing surfaces of the blastula together. In amniotes, gastrulation occurs in the following sequence: (1) the embryo becomes asymmetric; (2) the primitive streak forms; (3) cells from the epiblast at the primitive streak undergo an epithelial to mesenchymal transition and ingress at the primitive streak to form the germ layers.[7]
Symmetry breaking
[edit]In preparation for gastrulation, the embryo must become asymmetric along both the proximal-distal axis and the anteroposterior axis. The proximal-distal axis is formed when the cells of the embryo form the "egg cylinder", which consists of the extraembryonic tissues, which give rise to structures like the placenta, at the proximal end and the epiblast at the distal end. Many signaling pathways contribute to this reorganization, including BMP, FGF, nodal, and Wnt. Visceral endoderm surrounds the epiblast. The distal visceral endoderm (DVE) migrates to the anterior portion of the embryo, forming the anterior visceral endoderm (AVE). This breaks anterior-posterior symmetry and is regulated by nodal signaling.[7]

Germ layer determination
[edit]The primitive streak is formed at the beginning of gastrulation and is found at the junction between the extraembryonic tissue and the epiblast on the posterior side of the embryo and the site of ingression.[22] Formation of the primitive streak is reliant upon nodal signaling[7] in the Koller's sickle within the cells contributing to the primitive streak and BMP4 signaling from the extraembryonic tissue.[22][23] Furthermore, Cer1 and Lefty1 restrict the primitive streak to the appropriate location by antagonizing nodal signaling.[24] The region defined as the primitive streak continues to grow towards the distal tip.[7]
During the early stages of development, the primitive streak is the structure that will establish bilateral symmetry, determine the site of gastrulation and initiate germ layer formation.[25] To form the streak, reptiles, birds and mammals arrange mesenchymal cells along the prospective midline, establishing the first embryonic axis, as well as the place where cells will ingress and migrate during the process of gastrulation and germ layer formation.[26] The primitive streak extends through this midline and creates the antero-posterior body axis,[27] becoming the first symmetry-breaking event in the embryo, and marks the beginning of gastrulation.[28] This process involves the ingression of mesoderm and endoderm progenitors and their migration to their ultimate position,[27][29] where they will differentiate into the three germ layers.[26] The localization of the cell adhesion and signaling molecule beta-catenin is critical to the proper formation of the organizer region that is responsible for initiating gastrulation.
Cell internalization
[edit]In order for the cells to move from the epithelium of the epiblast through the primitive streak to form a new layer, the cells must undergo an epithelial to mesenchymal transition (EMT) to lose their epithelial characteristics, such as cell–cell adhesion. FGF signaling is necessary for proper EMT. FGFR1 is needed for the up regulation of SNAI1, which down regulates E-cadherin, causing a loss of cell adhesion. Following the EMT, the cells ingress through the primitive streak and spread out to form a new layer of cells or join existing layers. FGF8 is implicated in the process of this dispersal from the primitive streak.[24]
Cell signaling driving gastrulation
[edit]During gastrulation, the cells are differentiated into the ectoderm or mesendoderm, which then separates into the mesoderm and endoderm.[21] The endoderm and mesoderm form due to the nodal signaling. Nodal signaling uses ligands that are part of TGFβ family. These ligands will signal transmembrane serine/threonine kinase receptors, and this will then phosphorylate Smad2 and Smad3. This protein will then attach itself to Smad4 and relocate to the nucleus where the mesendoderm genes will begin to be transcribed. The Wnt pathway along with β-catenin plays a key role in nodal signaling and endoderm formation.[30] Fibroblast growth factors (FGF), canonical Wnt pathway, bone morphogenetic protein (BMP), and retinoic acid (RA) are all important in the formation and development of the endoderm.[21] FGF are important in producing the homeobox gene which regulates early anatomical development. BMP signaling plays a role in the liver and promotes hepatic fate. RA signaling also induce homeobox genes such as Hoxb1 and Hoxa5. In mice, if there is a lack in RA signaling the mouse will not develop lungs.[21] RA signaling also has multiple uses in organ formation of the pharyngeal arches, the foregut, and hindgut.[21]
Gastrulation in vitro
[edit]There have been a number of attempts to understand the processes of gastrulation using in vitro techniques in parallel and complementary to studies in embryos, usually though the use of 2D[31][32][33] and 3D cell (Embryonic organoids) culture techniques[34][35][36][37] using embryonic stem cells (ESCs) or induced pluripotent stem cells (iPSCs). These are associated with number of clear advantages in using tissue-culture based protocols, some of which include reducing the cost of associated in vivo work (thereby reducing, replacing and refining the use of animals in experiments; the 3Rs), being able to accurately apply agonists/antagonists in spatially and temporally specific manner[35][36] which may be technically difficult to perform during Gastrulation. However, it is important to relate the observations in culture to the processes occurring in the embryo for context.
To illustrate this, the guided differentiation of mouse ESCs has resulted in generating primitive streak–like cells that display many of the characteristics of epiblast cells that traverse through the primitive streak[31] (e.g. transient brachyury up regulation and the cellular changes associated with an epithelial to mesenchymal transition[31]), and human ESCs cultured on micro patterns, treated with BMP4, can generate spatial differentiation pattern similar to the arrangement of the germ layers in the human embryo.[32][33] Finally, using 3D embryoid body- and organoid-based techniques, small aggregates of mouse ESCs (Embryonic Organoids, or Gastruloids) are able to show a number of processes of early mammalian embryo development such as symmetry-breaking, polarisation of gene expression, gastrulation-like movements, axial elongation and the generation of all three embryonic axes (anteroposterior, dorsoventral and left-right axes).[34][35][36][38]
In vitro fertilization occurs in a laboratory. The process of in vitro fertilization is when mature eggs are removed from the ovaries and are placed in a cultured medium where they are fertilized by sperm. In the culture the embryo will form.[39] 14 days after fertilization the primitive streak forms. The formation of the primitive streak has been known to some countries as "human individuality".[40] This means that the embryo is now a being itself, it is its own entity. The countries that believe this have created a 14-day rule in which it is illegal to study or experiment on a human embryo after the 14-day period in vitro. Research has been conducted on the first 14 days of an embryo, but no known studies have been done after the 14 days.[41] With the rule in place, mice embryos are used understand the development after 14 days; however, there are differences in the development between mice and humans.
See also
[edit]References
[edit]Notes
[edit]- ^ Urry, Lisa (2016). Campbell Biology (11th ed.). Pearson. p. 1047. ISBN 978-0-13-409341-3.
- ^ a b c d e f Gilbert, Scott F.; Michael J. F. Barresi (2016). Developmental biology (Eleventh ed.). Sunderland, Massachusetts: Sinauer. ISBN 978-1-60535-470-5. OCLC 945169933.
- ^ Mundlos 2009: p. 422
- ^ a b McGeady, 2004: p. 34
- ^ Jonathon M.W., Slack (2013). Essential Developmental Biology. West Sussex, UK: Wiley-Blackwell. p. 122. ISBN 978-0-470-92351-1.
- ^ Hall, 1998: pp. 132-134
- ^ a b c d e Arnold & Robinson, 2009
- ^ Hall, 1998: p. 177
- ^ Gilbert, Scott F. (2000). "Figure 8.6, [Types of cell movements during...]". www.ncbi.nlm.nih.gov. Retrieved 11 May 2022.
- ^ Ereskovsky 2010: p. 236
- ^ Wolpert L (2008) The triumph of the embryo. Courier Corporation, page 12. ISBN 978-0-486-46929-4
- ^ Laubichler, M.D. and Davidson, E. H. (2008). "Boveri's long experiment: sea urchin merogones and the establishment of the role of nuclear chromosomes in development". Developmental Biology. 314(1):1–11. doi:10.1016/j.ydbio.2007.11.024.
- ^ McClay, David R.; Gross, J.M.; Range, Ryan; Peterson, R.E.; Bradham, Cynthia (2004). "Chapter 9: Sea Urchin Gastrulation". In Stern, Claudio D. (ed.). Gastrulation: From Cells to Embryos. Cold Spring Harbor Laboratory Press. pp. 123–137. ISBN 978-0-87969-707-5.
- ^ McClay, D. R. 2009. Cleavage and Gastrulation in Sea Urchin. eLS. doi:10.1002/9780470015902.a0001073.pub2
- ^ Hardin J D (1990). "Context-sensitive cell behaviors during gastrulation" (PDF). Semin. Dev. Biol. 1: 335–345.
- ^ Blum, Martin; Beyer, Tina; Weber, Thomas; Vick, Philipp; Andre, Philipp; Bitzer, Eva; Schweickert, Axel (June 2009). "Xenopus , an ideal model system to study vertebrate left-right asymmetry". Developmental Dynamics. 238 (6): 1215–1225. doi:10.1002/dvdy.21855. PMID 19208433. S2CID 39348233.
- ^ a b Gilbert, Scott F. (2000). "Axis Formation in Amphibians: The Phenomenon of the Organizer, The Progressive Determination of the Amphibian Axes". Developmental Biology. Sinauer Associates.
- ^ Gilbert, Scott F. (2000). "Figure 10.20, [Organization of a secondary axis...]". www.ncbi.nlm.nih.gov. Retrieved 1 June 2020.
- ^ Spemann H., Mangold H. (1924). "Über Induktion von Embryonanlagen durch Implantation artfremder Organisatoren". Roux' Arch. F. Entw. Mech. 100 (3–4): 599–638. doi:10.1007/bf02108133. S2CID 12605303.
- ^ De Robertis Edward (2006). "Spemann's organizer and self-regulation in amphibian embryos". Nature Reviews Molecular Cell Biology. 7 (4): 296–302. doi:10.1038/nrm1855. PMC 2464568. PMID 16482093.
- ^ a b c d e f Zorn A, Wells J (2009). "Vertebrate Endoderm Development and Organ Formation". Annu Rev Cell Dev Biol. 25: 221–251. doi:10.1146/annurev.cellbio.042308.113344. PMC 2861293. PMID 19575677.
- ^ a b Tam & Behringer, 1997
- ^ Catala, 2005: p. 1535
- ^ a b Tam, P.P.; Loebel, D.A (2007). "Gene function in mouse embryogenesis: get set for gastrulation". Nat Rev Genet. 8 (5): 368–81. doi:10.1038/nrg2084. PMID 17387317. S2CID 138874.
- ^ Sheng, Guojun; Arias, Alfonso Martinez; Sutherland, Ann (2021-12-03). "The primitive streak and cellular principles of building an amniote body through gastrulation". Science. 374 (6572) abg1727. doi:10.1126/science.abg1727. PMID 34855481. S2CID 244841366.
- ^ a b Mikawa T, Poh AM, Kelly KA, Ishii Y, Reese DE (2004). "Induction and patterning of the primitive streak, an organizing center of gastrulation in the amniote". Dev Dyn. 229 (3): 422–32. doi:10.1002/dvdy.10458. PMID 14991697. S2CID 758473.
- ^ a b Downs KM. (2009). "The enigmatic primitive streak: prevailing notions and challenges concerning the body axis of mammals". BioEssays. 31 (8): 892–902. doi:10.1002/bies.200900038. PMC 2949267. PMID 19609969.
- ^ Chuai M, Zeng W, Yang X, Boychenko V, Glazier JA, Weijer CJ (2006). "Cell movement during chick primitive streak formation". Dev. Biol. 296 (1): 137–49. doi:10.1016/j.ydbio.2006.04.451. PMC 2556955. PMID 16725136.
- ^ Chuai M, Weijer CJ (2008). "The mechanisms underlying primitive streak formation in the chick embryo.". Current Topics in Developmental Biology. Vol. 81. pp. 135–56. doi:10.1016/S0070-2153(07)81004-0. ISBN 978-0-12-374253-7. PMID 18023726.
- ^ Grapin-Botton, A.; Constam, D. (2007). "Evolution of the mechanisms and molecular control of endoderm formation". Mechanisms of Development. 124 (4): 253–78. doi:10.1016/j.mod.2007.01.001. PMID 17307341. S2CID 16552755.
- ^ a b c Turner, David A.; Rué, Pau; Mackenzie, Jonathan P.; Davies, Eleanor; Martinez Arias, Alfonso (2014-01-01). "Brachyury cooperates with Wnt/β-catenin signalling to elicit primitive-streak-like behaviour in differentiating mouse embryonic stem cells". BMC Biology. 12 63. doi:10.1186/s12915-014-0063-7. ISSN 1741-7007. PMC 4171571. PMID 25115237.
- ^ a b Warmflash, Aryeh; Sorre, Benoit; Etoc, Fred; Siggia, Eric D; Brivanlou, Ali H (2014). "A method to recapitulate early embryonic spatial patterning in human embryonic stem cells". Nature Methods. 11 (8): 847–854. doi:10.1038/nmeth.3016. PMC 4341966. PMID 24973948.
- ^ a b Etoc, Fred; Metzger, Jakob; Ruzo, Albert; Kirst, Christoph; Yoney, Anna; Ozair, M. Zeeshan; Brivanlou, Ali H.; Siggia, Eric D. (2016). "A Balance between Secreted Inhibitors and Edge Sensing Controls Gastruloid Self-Organization". Developmental Cell. 39 (3): 302–315. doi:10.1016/j.devcel.2016.09.016. PMC 5113147. PMID 27746044.
- ^ a b Brink, Susanne C. van den; Baillie-Johnson, Peter; Balayo, Tina; Hadjantonakis, Anna-Katerina; Nowotschin, Sonja; Turner, David A.; Arias, Alfonso Martinez (2014-11-15). "Symmetry breaking, germ layer specification and axial organisation in aggregates of mouse embryonic stem cells". Development. 141 (22): 4231–4242. doi:10.1242/dev.113001. ISSN 0950-1991. PMC 4302915. PMID 25371360.
- ^ a b c Turner, David Andrew; Glodowski, Cherise R.; Luz, Alonso-Crisostomo; Baillie-Johnson, Peter; Hayward, Penny C.; Collignon, Jérôme; Gustavsen, Carsten; Serup, Palle; Schröter, Christian (2016-05-13). "Interactions between Nodal and Wnt signalling Drive Robust Symmetry Breaking and Axial Organisation in Gastruloids (Embryonic Organoids)". bioRxiv 10.1101/051722.
- ^ a b c Turner, David; Alonso-Crisostomo, Luz; Girgin, Mehmet; Baillie-Johnson, Peter; Glodowski, Cherise R.; Hayward, Penelope C.; Collignon, Jérôme; Gustavsen, Carsten; Serup, Palle (2017-01-31). "Gastruloids develop the three body axes in the absence of extraembryonic tissues and spatially localised signalling". bioRxiv 10.1101/104539.
- ^ Beccari, Leonardo; Moris, Naomi; Girgin, Mehmet; Turner, David A.; Baillie-Johnson, Peter; Cossy, Anne-Catherine; Lutolf, Matthias P.; Duboule, Denis; Arias, Alfonso Martinez (October 2018). "Multi-axial self-organization properties of mouse embryonic stem cells into gastruloids". Nature. 562 (7726): 272–276. Bibcode:2018Natur.562..272B. doi:10.1038/s41586-018-0578-0. ISSN 0028-0836. PMID 30283134. S2CID 52915553.
- ^ Turner, David A.; Girgin, Mehmet; Alonso-Crisostomo, Luz; Trivedi, Vikas; Baillie-Johnson, Peter; Glodowski, Cherise R.; Hayward, Penelope C.; Collignon, Jérôme; Gustavsen, Carsten (2017-11-01). "Anteroposterior polarity and elongation in the absence of extra-embryonic tissues and of spatially localised signalling in gastruloids: mammalian embryonic organoids". Development. 144 (21): 3894–3906. doi:10.1242/dev.150391 (inactive 17 October 2025). ISSN 0950-1991. PMC 5702072. PMID 28951435.
{{cite journal}}: CS1 maint: DOI inactive as of October 2025 (link) - ^ "In vitro fertilization (IVF) - Mayo Clinic". www.mayoclinic.org. Retrieved 2022-04-11.
- ^ Asplund, Kjell (2020). "Use of in vitro fertilization—ethical issues". Upsala Journal of Medical Sciences. 125 (2): 192–199. doi:10.1080/03009734.2019.1684405. ISSN 2000-1967. PMC 7721055. PMID 31686575. S2CID 207896932.
- ^ Davis, Caitlin (2019-03-01). "The Boundaries of Embryo Research: Extending the Fourteen-Day Rule". Journal of Bioethical Inquiry. 16 (1): 133–140. doi:10.1007/s11673-018-09895-w. ISSN 1872-4353. PMID 30635823. S2CID 58643344.
Bibliography
[edit]- Arnold, Sebastian J.; Robertson, Elizabeth J. (2009). "Making a commitment: cell lineage allocation and axis patterning in the early mouse embryo". Nat. Rev. Mol. Cell Biol. 10 (2): 91–103. doi:10.1038/nrm2618. PMID 19129791. S2CID 94174.

- Catala, Martin (2005). "Embryology of the Spine and Spinal Cord". In Tortori-Donati, Paolo; et al. (eds.). Pediatric Neuroradiology: Brain. Springer. ISBN 978-3-540-41077-5.
- Ereskovsky, Alexander V. (2010). The Comparative Embryology of Sponges. Springer. ISBN 978-90-481-8574-0.
- Gilbert, Scott F. (2010). Developmental Biology (Ninth ed.). Sinauer Associates. ISBN 978-0-87893-558-1.
- Hall, Brian Keith (1998). "8.3.3 The gastrula and gastrulation". Evolutionary developmental biology (2nd ed.). The Netherlands: Kluwer Academic Publishers. ISBN 978-0-412-78580-1.
- Harrison, Lionel G. (2011). The Shaping of Life: The Generation of Biological Pattern. Cambridge University Press. ISBN 978-0-521-55350-6.
- McGeady, Thomas A., ed. (2006). "Gastrulation". Veterinary embryology. Wiley-Blackwell. ISBN 978-1-4051-1147-8.
- Mundlos, Stefan (2009). "Gene action: developmental genetics". In Speicher, Michael; et al. (eds.). Vogel and Motulsky's Human Genetics: Problems and Approaches (4th ed.). Springer. doi:10.1007/978-3-540-37654-5. ISBN 978-3-540-37653-8.
- Tam, Patrick P.L.; Behringer, Richard R. (1997). "Mouse gastrulation: the formation of a mammalian body plan". Mech. Dev. 68 (1–2): 3–25. doi:10.1016/S0925-4773(97)00123-8. PMID 9431800. S2CID 14052942.

Further reading
[edit]- Baron, Margaret H. (2001). "Embryonic Induction of Mammalian Hematopoiesis and Vasculogenesis". In Zon, Leonard I. (ed.). Hematopoiesis: a developmental approach. Oxford University Press. ISBN 978-0-19-512450-7.
- Cullen, K.E. (2009). "embryology and early animal development". Encyclopedia of life science, Volume 2. Infobase. ISBN 978-0-8160-7008-4.
- Forgács, G.; Newman, Stuart A. (2005). "Cleavage and blastula formation". Biological physics of the developing embryo. Cambridge University Press. Bibcode:2005bpde.book.....F. ISBN 978-0-521-78337-8.
- Forgács, G.; Newman, Stuart A. (2005). "Epithelial morphogenesis: gastrulation and neurulation". Biological physics of the developing embryo. Cambridge University Press. Bibcode:2005bpde.book.....F. ISBN 978-0-521-78337-8.
- Hart, Nathan H.; Fluck, Richard A. (1995). "Epiboly and Gastrulation". In Capco, David (ed.). Cytoskeletal mechanisms during animal development. Academic Press. ISBN 978-0-12-153131-7.
- Knust, Elizabeth (1999). "Gastrulation movements". In Birchmeier, Walter; Birchmeier, Carmen (eds.). Epithelial Morphogenesis in Development and Disease. CRC Press. pp. 152–153. ISBN 978-90-5702-419-1.
- Kunz, Yvette W. (2004). "Gastrulation". Developmental biology of Teleost fishes. Springer. ISBN 978-1-4020-2996-7.
- Nation, James L., ed. (2009). "Gastrulation". Insect physiology and biochemistry. CRC Press. ISBN 978-0-8493-1181-9.
- Ross, Lawrence M.; Lamperti, Edward D., eds. (2006). "Human Ontogeny: Gastrulation, Neurulation, and Somite Formation". Atlas of anatomy: general anatomy and musculoskeletal system. Thieme. ISBN 978-3-13-142081-7.
- Sanes, Dan H.; et al. (2006). "Early embryology of metazoans". Development of the nervous system (2nd ed.). Academic Press. pp. 1–2. ISBN 978-0-12-618621-5.
- Stanger, Ben Z.; Melton, Douglas A. (2004). "Development of Endodermal Derivatives in the Lungs, Liver, Pancreas, and Gut". In Epstein, Charles J.; et al. (eds.). Inborn errors of development: the molecular basis of clinical disorders of morphogenesis. Oxford University Press. ISBN 978-0-19-514502-1.
External links
[edit]Gastrulation
View on GrokipediaFundamentals
Definition and Core Process
Gastrulation is the phase of early embryonic development in which the blastula—a hollow sphere consisting of a single layer of cells enclosing a blastocoel—is transformed into the gastrula, a multilayered structure featuring three primary germ layers: ectoderm, mesoderm, and endoderm. This reorganization occurs through coordinated cellular behaviors that internalize specific cell populations, establishing the basic body plan of the embryo.[1][7] The core process involves distinct morphogenetic movements, such as invagination (inward folding of the cell sheet), ingression (individual cells delaminating and migrating inward), epiboly (expansion and thinning of the outer layer), involution (cells rolling inward at the margin), and convergence-extension (tissue narrowing and elongation). These actions, often initiated at a specific site like the vegetal pole in invertebrates or the primitive streak in vertebrates, rely on epithelial-to-mesenchymal transitions to enable cells to ingress and displace into interior positions, forming the inner endoderm and middle mesoderm beneath the protective ectoderm.[1][8] Germ layer formation during gastrulation positions ectoderm as the outermost layer destined for epidermal and neural tissues, mesoderm as the intermediate layer for connective, muscular, and vascular structures, and endoderm as the innermost layer for digestive and respiratory epithelia. This trilaminar organization, conserved across metazoans despite mechanistic variations, also orients embryonic axes (e.g., dorsal-ventral, anterior-posterior) critical for further differentiation.[1][8]Etymology
The term gastrulation was coined by German biologist Ernst Haeckel in 1872, in his work Die Kalkschwämme (Biology of Calcareous Sponges), to describe the embryonic process leading to the formation of the primitive gut.[9][10] It derives from the Ancient Greek gastḗr (γαστήρ), meaning "stomach" or "belly," reflecting the invagination and reorganization that establish the archenteron, the precursor to the digestive tract.[11][12] The noun gastrula, denoting the resulting embryonic stage, combines this root with the Neo-Latin diminutive suffix -ula.[11]Biological and Evolutionary Importance
Gastrulation establishes the foundational three primary germ layers—ectoderm, mesoderm, and endoderm—which differentiate into all major tissue types and organs in triploblastic animals, thereby initiating the morphogenesis of the body plan.[1] This process reorganizes the blastula's uniform cell layer into a structured gastrula, specifying and positioning these layers to enable subsequent organogenesis and neurulation.[1] By generating spatial organization, including anterior-posterior and dorsal-ventral axes, gastrulation ensures directional development critical for embryonic viability.[1] Biologically, gastrulation's cell rearrangements, such as invagination and convergence-extension, are essential for transitioning from a simple spherical embryo to a complex form capable of supporting differentiated functions, with disruptions often resulting in lethal developmental arrest or congenital anomalies.[13] The formation of the mesoderm, in particular, allows for the development of supportive structures like muscles and circulatory systems, underscoring gastrulation's role in enabling multicellular complexity beyond mere cell proliferation.[14] Evolutionarily, gastrulation exhibits deep conservation across metazoans, with core morphogenetic movements and regulatory networks—such as those involving BMP, Wnt, and Nodal signaling—predating the protostome-deuterostome divergence, indicative of shared ancestry in bilaterian lineages.[15] While modes vary (e.g., spiraling cleavage in protostomes versus radial in deuterostomes), the underlying genetic toolkit for germ layer specification remains homologous, suggesting that evolutionary innovations primarily modified deployment rather than inventing new mechanisms de novo.[16] This conservation highlights gastrulation as a pivotal innovation in metazoan evolution, facilitating the scalable complexity of animal body plans from simple invertebrates to vertebrates.[17]Morphogenetic Mechanisms
Fundamental Cell Movements
Gastrulation relies on a set of fundamental cell movements that reorganize the blastula into a multilayered gastrula, establishing the ectoderm, mesoderm, and endoderm germ layers through coordinated internalization, spreading, and reshaping of cell sheets. These movements, conserved across metazoans but varying in execution, include invagination, involution, ingression, delamination, epiboly, and convergence-extension, often involving changes in cell shape, adhesion, and motility driven by cytoskeletal dynamics and extracellular matrix interactions.[18][19] In vertebrates, these processes typically initiate at a specified dorsal region, such as the blastopore lip in amphibians, where cells undergo epithelial rearrangements to internalize presumptive mesendoderm.[19] Invagination entails the inward folding of an epithelial cell sheet, akin to dimpling a flexible membrane, which creates a pouch-like structure that contributes to cavity formation or tissue internalization. This movement is prominent in sea urchin embryos, where primary mesenchyme cells invaginate at the vegetal pole to form the archenteron, and in amphibian gastrulation at the dorsal blastopore lip.[20][21] It relies on apical constriction of cells via actomyosin contractility, reducing surface area and driving buckling without requiring widespread cell migration.[22] Involution involves the inward rolling of a coherent epithelial layer over an edge, such as cells from the marginal zone folding under the blastocoel roof in frog embryos to displace presumptive endoderm and mesoderm internally. This movement, distinct from invagination by its sheet-like migration around a lip or rim, extends the involuting layer to line embryonic cavities and is regulated by planar cell polarity and Wnt signaling.[19][23] Involution facilitates the transition from superficial to deep layers, ensuring proper germ layer positioning.[24] Ingression occurs when individual epithelial cells detach from a sheet, undergo epithelial-to-mesenchymal transition (EMT), and migrate independently into the interior, often as mesenchymal cells that later reintegrate into tissues. This is exemplified by primary mesenchyme ingression in echinoderms, where cells bottle-shape and delaminate via downregulation of E-cadherin and upregulation of snail transcription factors, contributing to skeleton formation.[20][22] Ingression allows dispersed cell populations to populate spaces between germ layers, contrasting with collective sheet movements.[18] Delamination separates cells from an epithelial layer into basal or apical sublayers, or as individuals, without extensive migration, often preceding other movements to refine tissue stratification. Seen in avian primitive streak formation, it involves partial EMT and is crucial for generating multilayered epithelia from monolayers.[21][25] Epiboly describes the thinning and spreading of an external epithelial sheet to envelop the embryo, driven by radial intercalation and membrane expansion, as in zebrafish where blastoderm cells expand over the yolk cell. This movement increases surface area coverage, coordinating with internalization to enclose yolk or internalize cells.[18][24] Convergence-extension reshapes tissues by cells converging toward the midline while intercalating and elongating perpendicularly, narrowing and lengthening the embryo along the anteroposterior axis. Powered by polarized protrusions and non-canonical Wnt/PCP signaling, it is essential in vertebrate notochord formation and amplifies axial structures during gastrulation.[18][22] These movements collectively ensure efficient germ layer segregation and embryonic axis establishment, with variations reflecting evolutionary adaptations.[26]Germ Layer Formation and Gastrula Structure
Gastrulation establishes the three primary germ layers—ectoderm, mesoderm, and endoderm—through coordinated cellular rearrangements that convert the blastula's single epithelial layer into a trilaminar structure.[27] The ectoderm comprises the presumptive outer layer, destined to form epidermal tissues and the nervous system; the mesoderm arises as an intermediate layer generating connective tissues, muscles, and circulatory elements; and the endoderm internalizes to line the primitive digestive cavity, giving rise to gut and associated organs.[5] These layers emerge via processes such as ingression and involution, where cells from the surface layer migrate inward, displacing or supplementing prior hypoblast-like cells in vertebrates.[28] A key mechanism in germ layer formation is the epithelial-to-mesenchymal transition (EMT), during which epithelial cells of the epiblast lose cell-cell adhesion, apical-basal polarity, and acquire migratory mesenchymal properties, enabling their contribution to mesoderm and endoderm.[28] This transition, regulated by signaling pathways like BMP and Wnt, allows cells to ingress through structures such as the primitive streak in amniotes or the blastopore in other taxa, positioning mesodermal precursors laterally and endodermal cells medially.[29] Post-EMT, some cells undergo mesenchymal-to-epithelial transition to reform epithelial sheets, stabilizing layer identities.[28] The resulting gastrula exhibits a characteristic structure with the ectoderm enveloping the embryo externally, the mesoderm interspersed as loosely organized sheets or cavities, and the endoderm forming a continuous internal lining around the archenteron—a fluid-filled cavity connected to the exterior via the blastopore.[5] This blastopore marks the site of cell internalization and defines embryonic axes, with the archenteron serving as the precursor to the gut.[27] In triploblastic animals, the trilaminar organization establishes the foundational body plan, with germ layer positions reflecting conserved morphogenetic principles across metazoans.[30]Molecular Regulation
Key Signaling Pathways
The molecular orchestration of gastrulation relies on conserved signaling pathways that integrate environmental cues, cell-cell interactions, and transcriptional responses to drive germ layer specification and tissue morphogenesis. Principal pathways include members of the transforming growth factor-β (TGF-β) superfamily such as Nodal, which initiates primitive streak formation; Wnt/β-catenin signaling, which promotes posterior mesoderm identity; bone morphogenetic protein (BMP) signaling, which establishes ventral fates; and fibroblast growth factor (FGF) signaling, which supports cell migration and survival during involution.[1][31] These pathways exhibit spatiotemporal dynamics, with mutual antagonism and synergy ensuring precise patterning, as disruptions in any one can arrest gastrulation progression.[32][33] Nodal signaling, part of the TGF-β family, is pivotal for inducing the primitive streak and specifying mesendodermal progenitors in vertebrates. In mice and amphibians, Nodal ligands secreted from extraembryonic or vegetal regions activate Smad2/3 transcription factors via receptors like Alk4/5/7, promoting brachyury (T) expression and epiblast competence for gastrulation entry as early as embryonic day 6.5 in mice.[1][34] Antagonists such as Lefty and Cerberus refine Nodal gradients to prevent ectopic induction, with genetic knockouts demonstrating that Nodal-null embryos fail to form mesoderm by E7.5.[33] Wnt/β-catenin signaling cooperates with Nodal to posteriorize the embryo and stabilize mesodermal fates during streak formation. Canonical Wnt ligands (e.g., Wnt3 in mice) inhibit GSK3β, leading to β-catenin nuclear accumulation and activation of targets like Tbx6 and Fgf8, which are essential for paraxial mesoderm development; in zebrafish, maternal Wnt8 reinforces involution at shield stage (5.25 hours post-fertilization).[31][35] Non-canonical Wnt pathways, such as Wnt/PCP, further regulate convergent extension movements by polarizing cytoskeletal dynamics via RhoA and JNK.[36] BMP signaling, also TGF-β-related, patterns the dorsoventral axis by promoting ventral and lateral mesoderm while being antagonized dorsally by secreted inhibitors like Chordin, Noggin, and Follistatin from the organizer region. In Xenopus, BMP4 gradients peak ventrally at stage 10.5, driving ventrolateral fate via Smad1/5/8; loss-of-function studies show uniform dorsalization in BMP receptor mutants.[31][1] Interactions with Wnt and Nodal modulate BMP thresholds, ensuring compartmentalized fates during gastrulation.[32] FGF signaling sustains mesodermal gene expression and facilitates epithelial-mesenchymal transition (EMT) and migration post-involution. FGF8 and FGF4 from the primitive streak activate ERK/MAPK cascades, inducing snail and twist for EMT while preventing apoptosis; in chick embryos, FGF inhibition halts head process extension by HH stage 4.[31][37] Crosstalk with BMP inhibits neural induction, reinforcing ectodermal competence boundaries.[35] These pathways are evolutionarily conserved yet modulated by context-specific regulators, with quantitative models revealing threshold-dependent responses that underpin robust gastrulation across taxa.[38]Genetic and Epigenetic Controls
Genetic regulation of gastrulation is mediated by hierarchical gene regulatory networks (GRNs) that coordinate transcription factor activity with extracellular signaling to specify germ layers and drive morphogenetic movements.[39] These networks integrate inputs from pathways like Wnt, Nodal, and BMP to activate lineage-specific genes, with conserved transcription factors such as Brachyury (T-box family) essential for mesoderm induction and axial elongation in vertebrates.[16] GATA4/5/6 factors similarly function across species to regulate endoderm and mesoderm formation, binding enhancers that respond to nodal signaling for timely gene activation during cell ingression.[40] Disruptions in these GRNs, as seen in Trim71 mutants, lead to dysregulated expression of mesodermal genes, impairing gastrulation progression and highlighting the precision of transcriptional hierarchies. A core set of approximately 75 transcription factors forms a conserved regulatory module across mammalian gastrulation, controlling epiblast delamination and primitive streak formation through shared cis-regulatory syntax.[41] This module, identified via single-cell atlases, underscores evolutionary stability despite species-specific timing, with factors like Sox17 and Foxa2 enforcing endodermal identity post-ingression.[42] In protostomes and deuterostomes, analogous TFs such as snail and twist in Drosophila orchestrate mesoderm invagination, reflecting modular GRN conservation amid divergent morphologies.[43] Epigenetic controls complement genetic programs by dynamically remodeling chromatin to permit or restrict access to developmental loci during the pluripotent-to-multipotent transition.[44] DNA methylation patterns shift globally in mouse epiblasts prior to gastrulation, with demethylation at bivalent promoters enabling poised expression of lineage genes like those in the T/Brachyury network.[45] Histone modifications, including H3K27me3 enrichment by Polycomb repressive complexes at CpG islands, maintain repression of non-gastrulation fates, while Trithorax-mediated H3K4me3 activates mesendodermal enhancers as cells ingress.[46] These marks interact cooperatively; for instance, low DNA methylation correlates with active histone acetylation, facilitating rapid transcriptional responses to signaling gradients.[47] Chromatin accessibility assays reveal gastrulation-stage surges in open regions near TFs like Gata6, correlating with nucleosome repositioning and variant histone incorporation to stabilize germ layer identities.[48] In human organoid models mimicking gastrulation, epigenetic profiling shows conserved principles of H3K4me3/H3K27me3 bivalency resolution, directing germ cell specification alongside somatic lineages.[49] Such reprogramming ensures irreversible commitment, as evidenced by elevated epigenetic age minima at gastrulation onset, underscoring its role as a causal bottleneck for developmental fidelity.[50]Comparative Gastrulation Across Taxa
Protostomes Versus Deuterostomes
A primary distinction between protostomes and deuterostomes arises during gastrulation with the fate of the blastopore, the initial opening formed in the embryo. In protostomes, the blastopore develops into the mouth, reflecting a "first mouth" developmental pattern.[2][51] In contrast, deuterostomes exhibit the blastopore becoming the anus, with the mouth forming secondarily from a separate opening.[2][51] This difference, observed consistently across major taxa such as annelids and mollusks for protostomes versus echinoderms and chordates for deuterostomes, underscores divergent strategies in gut formation and anterior-posterior axis establishment./13%3A_Module_10-_Animal_Diversity/13.21%3A_Embryological_Development) Preceding gastrulation, cleavage patterns further differentiate the groups, influencing subsequent morphogenetic movements. Protostomes typically undergo spiral cleavage, where daughter cells divide at oblique angles, resulting in a skewed arrangement relative to underlying cells and promoting determinate development with early cell fate commitment./13%3A_Module_10-_Animal_Diversity/13.21%3A_Embryological_Development)[51] Deuterostomes, however, feature radial cleavage, with divisions parallel or perpendicular to the polar axis, aligning cells in tiers and supporting indeterminate development where early blastomeres retain regulative potential./13%3A_Module_10-_Animal_Diversity/13.21%3A_Embryological_Development)[51] These patterns set the stage for gastrulation, where protostome invagination often emphasizes mesendoderm migration toward the blastopore to form the mouth-first archenteron, while deuterostome processes prioritize radial symmetry in germ layer reorganization. Coelom formation, which emerges post-gastrulation from mesodermal precursors, also varies mechanistically between the clades. Protostomes employ schizocoely, wherein the coelom arises by splitting solid mesodermal masses into cavities.[52] Deuterostomes utilize enterocoely, involving outgrowths or pouches from the archenteron wall that pinch off to form coelomic spaces.[52] These modes correlate with the blastopore's role: protostome schizocoely aligns with mesoderm originating laterally to the blastopore-derived mouth, whereas deuterostome enterocoely ties to mesodermal evagination near the blastopore-anus site.[52] Such differences impact body plan flexibility, with deuterostome regulative mechanisms allowing greater evolutionary adaptability in organ positioning compared to the more fixed protostome mosaic development./13%3A_Module_10-_Animal_Diversity/13.21%3A_Embryological_Development) While these traits define the protostome-deuterostome dichotomy, exceptions exist, such as variable blastopore fates in some protostomes, challenging strict delineations and prompting ongoing phylogenetic reevaluations based on molecular data./13%3A_Module_10-_Animal_Diversity/13.21%3A_Embryological_Development) Nonetheless, gastrulation's morphological signatures remain key for classifying bilaterian animals, informing evolutionary divergence estimated around 550-600 million years ago during the Ediacaran-Cambrian transition.[51]Invertebrate Models: Sea Urchins
Sea urchins, particularly species like Strongylocentrotus purpuratus, serve as a premier invertebrate model for studying deuterostome gastrulation due to their transparent embryos, external development, and ability to produce large numbers of synchronously cleaving eggs that are easily fertilized in vitro.[53] These features have facilitated over 150 years of research, enabling detailed observation of cellular and molecular events without invasive techniques.[53] As deuterostomes, sea urchins exhibit gastrulation patterns homologous to vertebrates, including radial cleavage and formation of the anus from the blastopore, contrasting with protostome mouth-first development.[54] Gastrulation commences at the blastula stage, around 9-10 hours post-fertilization at 15°C, with the vegetal plate cells initiating primary mesenchyme cell (PMC) ingression.[55] PMCs, derived from micromeres at the fourth cleavage, undergo epithelial-to-mesenchymal transition (EMT), detach from the vegetal epithelium via apical constriction and loss of cell-cell adhesions, and ingress into the blastocoel using filopodia for migration.[55] These cells, numbering about 20-40 per embryo, settle at the animal pole and vegetal ridge to secrete the larval skeleton, guided by chemotactic signals such as VEGF and FGF.[56] Disruption of these pathways, as shown in perturbation experiments, arrests PMC migration and skeletal formation.[56] Concurrent with or following PMC ingression, the remaining vegetal plate undergoes primary invagination to form the archenteron, the precursor to the gut.[55] This involves convergent extension of the endodermal epithelium, where bottle cells at the vegetal margin constrict apically, driving a purse-string-like folding, while myosin contractility and extracellular matrix remodeling facilitate tissue bending without requiring oriented cell division.[55] The archenteron then elongates through secondary mesenchyme cell (SMC) addition and filopodial traction, propelled by polarized actomyosin flows and adhesion to the blastocoel roof.[57] By late gastrulation, around 24-30 hours, the archenteron tip contacts the oral ectoderm, establishing the mouth via fusion, while SMCs contribute to coelomic pouches and pigment cells.[55] Gene regulatory networks (GRNs) orchestrate these movements, with transcription factors like ets1, alx1, and tbr specifying PMC fate and EMT, activated downstream of Delta-Notch signaling from micromeres.[58] Archenteron invagination relies on goosecoid and brachyury for epithelial integrity and elongation, modulated by Nodal, BMP, and Wnt pathways that pattern the vegetal-oral axis.[57] These networks, mapped through perturbation and cis-regulatory analysis, reveal robust linkages between gene expression and morphogenesis, as PMCs ingress even in isolated cells under specific conditions.[58] Sea urchin studies have thus illuminated conserved mechanisms, such as EMT and convergent extension, applicable to vertebrate gastrulation while highlighting echinoderm-specific adaptations like skeletal biomineralization.[55]Vertebrate Models: Amphibians
In amphibians such as Xenopus laevis, gastrulation transforms the spherical blastula into a multilayered gastrula through precise cell rearrangements that establish the ectoderm, mesoderm, and endoderm germ layers, with the process initiating at stage 10 (approximately 8-10 hours post-fertilization at 23°C). The dorsal blastopore lip emerges first as a thickened region in the marginal zone, where bottle cells—narrow, flask-shaped cells derived from superficial and deep marginal layers—undergo epithelial-to-mesenchymal transition (EMT) and ingress to initiate involution, forming the blastopore rim that serves as the portal for mesendodermal internalization.[59][60] Presumptive endoderm from the vegetal hemisphere and mesoderm from the equatorial marginal zone involute through this lip, displacing the blastocoel cavity and expanding the archenteron, a primitive gut cavity lined by endoderm.[61] Coordinated morphogenetic movements drive layer formation: epiboly thins and spreads the prospective ectodermal animal cap vegetally over the embryo, covering up to 60-70% of the surface by mid-gastrulation; involution directs deep marginal cells inward along the blastocoel roof via directed migration and traction forces; and convergence-extension intercalates mediolateral cells to elongate the anteroposterior axis by 2-3 fold while narrowing the mediolateral dimension, generating tensile forces that peak at 1.5-2.0 μN during early gastrulation and exceed 4.0 μN by neurulation onset.[62][63] These rearrangements position mesoderm between ectoderm and endoderm, with dorsal mesoderm fating to notochord and somites, ventral to blood and lateral plate, and lateral to pronephros, as traced by vital dye labeling experiments showing 80-90% fidelity in cell fate mapping.[60] The Spemann-Mangold organizer, identified in newt embryos in 1924 and homologous in Xenopus at the dorsal blastopore lip, functions as a signaling center by secreting BMP antagonists like chordin and noggin, which dorsalize ventral mesoderm and induce neural ectoderm in overlying tissues, as demonstrated by transplantation assays inducing secondary axes with 70-100% efficiency depending on graft timing.[64][65] Non-canonical Wnt/PCP signaling, involving RhoA GTPases and myosin II contractility, mediates convergence-extension independently of canonical Wnt/β-catenin pathways, with disruptions via dominant-negative constructs reducing extension by 50% or more in explants.[62] Cell adhesion molecules such as cadherins and integrins facilitate differential migration, with calcium-dependent adhesion gradients ensuring tissue integrity during shear stresses up to 10-20 dyn/cm².[66] EMT underlies bottle cell formation and involuting mesoderm motility in amphibians, enabling mesenchymal migration before reversion to epithelial states in target tissues.[67]Vertebrate Models: Amniotes and Mammals
In amniote embryos, gastrulation proceeds through the formation of a primitive streak, a transient structure that serves as the site for epithelial-to-mesenchymal transition (EMT) and ingression of epiblast cells to generate mesoderm and definitive endoderm.[42] This process establishes bilateral symmetry and the anteroposterior axis, with conserved roles for Wnt, Nodal, BMP, and FGF signaling pathways across amniotes.[68] The chick embryo provides a primary model for yolk-rich amniotes, where the epiblast and hypoblast form prior to streak initiation around 15 hours post-laying.[69] In chicks, primitive streak formation begins posteriorly near Koller's sickle, a region of cell intercalation that induces streak elongation anteriorly through epithelial rearrangements and proliferation, reaching full extension by Hamburger-Hamilton stage 4 (approximately 18-19 hours of incubation).[70] The hypoblast, an extraembryonic layer, positions the streak by secreting antagonists such as Cerberus, which inhibit Nodal and BMP signaling to prevent ectopic streak formation.00318-0) Ingression occurs via EMT at the streak, with cells migrating laterally and anteriorly to displace hypoblast-derived endoderm, forming paraxial mesoderm, lateral plate, and axial structures like Hensen's node at the anterior terminus.00041-1) Cell movements include midline convergence and counterrotational flows, driven by polarized intercalations.[42] Mammalian gastrulation, exemplified by the mouse, shares the primitive streak mechanism but adapts to a yolk-poor environment, initiating around embryonic day 6.25 (E6.25) at the distal-posterior epiblast without a hypoblast equivalent.[33] The anterior visceral endoderm (AVE) migrates posteriorly to break symmetry and restrict streak formation, guided by Nodal inhibition via Lefty and Cer1.[33] Streak extension to E7.5 involves Wnt3-mediated EMT and proliferation, with ingressing cells forming mesenchymal mesoderm that spreads as wings between epiblast and visceral endoderm.[42] Definitive endoderm emerges via subsequent mesenchymal-to-epithelial transition (MET) and intercalation into extraembryonic endoderm by E8.75, while mesoderm diversifies into axial, paraxial, intermediate, and lateral subtypes based on Hox gene expression and signaling gradients.[33] Unlike chicks, mouse ingression lacks pronounced convergent extension, relying more on local EMT without large-scale posterior convergence.[42] Key differences between avian and mammalian models include yolk dependency—chick gastrulation occurs on a yolky disc with hypoblast involvement, whereas mouse proceeds in a cup-shaped structure with AVE-directed patterning—and dynamic flows, with chicks exhibiting stronger rotational movements absent in mice.[68] Both, however, demonstrate regulative capacity, as evidenced by streak reformation after perturbation.[68] In humans, primitive streak appearance around day 14 post-fertilization mirrors mouse timing proportionally, though direct observation is limited.[42]Evolutionary Perspectives and Debates
Historical Research Milestones
In 1817, Christian Heinrich Pander published observations on chick embryo development, identifying the formation of three distinct germ layers—ectoderm, mesoderm, and endoderm—from the blastoderm, providing the initial empirical basis for recognizing gastrulation as the morphogenetic process that establishes these foundational tissues.[71] This trilaminar organization was later generalized by Karl Ernst von Baer, who in 1828 extended the germ layer concept to all vertebrates through comparative studies, emphasizing conserved developmental patterns across species and linking them to organ formation during gastrulation.[72] Ernst Haeckel coined the terms "gastrula" and "gastrulation" in 1872 while studying sponge development, describing the process as an invagination forming a primitive gut-like structure analogous across metazoans, and in 1874 proposed the gastraea hypothesis positing a hypothetical ancestor with gastrula-like organization to explain evolutionary conservation of germ layer origins.[42] These conceptual advances shifted focus from descriptive anatomy to dynamic cellular rearrangements, though Haeckel's recapitulationist interpretations later faced scrutiny for overemphasizing phylogenetic regression in ontogeny. Experimental embryology advanced in the early 20th century with vital dye staining techniques; Walter Vogt applied neutral red and Nile blue dyes to amphibian blastulae in 1923–1929, producing the first comprehensive fate maps that traced presumptive germ layer territories and quantified involution movements during gastrulation, revealing mesoderm's dual superficial and deep origins.[72] Concurrently, in 1924, Hans Spemann and Hilde Mangold demonstrated that the dorsal blastopore lip in amphibian gastrulae acts as an organizer, inducing secondary axes and neural tissue via host-graft transplants, a discovery earning Spemann the 1935 Nobel Prize and establishing induction as a causal mechanism coordinating germ layer specification and morphogenesis.[73] Mid-20th-century refinements included Johannes Holtfreter's 1943–1944 exogastrulation experiments on amphibian embryos, which dissociated mesoderm induction from morphogenetic movements, showing that presumptive endoderm and mesoderm could self-differentiate in vitro while ectoderm required signals, thus clarifying gastrulation's regulative versus mosaic elements.[74] These milestones, grounded in direct observation and manipulation, resolved early debates on preformation versus epigenesis, privileging causal interactions over rigid germ layer homologies.[72]Debates on Germ Layer Homology and Origins
The evolutionary origins of endoderm and mesoderm, the inner germ layers formed during gastrulation, have been debated for decades, with uncertainty surrounding their ancestral specification and interrelationship in early metazoans. Comparative analyses of transcription factors such as GATA4-6, Twist, Snail, and Brachyury indicate that these genes, which regulate germ layer formation in bilaterians, likely evolved from roles in basic cellular processes like adhesion and motility, supporting a gradual emergence rather than abrupt invention.[75] In diploblastic cnidarians, the inner cell layer expresses homologs of bilaterian endomesodermal markers, prompting debate over whether this reflects homology to endoderm or convergent co-option of ancient regulatory networks.[76] Sponge development provides limited support for germ layer homology, as embryonic cell layers in species like Amphimedon queenslandica exhibit transient patterning without fixed identities akin to eumetazoan ectoderm, endoderm, or mesoderm; instead, cells transdifferentiate flexibly post-embryogenesis. GATA expression in larval inner layers hints at a shared endomesodermal precursor, but overall evidence favors gastrulation as an eumetazoan innovation after the poriferan divergence around 600-800 million years ago.[17] Within triploblastic bilaterians, germ layer homology is inferred from conserved topological arrangements and gene regulatory networks, despite divergent cellular origins—such as endomesodermal ingression in many protostomes versus inductive separation in deuterostomes. Shifts in germ layer contributions, exemplified by variable neural crest versus mesodermal origins of vertebrate skull sutures (e.g., coronal suture as neural crest-mesoderm in mice but mesoderm-mesoderm in zebrafish), necessitate homology criteria beyond origin, including inductive signaling centers and evolutionary continuity.[77][75] Emerging molecular data challenge rigid germ layer boundaries, as ectodermal gonopores in xenacoelomorphs (basal bilaterians) deploy endodermal hindgut markers like caudal, brachyury, and Wnt signaling, suggesting functional homology to bilaterian anuses despite disparate origins and implying that strict endoderm-ectoderm distinctions may obscure deeper developmental equivalences. Such findings underscore ongoing debates on whether triploblasty arose via endodermal splitting or de novo mesoderm insertion, with implications for reconstructing the urmetazoan body plan.[78]Challenges from Non-Model Organisms
Non-model organisms, encompassing taxa beyond standard laboratory species such as Drosophila melanogaster, Xenopus laevis, and Mus musculus, exhibit gastrulation processes that diverge markedly in morphology, cell movements, and molecular regulation, complicating extrapolations from model systems. For instance, in cnidarians like Nematostella vectensis, embryos typically undergo invagination for endoderm internalization, but experimental cell dissociation induces a switch to multipolar ingression or cavitation, demonstrating context-dependent plasticity not routinely observed in bilaterian models.[26] Similarly, arthropods such as Chironomus riparius favor ingression over the invagination seen in Drosophila, with maternal factors like Fog/t48 mRNA dictating the mode, highlighting how subtle genetic variations yield alternative outcomes.[26] In vertebrates, non-model species further underscore this variability; catsharks (Scyliorhinus spp.) form flat gastrulation discs with crescent-shaped mesoderm ingressions adapted to large yolk reserves, differing from the ring-like mesoderm and involution in amphibians or ingression in teleosts.[79] Amniote reptiles employ bi-modal strategies, combining primitive streak-like ingression with yolk-constrained adaptations, challenging direct homology to avian or mammalian streaks.[26] Among mammals, rabbits and humans deviate from mice in gastrulation tempo and signaling: human primitive streak mesoderm requires SNAI2 expression and MEK-dependent epithelial-mesenchymal transition (EMT), absent in rodents, while somitogenesis periodicity differs (e.g., ~3 hours in humans versus ~2 hours in mice) due to variations in HES7 protein stability.[80] These observations pose technical challenges, including opaque embryos hindering live imaging, protracted developmental timelines, and paucity of genetic tools like CRISPR transgenics, which limit functional validation of mechanisms in species such as lampreys or bichirs.[26] Conceptually, non-bilaterian non-models like ctenophores and cnidarians lack a distinct mesoderm layer, with endoderm and ectoderm often arising via delamination or polyclism rather than canonical gastrulation ingressions, fueling debates on germ layer homology and the evolutionary origins of triploblasty.[81] Such diversity implies that yolk geometry, signaling thresholds (e.g., FGF/BMP gradients), and intrinsic biochemical rates drive adaptive innovations, rather than rigid conservation, necessitating broader comparative datasets to refine causal models of body plan formation.[79][80]In Vitro and Synthetic Models
Development of Gastruloids and Engineered Systems
Gastruloids are three-dimensional aggregates of pluripotent stem cells, such as mouse embryonic stem cells (mESCs) or human embryonic stem cells (hESCs), engineered to self-organize and recapitulate key aspects of post-implantation mammalian gastrulation, including symmetry breaking, primitive streak formation, and germ layer specification.[82] The first 3D gastruloids were generated in 2014 by aggregating approximately 300 mESCs in low-adherence conditions, yielding structures that elongated along an anterior-posterior axis within 5-7 days under serum-free media supplemented with N2B27 and minimal growth factors to promote self-organization without exogenous morphogens.[83] Concurrently, two-dimensional engineered models emerged using micropatterned substrates to confine hESCs to circular adhesive islands (0.5-1 mm diameter) coated with extracellular matrix, enabling patterned differentiation driven by uniform BMP4 exposure, which induced brachyury-expressing primitive streak-like cells at colony edges and subsequent germ layer patterning.[84] Protocols for 3D gastruloid generation typically involve dissociating pluripotent stem cells into single-cell suspensions via trypsin or Accutase, followed by plating at densities of 200-500 cells per aggregate in U-bottom 96-well low-attachment plates to form embryoid body-like structures.[85] Culture proceeds in defined media activating endogenous signaling pathways—such as WNT, BMP, NODAL, and FGF—often starting with Activin A and WNT agonists for primitive streak induction, transitioning to BMP4 for mesendoderm specification, with aggregates reaching 500-1000 μm in size by day 4-5 and exhibiting lumen formation and axial elongation by day 7.[86] For human models, similar aggregation uses hESCs or induced pluripotent stem cells (iPSCs) under feeder-free conditions, with ethical guidelines limiting progression beyond 14 days or implantation potential.[87] Engineered enhancements refine these systems for spatiotemporal control, incorporating micropatterned polydimethylsiloxane (PDMS) substrates or bioengineered microwell arrays to impose geometric constraints that influence symmetry breaking and tissue morphogenesis.[88] Microfluidic devices deliver precise morphogen gradients, mimicking in vivo signaling asymmetries, while synthetic biology tools—such as CRISPR-edited lines expressing inducible transgenes for pathway modulation—enable dissection of causal mechanisms like epithelial-mesenchymal transition during ingression.[89] Chimeric gastruloids, formed by mixing differentially labeled stem cell populations, further allow lineage tracing and perturbation studies, revealing non-cell-autonomous interactions in germ layer allocation.[90] These advancements, building on initial self-organizing aggregates, have scaled production to thousands of uniform structures for high-throughput screening, though models remain limited by absent extraembryonic support and incomplete somitogenesis.[91]Achievements and Empirical Limitations
Gastruloids, aggregates of pluripotent stem cells that self-organize to mimic early gastrulation, have successfully recapitulated key aspects of germ layer specification and patterning in mammalian models. For instance, human gastruloids derived from embryonic stem cells exhibit primitive streak-like structures, epiblast cavitation, and differentiation into the three germ layers within 72-96 hours post-aggregation, enabling high-throughput screening of signaling pathways such as Wnt and Nodal.[92] Recent protocols incorporating retinoic acid pulses have induced posterior axial elongation and somitogenesis-like oscillations, producing trunk-like structures with segmented mesoderm by day 5-7, thus modeling post-gastrulation morphogenesis.[93] These systems facilitate genetic perturbations, revealing causal roles for metabolites like mannose in mesoderm induction, as demonstrated in large-scale metabolomic analyses of mouse gastruloids.[94] Engineered variants, including vascularized gastruloids, have further advanced modeling of tissue interactions, such as cardiac specification and primitive vascular networks emerging by day 4-6 in human systems.[95] [96] Microraft array technologies have scaled gastruloid production to thousands per experiment, allowing image-based phenotyping and sorting for aberrant morphologies, which supports quantitative studies of developmental robustness and genetic variants.[97] Such platforms have quantified spatiotemporal gene expression, confirming self-organization driven by diffusible morphogens without external scaffolds, thereby validating intrinsic cellular programs in vitro.[98] Despite these advances, gastruloids exhibit empirical limitations in faithfully replicating in vivo gastrulation dynamics. Basic models lack extraembryonic tissues, resulting in incomplete anterior-posterior axis formation and absent structures like the anterior visceral endoderm, which restricts modeling of full body plan establishment.[92] Stochastic self-organization leads to morphological variability, with size-dependent disruptions in patterning—gastruloids exceeding 500 μm often fail to sustain coordinated cell migrations, yielding fragmented rather than elongated axes.[99] They do not incorporate systemic cues, such as maternal circulation or immune modulation, limiting insights into peri-implantation interactions and vascular integration beyond rudimentary sprouts.[95] Metabolic perturbations, while informative, produce pleiotropic effects that confound pathway-specific causality due to the absence of compartmentalized niches.[94] Furthermore, human gastruloids halt progression around Carnegie stage 8-10 equivalents, failing to transition to organogenesis without additional engineering, underscoring gaps in sustaining long-term multicellular coordination.00920-4) These constraints highlight that while gastruloids excel in dissecting molecular mechanisms, they underrepresent the biomechanical and environmental feedbacks essential for holistic embryogenesis.Recent Advances and Future Directions
Spatiotemporal Mapping and Mechanochemical Insights
Recent advances in spatiotemporal mapping of gastrulation have integrated spatial transcriptomics, single-cell RNA sequencing, and computational alignment tools to reconstruct cellular trajectories across embryonic stages. In mouse models, a 2023 study generated a single-cell resolution spatio-temporal transcriptome of germ-layer populations during gastrula stages, revealing dynamic gene expression patterns tied to cell positioning and migration. Building on this, a 2025 atlas combined spatial and temporal coordinates to map cellular diversity in mouse embryogenesis from gastrulation onward, enabling queries into lineage progression and tissue organization. Similarly, tools like moscot have facilitated alignment of 1.7 million cells across 20 time points in mouse embryos, reconstructing developmental trajectories with high fidelity and highlighting conserved spatiotemporal motifs in germ layer formation. These mappings underscore the precision of gastrulation, where epiblast cells ingress in a temporally orchestrated manner to form mesoderm and endoderm, driven by gradients of signaling molecules like BMP and Nodal.[100]00818-6)[101] Human gastrulation mapping has progressed with 3D reconstructions and spatial transcriptomics of intact embryos. A 2024 analysis of a gastrulating human embryo provided cellular and molecular details of primitive streak formation and germ layer specification, identifying key transcription factors such as SOX17 and GATA6 in endoderm progenitors. In 2025, spatial transcriptomics of a Carnegie stage 7 human embryo resolved single-cell resolution maps, correlating positional data with gene modules for mesendoderm differentiation and highlighting species-specific timings relative to mouse models. Live imaging complemented these efforts; for instance, 2025 work described evolving mechanical properties in embryonic tissues via spatio-temporal quantification, revealing rapid transitions in cell stiffness and contractility during invagination and convergence. Such data empirically link spatiotemporal dynamics to causal drivers, including oscillatory signaling in primitive streak elongation observed through time-lapse microscopy.00357-X)[102][103] Mechanochemical insights elucidate how physical forces interplay with biochemical cues to orchestrate gastrulation movements. A 2023 computational model integrated cell adhesion, cortical tension, and chemotactic signaling to recapitulate primitive streak ingression and convergence-extension flows in chick embryos, predicting outcomes under perturbations like altered BMP gradients. This aligns with experimental evidence that mechanical feedback, such as myosin-driven contractility, coordinates epiblast cell behaviors during epithelial-to-mesenchymal transition (EMT), a core mechanochemical process enabling delamination. Recent 2025 modeling advances in avian systems further detailed primitive streak formation, showing how differential adhesion and tissue viscosity generate self-organizing flows independent of global gradients, validated against live-imaging data. In luminal epithelia analogous to gastrulating tissues, mechanochemical patterning via actomyosin pulses localizes organizers, suggesting conserved mechanisms across vertebrate gastrulation modes. These models emphasize causal realism: chemical signals initiate but mechanical properties amplify and stabilize morphogenetic patterns, with empirical perturbations confirming bidirectional feedbacks between tension and gene expression.[104][105][106]Implications for Human Development and Disorders
Disruptions in human gastrulation, which occurs during the third week of embryonic development and establishes the trilaminar germ layers and body axes, can result in early embryonic lethality or severe congenital malformations involving multiple germ layers. Abnormalities such as failed primitive streak formation or defective cell migration have been linked to conditions including conjoined twinning, chordomas, and caudal dysgenesis syndromes like sirenomelia, where perturbations in preimplantation and gastrulation processes lead to incomplete axial elongation and fusion anomalies.[107][108] Currarino syndrome, characterized by sacral agenesis, presacral masses, and anorectal malformations, exemplifies a gastrulation-related disorder tied to mutations in the MNX1 gene (formerly HLXB9), which encodes a transcription factor critical for caudal development and primitive streak function.[108] Genetic mutations affecting gastrulation regulators further underscore these implications, often manifesting as multisystem defects due to impaired organizer signaling or epithelial-mesenchymal transitions. For instance, ZIC2 mutations cause holoprosencephaly by disrupting mid-gastrulation organizer activity, leading to arrested anterior neural development and midline facial anomalies.[109] Similarly, NIPBL haploinsufficiency in Cornelia de Lange syndrome alters gastrulation-stage gene expression, phenocopying limb, craniofacial, and growth defects observed in affected individuals.[110] Expanded HTT alleles associated with Huntington's disease impair epiblast patterning during gastrulation, detectable as early as two weeks post-fertilization in embryonic models, potentially contributing to later neurodegenerative pathology through disrupted early cell fate decisions.[111] These gastrulation-linked disorders highlight the process's vulnerability to genetic and environmental insults, with implications for miscarriage rates—estimated at 10-20% of clinically recognized pregnancies often tracing to peri-implantation failures extending into gastrulation—and broader developmental screening. Advances in stem cell-derived gastruloid models enable spatiotemporal dissection of these defects, revealing mechanochemical failures that inform non-invasive prenatal diagnostics and potential regenerative interventions, though empirical limitations persist in replicating human-specific axis formation.[112][113]References
- https://en.wiktionary.org/wiki/gastrulation