Recent from talks
Nothing was collected or created yet.
Campylobacterales
View on Wikipedia
| Campylobacterales | |
|---|---|
| |
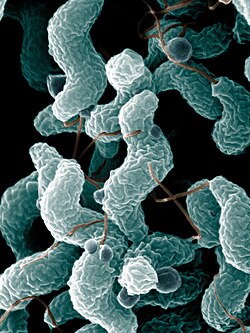
| Campylobacter jejuni bacteria | |
| Scientific classification | |
| Domain: | Bacteria |
| Kingdom: | Pseudomonadati |
| Phylum: | Campylobacterota |
| Class: | "Campylobacteria" Waite et al. 2017 |
| Order: | Campylobacterales Garrity et al. 2006 |
| Families and genera | |
| |
The Campylobacterales are an order of Campylobacterota which make up the epsilon subdivision, together with the small family Nautiliaceae. They are Gram-negative. Most of the species are microaerophilic.[1]
Molecular signatures
[edit]Comparative genomic analysis has led to the identification of 49 proteins which are uniquely found in virtually all species of the order Campylobacterales. Additionally, two conserved signature indels have been identified which, along with the proteins, serve as molecular markers for the order. The indels are a three-amino-acid insertion in the B protein of the Uvr ABC system, and a two-amino-acid deletion in phenylalanyl-tRNA synthetase.[2]
Phylogeny
[edit]The currently accepted taxonomy is based on the List of Prokaryotic names with Standing in Nomenclature (LPSN)[3] and National Center for Biotechnology Information (NCBI)[4]
| 16S rRNA based LTP_10_2024[5][6][7] | 120 marker proteins based GTDB 09-RS220[8][9][10] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
Unassigned genera:
- "Flexispira" Bryner 1987
- "Candidatus Thioturbo" Muyzer et al. 2005
- "Thiofractor" Makita et al. 2012
- "Thiobarbaceae" Assié et al. 2020
- "Candidatus Thiobarba" Assié et al. 2020
See also
[edit]References
[edit]- ^ Garrity, George M.; Brenner, Don J.; Krieg, Noel R.; Staley, James T. (eds.) (2005). Bergey's Manual of Systematic Bacteriology, Volume Two: The Proteobacteria, Part C: The Alpha-, Beta-, Delta-, and Epsilonproteobacteria. New York, New York: Springer. ISBN 978-0-387-24145-6.
- ^ Gupta, R. S. (2006). Molecular signatures (unique proteins and conserved indels) that are specific for the epsilon proteobacteria. BMC Genomics. 7:167.
- ^ A.C. Parte; et al. "Campylobacterales". List of Prokaryotic names with Standing in Nomenclature (LPSN). Retrieved 2023-09-09.
- ^ Sayers; et al. "Campylobacterales". National Center for Biotechnology Information (NCBI) taxonomy database. Retrieved 2023-09-09.
- ^ "The LTP". Retrieved 10 December 2024.
- ^ "LTP_all tree in newick format". Retrieved 10 December 2024.
- ^ "LTP_10_2024 Release Notes" (PDF). Retrieved 10 December 2024.
- ^ "GTDB release 09-RS220". Genome Taxonomy Database. Retrieved 10 May 2024.
- ^ "bac120_r220.sp_labels". Genome Taxonomy Database. Retrieved 10 May 2024.
- ^ "Taxon History". Genome Taxonomy Database. Retrieved 10 May 2024.

